Correlation and Path Coefficient Analyses of the Morphological Characteristics and Body Weight of Penaeus monodon (Crustacea, Decapoda, Penaeidae)
Song Jiang1,2,3, Falin Zhou2,3, Qibin Yang3, Lishi Yang2,3, Tao Li1, Shigui Jiang1,2,3, Jianhua Huang2,3*
1Shenzhen Base of South China Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences, Shenzhen 518108, P.R. China
2South China Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences, Guangzhou 510300, P.R. China
3Key Laboratory of South China Sea Fishery Resources Exploitation and Utilization, Ministry of Agriculture, Guangzhou, 510300, P.R. China
- Corresponding Author:
- Jianhua Huang
South China Sea Fisheries Research Institute
Chinese Academy of Fishery Sciences
Guangzhou 510300, P.R. China
Tel: (979)845-5650
E-mail: hjh210440@sina.com
Received date: July 31, 2017; Accepted date: August 22, 2017, 2017; Published date: August 29, 2017
Citation: Jiang S, Zhou F, Yang Q, Yang L, Tao Li, et al. Correlation and Path Coefficient Analyses ofthe Morphological Characteristics and Body Weight of Penaeus monodon (Crustacea, Decapoda, Penaeidae). Insights Aquac Cult Biotechnol 2017, 1:2.
Abstract
The acute toxicity of glutaraldehyde, dibromohydantoin, methionine iodine and bleaching powder to juvenile Pinctada maxima was studied. The results showed that the order of toxicity was bleaching powder>dibromohydantoin>m ethionine iodine>glutaraldehyde and the median lethal concentrations (LC50) of above disinfectants were 13.05 mg/L, 27.49 mg/L, 53.20 mg/L and 67.36 mg/L, respectively in 24 h and the LC50 were 10.15 mg/L, 9.35 mg/L, 29.27 mg/L and 35.58 mg/L respectively in 48 h; and their SC (safe concentration) were 1.86 mg/L, 0.32 mg/L, 2.66 mg/L and 7.52 mg/L respectively. The research indicated that we can use glutaraldehyde to disinfect P. maxima under safe concentration, but should not use bleahing powder, and can use dibromohydantoin and methionine iodine carefully.
Keywords
Penaeus monodon; Economic traits; Correlation analysis; Path analysis; Multiple regression equation
Introduction
Penaeus monodon is one of the world’s most commercially important cultured penaeid shrimp. About two decades ago, rapid development and expansion of prawn farming occurred throughout much of southeast Asia [1,2]. Since then, China has successfully used artificial breeding technology and directional selection to improve P. monodon yield [3-5]. Selective breeding is essential to increase efficiency and profitability of production, with body weight among the key selective breeding traits that drives profitability in P. monodon farming [6]. However, determining body weight is frequently difficult due to measuring locations, tools and environments, and thus morphological indicators such as body length and carapace length are often preferable and easier to obtain. Understanding the relationships between body weight and other morphological characteristics is therefore vital for selection [7].
Path coefficient analysis can be used to determine selection criteria by measuring the direct influence of one variable on another and by separating the correlation coefficient into direct and indirect effects, such as seen in several aquatic organism studies [6,8]. For example, Wang et al. [9] used path analysis on three-month-old juvenile turbot (Psetta maxima) to obtain the body weight multiple regression equation with body length, height and thickness as independent variables. Harue et al. [10] used multiple correlation analysis on farmed Red Sea carp to estimate the impact of standard length and weight on body fat content. Zhan et al. [11] used correlation, path and multiple regression analyses to establish the body weight linear regression equation for body length, head length and tail height in Paralichthys olivaceus. Debowski et al. [12] used multiple regressions to study the relationship between body fat content and body length and weight of Salmo sala. Ahmed et al. [13] used multiple correlation analysis to analyze body length and weightrelated growth parameters of fish, whales and shellfish.
Detailed path coefficient and correlation analyses can help identify the relationships between body weight and morphological characteristics in P. monodon. In particular, path coefficient analysis can separate direct and indirect effects and measure the relative importance of the potential causal factors involved [14,15]. The objectives of the current study were to: (a) determine the impact of morphological traits on Body Weight (BW); (b) estimate correlation coefficients for phenotypic characteristics between Body Length (BL), Carapace Length (CL), Carapace Width (CW), Carapace Height (CH), pleon segment lengths and BW; (c) evaluate the relative contribution of each morphological characteristic on BW using path coefficient analysis; and (d) establish the relationship between all measured morphological characteristics and BW.
Materials and Methods
Sampling and data collection
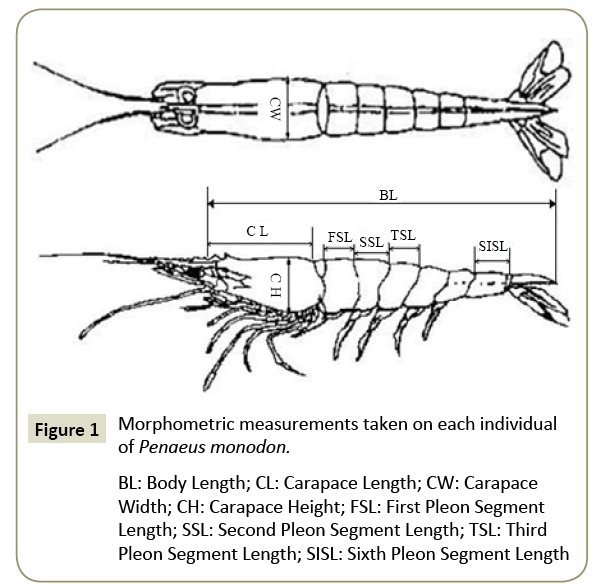
We collected P. monodon specimens from four different populations (Indonesia, Thailand, Mozambique and China). Sampling sites, sampling localities, geographical coordinates and number of collected specimens are presented in Table 1. Nine morphometric measurements were made on each specimen (Figure 1): BW, BL, CL, CW, CH and first, second, third and sixth pleon segment lengths (FSL, SSL, TSL and SISL, respectively). Body features were measured with a digital vernier caliper (Mitutoyo 500-744, Japan, accurate to ± 0.01 mm) and BW was obtained by using an electronic balance (Mettler Toledo 504, Switzerland, accurate to ± 0.0001 g).
| Populations | Locality | Latitude, Longitude | Number |
|---|---|---|---|
| Pop.1 | Banda Aceh, Republic of Indonesia | 05°30'N,94°40'E | 193 |
| Pop.2 | Khanom, Kingdom of Thailand | 09°12'N,100°03'E | 130 |
| Pop.3 | Mozambique Channel | 19°10'S,35°56'E | 85 |
| Pop.4 | Sanya, China | 18°47'N,109°26'E | 62 |
Table 1: Populations, sampling localities, geographical coordinates and number of collected specimens.
Data statistics
Observed data of the nine morphometric measurements were used for Pearson’s correlation (simple correlation) and path coefficient analyses. For each specimen, Pearson’s correlation coefficient (r) was calculated between every two morphometrics to indicate the relationship between the two traits. Pearson’s correlation and path coefficient analyses were performed using SPSS (version 19.0, SPSS Inc. 2010) and Excel 2003 (Microsoft Corporation, Redmond, WA, USA). The general form of the equation was used as follows [8]:
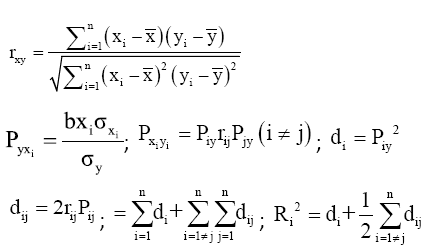
Where, rxy is the correlation coefficient of xi to yi, xi is the flag value of the independent variable, yi is the flag value of the dependent variable, Pyxi is the direct path coefficient of independent variable xi to dependent variable yi, bxi is the regression coefficient of the independent variable, σxi is the standard deviation of the independent variable, σy is the standard deviation of the dependent variable, Pij is the indirect path coefficient that trait i generated through trait j, di is the direct coefficient of trait i to dependent variable y, dij is the indirect coefficient of trait i to dependent variable y through trait j, d is the total determination coefficient of all traits, and Ri 2 is the total determination coefficient of all traits to trait y.
The regression equation linear model of quality traits Y was as follows:
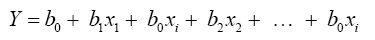
Where, Y is the dependent variable, b0 is a constant and bi is the partial regression coefficient of independent variable xi to dependent variable Y.
The mean, standard deviation and maximum and minimum of all P. monodon measurements were recorded. The coefficient of variation (CV%) was computed as: CV%=100 × SD/X, where SD is the standard deviation and X is the mean of the morphometric measurements of P. monodon.
Results
Descriptive statistics of the P. monodon samples are presented in Table 2. The coefficients of variation (CV) reflected the varying degrees within the samples. The CVs estimated for morphological characteristics within the samples were different from each other, with the CV estimated for BW the highest (42.98%), suggesting that BW exhibited considerable variation, which is an advantage for the selective breeding of P. monodon.
| Traits | Mean | SD | Range | CV (%) |
|---|---|---|---|---|
| BL | 209.92 | 29.87 | 240.50-261.20 | 14.23 |
| CL | 66.40 | 13.15 | 45.57-86.31 | 19.80 |
| CW | 34.03 | 6.90 | 22.47-44.51 | 20.29 |
| CH | 38.51 | 7.34 | 25.56-50.42 | 19.06 |
| FSL | 21.68 | 3.36 | 15.25-28.18 | 15.48 |
| SSL | 18.96 | 2.47 | 13.27-24.71 | 13.01 |
| TSL | 18.93 | 2.63 | 14.54-24.89 | 13.92 |
| SISL | 28.33 | 3.55 | 21.81-34.94 | 12.54 |
| BW | 149.22 | 64.13 | 56.24-261.19 | 42.98 |
SD: Standard Deviation; CV: Coefficient of Variation; BL: Body Length (mm); CL: Carapace Length (mm); CW: Carapace Width (mm); CH: Carapace Height (mm); FSL: First Pleon Segment Length (mm); SSL: Second Pleon Segment Length (mm); TSL: Third Pleon Segment Length (mm); SISL: Sixth Pleon Segment Length (mm); BW: Body Weight (g)
Table 2: Descriptive statistics of morphometric characteristics of P. monodon specimens.
The correlations were highly significant for all variables (P<0.01, Table 3), indicating that the variables had important practical significance for correlation analysis. Results showed that BW was positively correlated with all other variables, and the order of relevance was 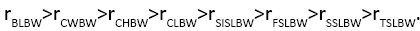
| Variable | BW | BL | CL | CW | CH | FSL | SSL | TSL | SISL |
|---|---|---|---|---|---|---|---|---|---|
| BW | 1.000 | ||||||||
| BL | 0.985 | 1.000 | |||||||
| CL | 0.962 | 0.964 | 1.000 | ||||||
| CW | 0.973 | 0.973 | 0.954 | 1.000 | |||||
| CH | 0.964 | 0.963 | 0.945 | 0.963 | 1.000 | ||||
| FSL | 0.868 | 0.871 | 0.866 | 0.866 | 0.867 | 1.000 | |||
| SSL | 0.776 | 0.779 | 0.733 | 0.763 | 0.761 | 0.631 | 1.000 | ||
| TSL | 0.817 | 0.829 | 0.766 | 0.811 | 0.795 | 0.692 | 0.740 | 1.000 | |
| SISL | 0.924 | 0.934 | 0.923 | 0.901 | 0.904 | 0.866 | 0.719 | 0.790 | 1.000 |
Table 3: Correlation coefficients between morphological characteristics of P. monodon.
The significant correlation between BW and all other traits made it difficult to assess the major factors associated with BW from simple correlation analysis alone. Therefore, path coefficient analyses between each trait and BW, including endogenous and dependent or exogenous and independent variables, were performed to clarify the relationships. The results showed that CL and CW were highly significant and significant to BW, respectively (Table 4), suggesting that these two morphological traits had strong direct effects on BW. The direct effect of BL on BW was obviously greater than the indirect effect of BL on BW through other traits. In addition, while the correlation coefficient between CW and BW was very large, the direct impact on BW was small. This indicated that the indirect effect of CW on BW (via other traits) was much larger than its direct effect, suggesting that CW affected BW through BL indirectly.
| Trait | Correlation coefficient | Direct effect | Indirect effect | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| BL | CL | CW | CH | FSL | SSL | TSL | SISL | Σ | |||
| BL | 0.985** | 0.557** | 0.103 | 0.178 | 0.111 | -0.001 | 0.016 | 0.003 | 0.019 | 0.429 | |
| CL | 0.962** | 0.107 | 0.537 | 0.175 | 0.109 | -0.001 | 0.015 | 0.003 | 0.018 | 0.856 | |
| CW | 0.973** | 0.183* | 0.542 | 0.102 | 0.111 | -0.001 | 0.015 | 0.003 | 0.018 | 0.790 | |
| CH | 0.964** | 0.115 | 0.536 | 0.101 | 0.176 | -0.001 | 0.015 | 0.003 | 0.018 | 0.849 | |
| FSL | 0.868** | -0.001 | 0.485 | 0.093 | 0.158 | 0.100 | 0.013 | 0.003 | 0.017 | 0.869 | |
| SSL | 0.776** | 0.020 | 0.434 | 0.078 | 0.140 | 0.088 | -0.001 | 0.003 | 0.014 | 0.756 | |
| TSL | 0.817** | 0.004 | 0.462 | 0.082 | 0.148 | 0.091 | -0.001 | 0.015 | 0.016 | 0.813 | |
| SISL | 0.924** | 0.020 | 0.520 | 0.099 | 0.165 | 0.104 | -0.001 | 0.014 | 0.003 | 0.905 | |
* Significant at P<0.05, ** Significant at P<0.01
Table 4: Path analysis of the effects of various traits on body weight of P. monodon.
The determination coefficients of various traits on BW are shown in Table 5. The direct determination coefficients of BL, CL, CW and CH to BW were 31.0, 1.1, 3.3 and 1.3%, respectively. The indirect determination coefficients of CL, CW and CH to BW through BL were 5.7, 9.9 and 6.2%, respectively.
| Trait | Direct determination coefficient | Indirect determination coefficient | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| BL | CL | CW | CH | FSL | SSL | TSL | SISL | ∑ | ||
| BL | 0.310 | 0.057 | 0.099 | 0.062 | 0.000 | 0.009 | 0.002 | 0.010 | 0.549 | |
| CL | 0.011 | 0.019 | 0.012 | 0.000 | 0.002 | 0.000 | 0.002 | 0.046 | ||
| CW | 0.033 | 0.020 | 0.000 | 0.003 | 0.001 | 0.003 | 0.060 | |||
| CH | 0.013 | 0.000 | 0.002 | 0.000 | 0.002 | 0.017 | ||||
| FSL | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | |||||
| SSL | 0.000 | 0.000 | 0.000 | 0.001 | ||||||
| TSL | 0.000 | 0.000 | 0.000 | |||||||
| SISL | 0.000 | 0.000 | ||||||||
Table 5: Determination coefficients of various traits on body weight of P. monodon.
Based on the path analysis and multiple correlation analysis results, multiple regression of the main morphological characteristics was performed (Table 6). BW was regarded as the dependent variable and the other traits as independent variables. The partial regression coefficients of BL and CW to BW were significant (P<0.05), and the multiple regression equation was: BW=1.546BL+2.533CW-261.522.
| Partial regression coefficient | Standard error | t | Significance | |
|---|---|---|---|---|
| Intercept | -254.377 | 12.379 | -20.548 | 0.000 |
| BL | 1.196 | 0.187 | 6.394 | 0.000 |
| CL | 0.523 | 0.299 | 1.746 | 0.084 |
| CW | 1.697 | 0.671 | 2.528 | 0.013 |
| CH | 1.008 | 0.526 | 1.916 | 0.058 |
| FSL | -0.017 | 0.654 | -0.026 | 0.979 |
| SSL | 0.519 | 0.631 | 0.822 | 0.413 |
| TSL | 0.093 | 0.684 | 0.136 | 0.892 |
| SISL | 0.353 | 0.853 | 0.414 | 0.680 |
Table 6 Partial regression coefficient test for various traits to body weight of P. monodon.
Predicted by the regression equation, no significant differences were found between the estimated and actual observed values. Thus, the above equation can be used conveniently and reliably in actual production.
Discussion and Conclusion
Phenotype correlation coefficients are used to comprehensively reflect relationships among variables. Since the influence of other variables is not eliminated, the direct effect of the independent variables on the dependent variables and their indirect effect through other independent variables may act against each other, and consequently phenotype correlation may be unable to accurately explain the relationship among all variables. Conversely, path coefficients are standardized partial regression coefficients that can differentiate direct and indirect effects without being influenced by other variables, and can therefore accurately reflect the relative importance of the results [16,17]. Our study showed that all nine quantitative traits of P. monodon showed positive correlations to each other, though the correlation coefficients between BW and BL (0.985), BW and CW (0.973), BW and CH (0.964) and BW and CL (0.962) were higher those between BW and SSL (0.776), BW and TSL (0.817) and BW and FSL (0.868). These results suggest that the four above mentioned variables exhibited a stronger influence on BW compared with the other traits, which is in accordance with the path coefficient results reported by Li et al. [18] and Zhang et al. [19] for Exopalaemon carinicauda and Exopalaemon modestus, respectively. However, correlation analysis alone cannot determine whether the traits with the largest correlation are the major factors that affect the dependent variables. Therefore, we used path analysis to not only reflect the relationship between variables, but also categorize the relationships between characteristics into direct and indirect effects, and thus determine the major factors affecting the BW of P. monodon.
In this study, the direct determination coefficient of BL (0.310) was the highest among all traits, while the direct determination coefficients of CW, CH, CL, FSL, SSL, TSL and SISL (0.033, 0.013, 0.011, 0.000, 0.000, 0.000 and 0.000, respectively) were much lower, suggesting that BL was the most important determinant of BW, consistent with the results of Yang et al. [20]. The phenotypic correlation coefficients of BL, CL, CW and CH with BW were basically the same, but the path analysis results suggested that the direct determination coefficients of these four phenotypes to BW had obvious differences. Specifically, CL, CW and CH influenced BW through BL indirectly, which indicated that the correlation between the variables did not accurately reflect their real relationship.
The main independent variable influencing a dependent variable can be determined when the sum of the multiple coefficient, or the sum of the single determination coefficient of variables to the dependent variable, or the sum of the pairwise coefficient are equal to or higher than 0.85. In this study, the summed (total) determination coefficient of BL and CW to BW was 0.952, indicating that BL and CW were the main characteristics that influenced BW. Hence, as far as P. monodon is concerned, morphological characteristics showed significant regression correlation with BW and indicated that path coefficient analysis was suitable for determining the real relationship between morphological traits and BW, as observed in Zhang et al. and Li et al. Due to the high genetic correlation between BL, CL, CW and CH, it would be appropriate to select BW and BL followed by CW as the main selection characteristics in the breeding of P. monodon.
Acknowledgement
This research was supported by the Development of Biology Industry in Shenzhen (NYSW20140331010053); National 863 Program (2012AA10A409), China Agriculture Research System (CARS-47); Special Scientific Research Funds for Central Non-profit Institutes, South China Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences 2015TS10);Guangdong Oceanic and Fisheries Project of China (A201601A14ã€ÂÂ2017A0014);Guangdong Province Science and Technology Project (2017A030303001).
References
- Hsu Y, Yang Y, Chen Y, Tung M, Wu J, et al. (1995) Development of an in vitro subculture system for the oka organ (lymphoid tissue) of Penaeus monodon. Aquaculture 136: 43-55.
- Ye L, Jiang S, Zhu X, Yang Q, Wen W, et al. (2009) Effects of salinity on growth and energy budget of juvenile Penaeus monodon. Aquaculture 290: 140-144.
- Yang Q, Wen W, Huang J, Chen X, Zhou F (2010) Growth and survival of families established based on 4 different populations of black tiger shrimp Penaeus monodon. South China Fisheries Science 6: 36-40.
- Sun M, Huang J, Yang Q, Zhou F, Su T, et al. (2012) Morphological variability in reciprocal hybrids F1 of Penaeus monodon from four geographical populations. Journal of Fisheries of China 36: 1512-1519.
- Zhang J, Huang Z, Lin H, Niu J, Lv G, et al.(2012) Effects of dietary protein content on growth performance and digestive enzyme activities of black tiger shrimp, Penaeus monodon. Marine Fisheries 34: 429-437.
- Krishna G, Gopikrishna G, Gopal C, Jahgeerdar S (2011) Genetic parameters for growth and survival in Penaeus monodon cultured in India. Aquaculture 318: 74-78.
- Sarawgi AK, Rastogi NK, Soni DK (1997) Correlation and path analysis in rice accessions from Madhya Pradesh. Field Crops Res 52: 161-167.
- RodrıÌÂÂguez DJ, Angulo-Sánchez JL, RodrıÌÂÂguez-GarcıÌÂÂa R (2001) Correlation and path coefficient analyses of the agronomic trait of a native population of guayule plants. Ind Crops Prod 14: 93-103.
- Wang X, Ma A, Xu K, Lei J, Yang Z, et al. (2008) Relationship between morphometric attributes and body weight of juvenile turbots Scophthalmus maximus. Acta Zoologica Sinica 54: 540-545.
- Harue K, Mutsuyosi T, Katsuya M (2000) Estimation of body fat content from standard body length and body weight on cultured red sea bream. Fisheries Science 66: 365-371.
- Zhan W, Lou B, Mao G, Xu D, Shi H, et al. (2010) Correlation and path analysis of growth traits of olive flounder (Paralichthys olivaceus). Journal of Quanzhou Normal University (Natural Science) 28: 29-32.
- Debowski P, Dobosz S, Robak S (1999) Fat level in body of juvenile Atlantic salmon (Salmo salar L.) and sea trout (Salmo trutta m. trutta L.) and method of estimation from morphometric data. Archives of Polish Fisheries 7: 237-243.
- Ahmed M, Abbas G (2000) Growth parameters of the finfish and shellfish juveniles in the tidal waters of Bhanbhore, Korangi Creek and Miani Hor Lagoon. Pak J Zoology 32: 21-26.
- Wright S (1921) Correlation and causation. Agric Res 20:557-585.
- Wright S (1934) The method of path coefficients. Annals of Mathematical Statistics 5:161-215.
- Sheng Z, Wu C (1999) Quantitative genetics. China Agriculture Press, Beijing, pp: 16-24.
- Wu Y, Chen A, Yao G (2010) The relationship between shell morphology and body weight of Meretrix meretrix. Marine Fisheries 32: 320-325.
- Li Y, Liu P a, Li (2005) Correlation and path analysis of morphometric traits on body weight for Exopalamon carinicauda. Progress in Fishery Sciences 33: 59-65.
- Zhang MY, Liu K, Duan JR (2010) Path analysis of effects of morphometric traits on body weight of Exopalaemon modestus in Taihu Lake. Chinese Agricultural Science Bulletin 26: 417-421.
- Yang C, He T, Liu X, Huang H, Xiang J (2011) Stepwise regression analysis of morphometric attributes on body weight of Penaeus vannamei. Acta Agriculturae Boreali-occidentalis Sinica 20: 15-20.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences