A Follow-Up Study Health Risk Assessment of Heavy Metal Leachability from Household Cookwares
Muyiwa Ogidi1, MKC Sridhar2* and AO Coker2
1Department of Environmental Health Sciences, Faculty of Public Health, College of Medicine, University of Ibadan, Ibadan, Nigeria
2Department of Civil Engineering (Public Health and Environmental Engineering), Faculty of Technology, University of Ibadan, Ibadan, Nigeria
- *Corresponding Author:
- MKC Sridhar
Department of Environmental Health Sciences
Faculty of Public Health, College of Medicine
University of Ibadan, Ibadan, Nigeria.
Tel: 08037273836
E-mail: mayucares@gmail.com
Received date: October 17, 2017; Accepted date: November 02, 2017; Published date: November 09, 2017
Citation: Ogidi MA, Sridhar MKC, Coker AO (2017) A Follow-Up Study Health Risk Assessment of Heavy Metal Leachability from Household Cookwares. J Food Sci Toxicol Vol.1 No.1:3
Abstract
Leaching of heavy metals from household cookware’s can be a public health concern. Therefore, in this present study, some selected heavy metals namely: iron (Fe), aluminum (Al), zinc (Zn), lead (Pb), chromium (Cr) and nickel (Ni) were analyzed using atomic absorption spectrophotometery in beans and tomatoes sauce cooked in aluminum and cast-iron cookware’s commonly used in Nigeria.
These metals were also determined in uncooked tomatoes sauce, and beans used as controls. Results obtained indicated that, the lowest concentrations of most of the metals were recorded in uncooked food items exceptions were that of Pb,Ni (tomatoes sauce only) and Zn (tomatoes sauce and beans). It was also noticed from results recorded that, the highest mean concentration of Iron (Fe) (1068
mg/kg) was obtained in tomatoes sauce cooked in cast iron pot, Aluminum (Al) recorded its highest mean concentration (850 mg/kg) in tomatoes sauce cooked in aluminum pot. Chromium (Cr) recorded the highest mean concentration (9.4mg/kg) in tomatoes sauce cooked in aluminum pot. Lead (Pb), Zinc (Zn) and Nickel(Ni) recorded its highest mean concentrations (31 mg/Kg), (22.6 mg/kg) and (11.9
mg/Kg) respectively in uncooked tomatoes sauceResults obtained in this study further revealed that, cooking utensils can leach some quantities of trace metals into food during processing, hence resulting in slight increase in the concentration of some of these metals in processed foods. The general results showed that concentration levels of Lead (Pb), Nickel (Ni) and Zinc (Zn) obtained were within
their recommended safe limits whereas, concentrations levels of Aluminum (Al), Iron (Fe) and Chromium (Cr) were above their maximum acceptable levels.The study has also shown that tomatoes sauce and beans foodstuffs have high potentials of leaching these metals from cookwares possibly due to pH and contact time. These results were discussed based on their environmental and
health implications. The results show that the aluminum cookware poses a greater danger to public health when compared to cast iron cookware by approximately 1.7-folds through tomatoes sauce and 1.6-folds through beans.
Keywords
Heavy metals; Leaching; Cookware; Beans; Tomato sauces
Introduction
Most people are aware of air pollution, water pollution and the dangers of household chemicals. Studies are now showing that certain cookware can also be contributing toxic pollutants to our bodies. Pots, pans and other cookware are made from a variety of materials. These materials can enter the food that we cook in them. There are countless types of cookware, made of cast iron, aluminum, copper, ceramic and enamel, glass, stainless steel, etc. The most commonly used cooking pots in Nigeria, are those made of aluminum, cast iron and stainless steel. The choice of these pots is because they are the most popular and economical and commonly found in most Nigerian markets in addition to the fact that they are easy to clean, have unique surfaces that cannot crack easily, difficult to rust and high durability.
According to International Occupational Safety and Health Information Centre (CIS), there are 35 metals that are of concern to humans due to occupational or residential exposure and it has been estimated that around one billion people worldwide suffer from some form of diseases attributed to those metals [1]. Regardless of their effect, many of them play a crucial role in all life forms. For example, arsenic (As), copper (Cu), iron (Fe) and nickel (Ni) are considered essential at low concentrations but are toxic at high levels. However, elements like aluminum (Al), beryllium (Be) and lead (Pb) have no biological significance [2].
Although metal toxicity depends on the amount ingested, chronic exposure to certain metals, such as cadmium (Cd) and lead (Pb), can cause severe toxic effects, even in low amounts. Humans are exposed to metals through different exposure pathways, the most common being inhalation of contaminated air and ingestion of products such as water, medicinal herbs, and food [3,4]
Metals tend to bioaccumulate thus their concentration increases in a biological system over time. This is because they are stored faster than being metabolized or excreted [5]. Unlike organic molecules, metals do not require bioactivation or undergoes enzymatic modification that produces a reactive chemical species for detoxification process [6]. However, metals use other mechanisms, such as long-term storage (e.g. Iron) and biliary and/or urinary excretion [6]. This paper reports the selected metal leachability from common cooking pots and the processes practices by a cross section of Nigerian population.
Materials and Methods
Study site
The study was carried out in Apete, one of the major towns in Iddo Local Government of Ibadan, Oyo State, Nigeria, which has an estimated population of 103,261 as at the 2006 census. It is a residential area with mostly low-income earners. It lies between latitude 07°27′35′′N of the Equator and longitudes 03°53′04′′E of the Greenwich Meridian. One of the most striking lifestyles of the people of Apete is that; people work from dawn to dusk and enjoy a sumptuous meal on return home. Aluminum and castiron cookwares are mostly used for cooking at home due to the socio-economic status of the residence.
Sampling procedure
Cookware: Two kinds of cookware were selected for this research work: Aluminum and Cast iron. These are the common cookware used in households of the study area. The cookware samples were rinsed with clean water to remove dirt because of their newness before using them.
Food items: Tomatoes and beans were used for this research work. Beans were chosen because of its long cooking time (contact time), but also high cooking temperature. Tomato sauce was chosen because of its low pH value.
Two portions of 500 g each of the beans and tomatoes samples were weighed and cooked in an aluminum pot and cast-iron pot for 120 min and 30 min, respectively, using a gas stove. Samples were collected and recorded as (Day 1 samples). Subsequently, the remaining food items were stored in the pot for two more days while reheating to reproduce the typical household method of preserving food before additional meals. The reheating was repeated four times for 15min for beans and 5 min for the tomatoes sauce on each occasion. After which another set of samples were taken and recorded as (Day 2 samples). The samples were then oven dried at 65-70°C to a constant weight. After drying, the dry mass and % moisture content of the food sample was determined using the following equation:

Where;
W1=weight of container (g),
W2=initial weight of container with food sample,
W3=final weight of container with sample
Digestion of samples
Samples were digested according to the method described by the Association of Official Analytical Chemists [7]. 2 g of soft samples were weighed using an analytical balance and placed into acid washed digestion tubes (PTFE-TFM liners) onto which 16 ml HNO3and 4 ml H2O2 were added before being placed into the microwave digester. The digested samples were cooled, and individually filtered through a Whatman No. 1 filter paper into a 50 mL standard flask and made up to the mark with distilled water. The extracts were transferred quantitatively into preheated plastic sample bottles, tightly closed, and kept in a refrigerator at a temperature around 4°C. A blank digest was carried out in the same way.
Analysis of heavy metals
Concentrations of Al, Pb, Ni, Fe, Zn and Cr in the filtrate of digested food samples were estimated by using an atomic absorption spectrophotometer. The instrument was fitted with specific lamp of metal. The instrument was calibrated using manually prepared standard solution of respective heavy metals as well as drift blanks. Standard stock solution of 1000 mg/l for all the metals was obtained from University of Ibadan Central Research Laboratory, Nigeria. These solutions were diluted for desired concentrations to calibrate the instrument.
Data analysis: The statistical analysis of the results was done using data analysis tool in Microsoft Excel 2013.
Daily intake of metal: For preliminary risk assessment of consumer exposure to heavy metals, the average metal content in each food item was calculated and multiplied by the respective consumption rate. The daily intake rate was determined by the following equation [8,9]:
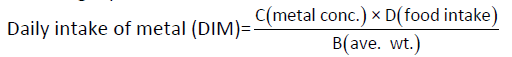
Where 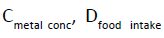 and
and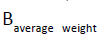 represent the heavy metal concentrations in food (mg/Kg), daily intake of food and average body weight, respectively. The required amount of vegetables in our daily diet must be 300 to 350 g/person has been suggested by WHO guideline [10]. Daily intake of 200 g of beans was used in this study. The average body weight
represent the heavy metal concentrations in food (mg/Kg), daily intake of food and average body weight, respectively. The required amount of vegetables in our daily diet must be 300 to 350 g/person has been suggested by WHO guideline [10]. Daily intake of 200 g of beans was used in this study. The average body weight 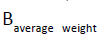 was taken as 60 kg for adults according to [11].
was taken as 60 kg for adults according to [11].
Health risk index (HRI)
Health risk index for the locals (consumers) through the consumption of contaminated food was assessed by the ratio of daily intake of metal (DIM) to the oral reference dose (RfDo) for each metal [12]. If the value of HRI is less than 1, then the exposed local population (consumers) is said to be safe, if HRI is equal to or higher than 1, is considered as not safe for human health, therefore potential health risk occurred, and related interventions and protective measurements should be taken [12]. An estimate of risk to human health through consumption of food was calculated by the following equation:
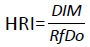
Where, RfDo is the oral reference dose. RfDo is an estimate of a daily oral exposure for the human population, which does not cause deleterious effects during a lifetime [13].
Hazard index (HI)
To estimate the risk to human health through more than one heavy metal (HM), the hazard index (HI) has been developed. The hazard index is the sum of the hazard quotients for all heavy metals, which was calculated by the following equation [14].
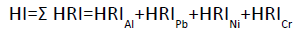
It assumes that the magnitude of the adverse effect will be proportional to the sum of multiple metal exposures. It also assumes similar working mechanisms that linearly affect the target organ. Iron (Fe) and Zinc (Zn) are essential elements for human health, the hazard index for the toxic elements Chromium (Cr), Aluminum (Al), Nickel (Ni), and Lead (Pb) are thus calculated in this study.
Results and Discussion
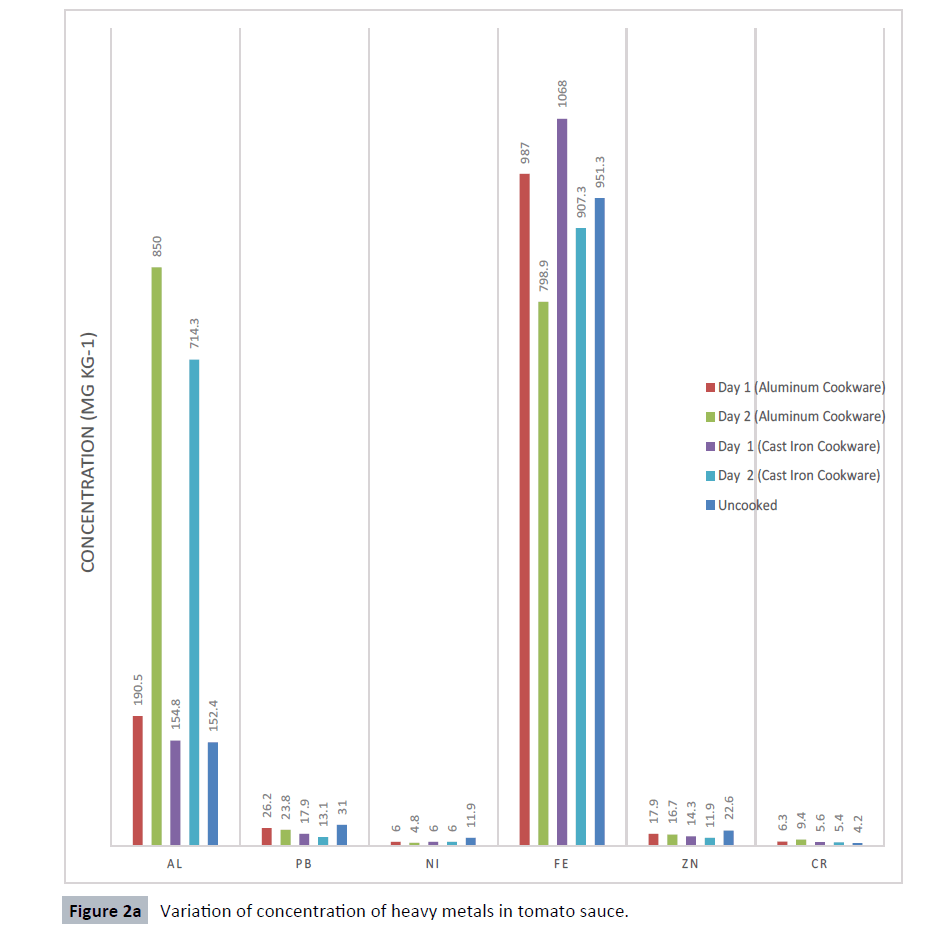
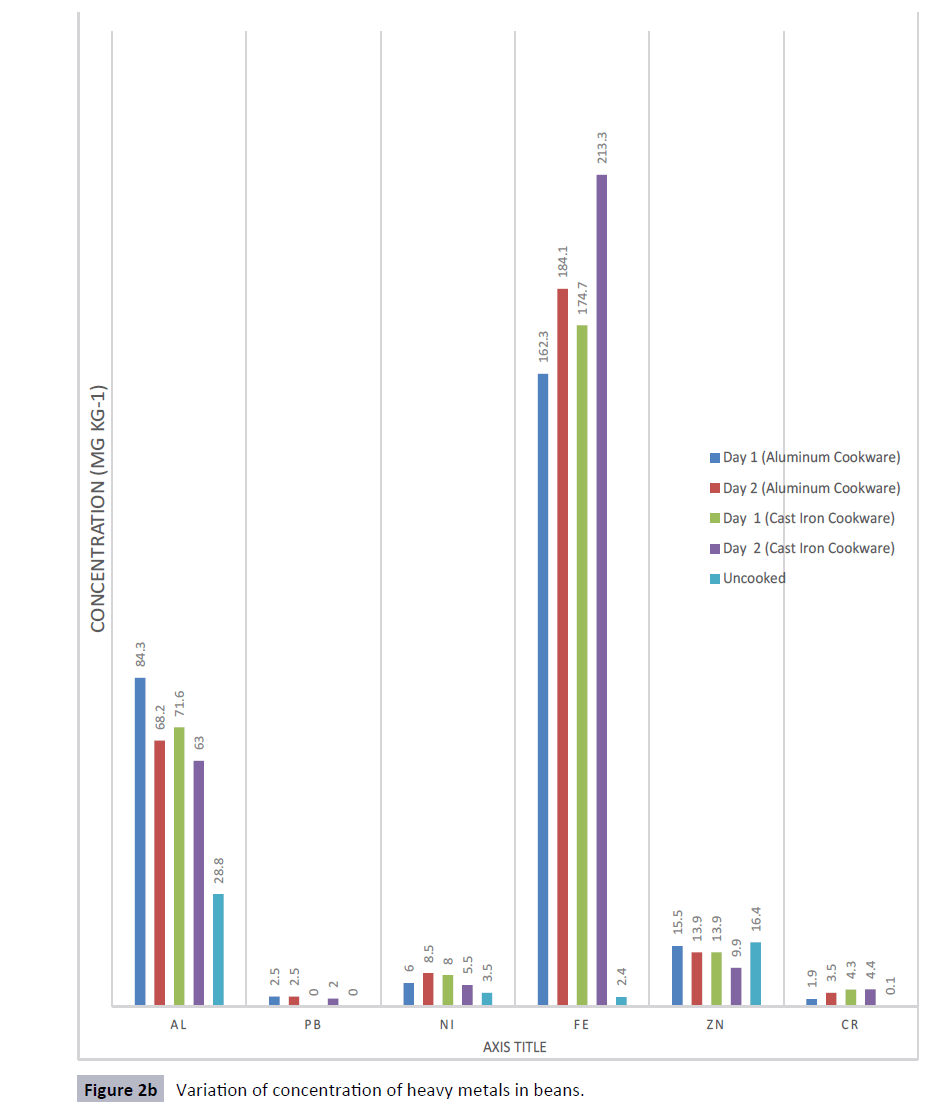
The pH level of samples was obtained as shown in Table 1. Heavy metals concentrations is shown for the tomatoes sauce and beans cooked in aluminum and cast-iron cookware (Figures 1 and 2). Results obtained revealed that the different food items studied recorded variable levels of heavy metals. Maximum concentration of Al was found in tomatoes sauce cooked in aluminum pot (850 mg kg-1) and the minimum concentration was found in beans cooked in cast iron pot (63.0 mg kg-1). Maximum concentration of Pb was found in tomatoes sauce cooked in aluminum pot (26.2 mg kg-1) and the minimum concentration was found in beans cooked in cast iron pot (2.0 mg kg-1). For Ni, the maximum concentration was found in beans cooked with aluminum pot (8.5 mg kg-1) and the minimum concentration was found in beans cooked with cast iron pot (5.5 mg kg-1). Maximum concentration of Fe was found in tomatoes sauce cooked with cast iron pot (1068 mg kg- 1) and the minimum was found in beans cooked with aluminum pot (162.3 mg kg-1). Maximum concentration of Zn was found in tomatoes sauce cooked in aluminum pot (17.9 mg kg-1) and the minimum was found in beans cooked with cast iron pot (9.9 mg kg-1). For Cr, the maximum concentration was found in tomatoes sauce cooked in aluminum pot (9.4 mg kg-1) and the minimum was found in beans cooked in aluminum pot (1.9 mg kg-1).
| Tomatoes Sauce | Beans | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Uncooked | Al Pot (Day 1) | Al Pot (Day 2) | Fe Pot (Day 1) | Fe Pot (Day 2) | Uncooked | Al Pot (Day 1) | Al Pot (Day 2) | Fe Pot (Day 1) | Fe Pot (Day 2) | |
| pH | 3.75 | 4.41 | 4.38 | 4.46 | 4.4 | 7.11 | 7.92 | 7.32 | 7.5 | |
Table 1: Variation in pH of samples.
Figure 2a:Variation of concentration of heavy metals in tomato sauce.
Aluminum solubility increases at a pH lower than 4.5 and above 8.5 [15]. The pH values of the second day tomatoes sauce were 4.3; this can be attributed to the high value of Al metals recorded. Leaching of metals under the preparation of lower pH (4.25) food in an aluminum cooking pot increase the concentration of Al metal by 20.3 mg/kg [16]. The results recorded for Al indicated that, the metal was leached from the cookware into the food item cooked. While acids give the highest figures of aluminum intake in cooked foodstuffs, alkaline foodstuffs (less common) and foodstuffs with much added salt, do increase the aluminum uptake [17,18]. This can be attributed to the lower level of aluminum recorded in the beans samples of both aluminum pots and cast-iron pots when compared with the tomatoes sauces (acidic foodstuff) samples. The longer the cooking time, the greater the accumulation of aluminum and about 3.5 mg/day quantity of Al is added to the daily diet using aluminum pot for food processing [19]. Aluminum is excreted by the kidneys, and only a small amount of aluminum is absorbed [20]. However, soluble aluminum salts are more easily absorbed.
The results obtained, indicates a reduction in the concentration level of lead in the cooked tomatoes sauce compared to the uncooked. This reduction may be attributed to the low pH of tomatoes sauce. Lead resists attack by many acids, including sulfuric acid [21]. Lead was not detected in the uncooked samples of beans. This may be attributed to the fact that, lead in the soil is only poorly taken up by plant roots and is not transported away from the roots to the rest of the plant crop, particularly the seeds. Therefore, lead levels in the plant are to a large extent governed by air-borne lead contamination which makes leaves and leafy vegetables most vulnerable to the air-borne deposition. Lead is a classical chronic or cumulative poison. In humans, lead can result in a wide range of biological effects depending upon the level and duration of exposure. Health effects are generally not observed after a single exposure. Lead poisoning effect includes; hematological effects, neurological and behavioral effects, renal effects, cardiovascular effects, and effects on the reproductive system.
Nickel is probably an essential element for biological organisms [22], even though nickel deficiency has not been demonstrated in human beings [21]. Nickel is found in small quantities in many foodstuffs (0.001-0.01 mg/kg) and in higher concentrations in foodstuffs such as grains, nuts, cocoa products and seeds (up to 0.8 mg/kg) [23]. The increase in level of nickel from the uncooked tomatoes sauce to the cooked one is attributed to leaching of the metal from the cookwares. This is consistent with the findings of several researchers that, some quantities of nickel are always transferred into foodstuffs cooked [24-27]. The daily intake of nickel via foodstuff is estimated at 0.15-0.7 mg/day [26]. Food intake and gastric emptying are of substantial significance for the bioavailability of nickel from aqueous solutions. The absorption of free nickel ions released in the gastrointestinal tract may be 40 times higher than that of complex-bound nickel from foodstuff [28]. However, some patients with certain types of nickel dermatitis may get a flare-up of eczema through ingestion of even small amounts of orally ingested nickel, e.g. from foodstuffs rich in nickel or foodstuff or drinks contaminated via nickel containing materials [29].
Iron is an essential trace metal. The high level of iron content in the cast iron pot may be attributed to the fact that in cast iron alloy, iron constitutes more than fifty per cent as ferrous alloy with 3-4.5 wt% C [30]. The high level of iron in the tomatoes sauce show that leaching of iron is promoted in acidic medium with pH less than 4.5. Iron leaching is dependent on the acidity, moisture content and cooking time of the food preparation. More acidic, moister foods and foods cooked longer have higher iron content than controls when cooked with an iron ingot or pot [31,32]. Ingestion of soluble iron salts by children in doses exceeding 0.5 g of iron can give rise to severe lesions in the gastrointestinal tract, followed by metabolic acidosis, shock and toxic hepatitis [30].
Zinc is an essential trace metal. The results obtained indicated reduction in the concentration level of zinc in the cooked tomatoes sauce and beans. This reduction is more significant in cast iron pot than the aluminum pot. This reduction can be attributed to coating of iron and steel with zinc and to prevent corrosion, which is the most important use of zinc [21]. Zinc protects iron from rusting because it is the stronger reducing agent of the two metals [21]. The required daily intake of zinc for adults is about 15 mg/day and the requirement varies with age [20]. The average daily intake of Zn is 15-20 mg/day [11]. In human’s high levels of zinc cause acute effects such as vomiting and gastrointestinal irritation (nausea, cramps, diarrhea), however when bound to food components (i.e., meat, oysters) these effects are expected to be less. No information is available on toxic effects in man due to chronic excessive intake of zinc; however impaired copper uptake in humans has been noted following the chronic elevated intake of zinc. Some effects of zinc therefore may be secondary to impaired copper utilization (i.e., anemia).
Chromium is an essential nutrient required by the human body to promote the action of insulin in the body tissue hence helping in the utilization of glucose, protein and fat especially if within allowable limit [33,34]. Studies have shown that, most foodstuffs contain less than 0.1 mg Chromium per kg [35]. There is no significant difference in the level of chromium leached from both cookware used in this study. These results show that consuming acidic foodstuff cooked with both pot aluminum and cast-iron pot may cause chromium toxicity. Toxic aspects of chromium are related to Cr (VI) [35], due to its high absorption, easy penetration of the cell membranes and its genotoxicity and oxidising properties.
Health risk assessment
Daily intake of metal (DIM) calculated in food cooked in both aluminum and cast-iron cookware examined are above the acceptable Daily intake of metal for Al, Fe and Cr set by FAO/ WHO. While that of Pb, Ni and Zn where below the acceptable standards. The summary of daily intake of metal in food samples and acceptable daily intake are presented in Table 2.
| Cookware | Daily intake of metals in tomatoes sauce | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Al | Pb | Ni | Fe | Zn | Cr | ||||||
| Aluminum | 0.95 | 0.12 | 0.02 | 3.99 | 0.08 | 0.03 | |||||
| Cast Iron | 0.77 | 0.07 | 0.03 | 4.54 | 0.06 | 0.03 | |||||
| WHO/FAO | 0.14 | 0.13 | 0.7 | 0.8 | 1 | 0 | |||||
| Daily intake of metals in beans | |||||||||||
| Aluminum | 0.254 | 0.008 | 0.024 | 0.577 | 0.049 | 0.009 | |||||
| Cast Iron | 0.224 | 0.003 | 0.023 | 0.935 | 0.040 | 0.015 | |||||
| WHO/FAO | 0.14 | 0.13 | 0.7 | 0.8 | 1 | 0 | |||||
Table 2: Daily intakes of metals (DIM) in tomatoes sauce.
The highest DIM values were found in tomatoes sauce cooked in cast iron pot and the lowest were found in beans cooked with aluminum pot. It was observed generally that, the DIM values in aluminum pots were higher than that of cast iron pot, except for Fe and Cr that shows slight higher value in cast iron pot.
| Cookware | Health Risk Index (HRI) | |||||
|---|---|---|---|---|---|---|
| Tomatoes sauce | ||||||
| Al | Pb | Ni | Fe | Zn | Cr | |
| Aluminum | 0.95 | 29.75 | 1.2 | 5.71 | 0.28 | 0.021 |
| Cast iron | 0.77 | 16.38 | 1.5 | 6.48 | 0.20 | 0.018 |
| Beans | ||||||
| Aluminum | 0.25 | 2.08 | 1.20 | 0.82 | 0.16 | 0.006 |
| Cast iron | 0.22 | 0.83 | 1.13 | 1.34 | 0.13 | 0.0096 |
Table 3: Health risk index (HRI) for heavy metals in tomatoes sauce and beans.
The health risk assessment associated with heavy metals (Al, Pb, Ni, Fe, Zn, and Cr) leaching of cookware, estimated exposure and risk index were calculated as shown in Table 3. The result revealed that, Health risk index for Al, Zn and Cr for both cookware leaching studied are lower than 1 indicating safe for the consumer. However, Pb, Ni and Fe were found to show HRI value greater than 1. But the value of Pb in cast iron cookware and Fe in aluminum cookware of beans samples both recorded a HRI value lower than 1. This result indicates the possibility of accumulation of Pb, Ni and Fe above the required level in the body of consumer using these cookwares.
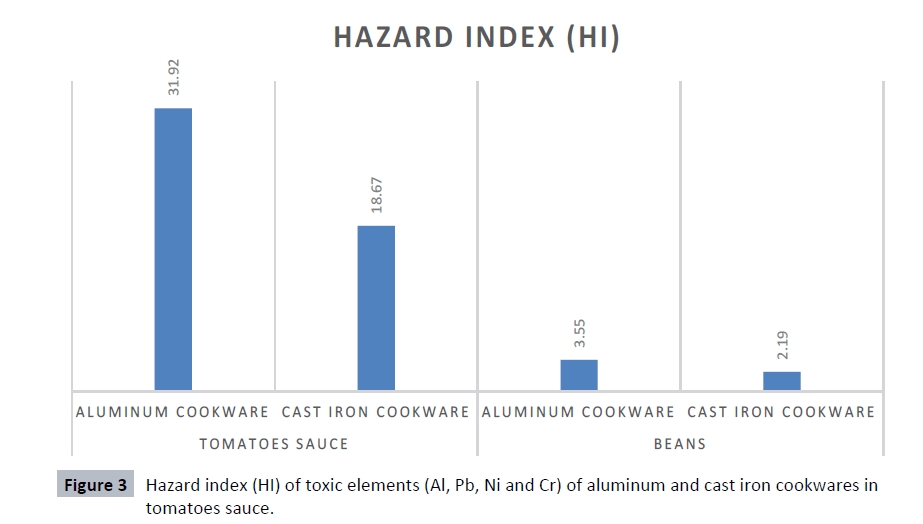
Hazard index
The Hazard Index (HI) has been calculated for the leaching of toxic element Al, Pb, Ni, and Cr, in aluminum and cast iron cookwares used for tomatoes sauce and beans. The sequence of HI followed the decreasing order from aluminum pot to cast iron pot. Both cookwares studied under tomatoes sauce and beans, recorded a Hazard Index (HI) value greater than 1. When the hazard index exceeds 1.0, there is concern for potential health effect [36]. The results as shown in Figure 3 indicate that, the HI value of tomatoes sauce cooked in aluminum cookwares has a higher HI value than that of tomatoes sauce cooked in cast iron cookware. Similarly, the beans cooked in aluminum cookwares have a higher HI value than the beans cooked in cast iron cookware.
Conclusion
The results obtained in the study have revealed that different foods have different potential of accumulating trace metals depending on the pot used, the type of food and the process applied. Also, the study has shown that cooking utensils like aluminum and cast-iron pots which are usually used in food processing in Apete have a positive impact on the metal transfer into the foodstuffs.
The DIM and HI values for the metals indicated that the population is more exposed to the toxic substances present in food substances which are frequently eaten by the population. This is because the values obtained from these indices with reference to the food samples exceeded the permissible limit [36]. The results show that the aluminum cookware poses a greater danger to public health when compared to cast iron cookware by approximately 1.7-folds in tomatoes sauce and 1.6-folds in beans. The assessments of metals in the cooked food with both cookwares confirm that those pots are a potential source of metals contamination of the foods and constituted a health risk for people who used them regularly. Based on the results obtained, prolonged usage of these pots in cooking and storing should be discouraged since this metal present a risk for the consumers particularly in the prolonged exposure even at low concentration.
While both cookware studied may pose health concerns, it is probably advisable to discontinue the use of aluminum cookware in preparing/processing acidic foods such as tomatoes sauce studied. Storage of food acidic foods in aluminum containers should also be discouraged to exposure of food to leaching even at low concentration.
References
- WHO (2011) World report on disability 2011. https://www.who.int/disabilities/world report/2011/en/index.html Accessed from 11 June, 2017.
- Trichet J, Defarge C (1995) Non-biologically supported organomineralization. Bulletin-Institute Oceanographique Monaco-Numero Special, pp: 203-236.
- Nowak B, Chmielnicka J (2000) Relationship of lead and cadmium to essential elements in hair, teeth and nails of environmentally exposed people. Ecotoxicol Environ Saf 46: 265-274.
- Abou-Arab AK Abou MA (2000) Heavy metals in Egyptian spices and medicinal plants and the effect of processing on their levels. J Agric Chem 48: 2300-2304.
- Hare L (1992) Aquatic insects and trace metals: bioavailability, bioaccumulation and toxicity. Crit Rev Toxicol 22: 327-369.
- Waalkes M (1995) Metal carcinogenesis. In: Goyer R.A. and C.D. Klaassen eds. Metal toxicology. New York: Academic Press, pp: 47-67.
- AOAC (Association of Official Analytical Chemists) (1980). Official Methods of Analysis of the AOAC (W. Hortwitz, ed. 13th edition) AOAC, Washington D.C. USA, pp: 129-146.
- Arora M, Kiran B, Rani S, Rani A, Kaur B, et al (2008) Heavy metal accumulation in vegetable irrigated with water from different sources. Food Chem 111: 811-815.
- Sajjad K, Robina F, Shagufta S, Aziz M, Maria S (2009) Health risk assessment of heavy metals for population via consumption of vegetables. World Appl Sci J 6: 1602-1606.
- WHO (1998) Health Guideline for the use of wastewater in agriculture and aquaculture. Report of WHO Science Group, World Health Organization, Geneva, Switzerland. Tech Rep Sci 778: 10.
- World Health Organization [WHO] (1993) Evaluation of certain food additives and contaminants. In: Forty-First Report of the joint FAO/WHO Expert Committee on Food Additives, WHO Geneva, Switzerland.
- USEPA (2013) Reference dose (RfD): Description and use in health risk assessments, Background Document 1A, Integrated risk information system (IRIS); United States Environmental Protection Agency: Washington, DC.
- USEPA (2010) Risk-based Concentration Table. United State Environmental Protection Agency, Washington, DC.
- Guerra K, Konz J, Lisi K, Neebrem C (2010) Exposure Factors Handbook. USEPA, Washington DC.
- Elinder CG, Sjogren B (1986) Aluminum. Handbook on the Toxicology of Metals 2: 1-25.
- Semwal AD, Padmashree A, Khan MA, Sharma GK, Bawa AS (2006) Science of food and agriculture 86: 24-25.
- Hughes JT (1992) Aluminum and your health. British Library Cataloguing in Publication Data, Rimes House.
- Gramiccioni L, Ingrao G, Milana MR, Santaroni P, Tomassi G (1996) Aluminum levels in Italian diets and in selected foods from aluminum utensils. Food Addit Contam 13: 767-774.
- Greger JL, Goetz W, SullivanD (1985) Aluminum levels in foods cooked and stored in aluminum pans, trays and foil. J Food Prot 48: 772-777.
- JECFA (1989) Evaluation of certain food additives and contaminants. Thirty-third report of the Joint FAO/WHO Expert Committee on Food Additives. World Health Organization, Technical Report Series, p: 776.
- Beliles RP (1994) The metals. Patty’s Industrial Hygiene and Toxicology 2 (Part C), pp: 2106-2124.
- Agency for Toxic Substances and Disease Registry (ATSDR) (1995) Toxicological profile for nickel. Draft for public comment. U.S. Department of Health and Human Services. Public Health Service. Agency for Toxic Substances and Disease Registry.
- Kumar R, Srivastava K, Srivastava SP (1994) Leaching of heavy metals (Cr, Fe and Ni) from stainless steel utensils I food stimulants and food materials. Bull Environ Contam Toxicol 53: 259-266.
- Agarwal P, Srivastava S, Srivastava MM, Prakash S, Ramanamurthy M, et al (1997) Studies on leaching of Cr and Ni from stainless steel utensils in certain acids and in some Indian drinks. Sci Total Environ 199: 271-275.
- Berg T, Petersen A (2000) The release of nickel and other trace elements from electric kettles and coffee machines. Food Addit Contam 17: 189-196.
- Codex Alimentarius Commission (1995) Doc. no. CX/FAC 96/17. Joint FAO/WHO food standards programme. Codex general standard for contaminants and toxins in foods.
- Sunderman FW Jr, Hopfer SM, Sweeney KR, Marcus AH, Most BM et al (1989) Nickel absorption and kinetics in human volunteers. Pro Soc Exp Biol Med 191: 5-11.
- Veien NK, Menné T (1990) Nickel contact allergy and a nickel-restricted diet. Semin Dermatol 9: 197-205.
- Elinder CG (1986) Zinc. In: Friberg L., Nordberg G.F., Vouk V.B. Hand book on the toxicology of metals. Second edition. Elsevier, Amsterdam, New York, Oxford.
- Brittin HC, Nossaman CE (1986) Use of iron cookware. Home Econ Res J 15: 43-51.
- Kollipara U K, Brittin HC (1996) Increased iron content of some Indian foods due to cookware. J Acad Nutr Diet 96: 508-510.
- Anderson RA, Bryden NA (1992) Dietary chromium intake. Freely chosen diets, institutional diet and individual foods. Biol Trace Elem Res 32: 117-121.
- O’flaherty EJ (1993) A physiologically based model for the ingestion of chromium (iii) and chromium (iv) by humans. Toxicol Appl Pharmacol 118: 16-29.
- Nordic Council of Ministers (1995 Risk evaluation of essential trace elements – essential versus toxic levels of intake. Report of a Nordic project group. Ed.: Oskarsson, A. Nordic Council of Ministers, Copenhagen, Denmark.
- Huang ML, Zhou SL, Sun B, Zhao QG (2008) Heavy metals in wheat grains: Assessment of potential health risk for inhabitants in Khunshan, China. Sci. Total Environ 405: 54-61.
- FAO/WHO (2000) Evaluation of certain food additives and contaminants. Geneva, World Health Organization, Joint FAO/WHO Expert Committee on Food Additives, World Health Organization Technical Report Series 859: 29-35.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences