Acute Disseminated Encephalomyelitis (ADEM) in 6-year-old Libyan Boy: Case Report
1Department of Pathology, School of Medicine, The University of Zawia, Alzawia, Libya
2Department of Paediatric Respiratory, Tripoli Medical centre (TMC), Tripoli, Libya
- *Corresponding Author:
- Tawel HM
Department of Pathology, School of Medicine
The University of Zawia, Alzawia, Libya
Email: hodatawel@gmail.com
Received Date: February 06, 2019; Accepted Date: February 20, 2019; Published Date: February 28, 2019
Citation: Tawel HM, Elmehedwi IM, Ahmed FA (2019) Acute Disseminated Encephalomyelitis (ADEM) in 6-year-old Libyan Boy: Case Report. J Brain Behav Cogn Sci Vol.2 No.1:6
Copyright: © 2019 Tawel HM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
ADEM is demyelinating immune-mediated inflammatory disease affecting the CNS white matter following infection or vaccination. It is associated with influenza, rabies, measles, mumps, rubella, smallpox, pertussis, diphtheriatetanus- polio (DPT), Japanese B encephalitis, pertussis, and hepatitis B infection/and vaccine. Case definitions for ADEM is based on existence of multifocal neurologic deficit, MRI findings of diffuse white matter lesions, the monophasic course of the disease with no relapse for 3 months, and absence of evidences for other acute demyelinating disorders of CNS. Early diagnosis and treatment carry advantageous of preventing severe residual neurological deficits. Our case study suggests ADEM as the patient encountered level-1 criteria for its diagnosis. Detailed vaccination history when assessing patients with neurological manifestations should be obtained. Also, physicians should be aware of developing ADEM following seasonal influenza vaccination.
Keywords
Meninges encephalitis; Japanese B encephalitis; Neurological refluxes; Influenza vaccine
Introduction
Acute Disseminated Encephalomyelitis (ADEM) is a rare autoimmune disorder that causes inflammation of the myelinated insulation that affects the brain and spinal cord. ADEM is normally followed by viral or bacterial infection in 50-75% cases usually, in the form of a sore throat or cough. But none of these infectious agents are directly related to the disease. In general, ADEM begins about 7-14 days after the infection. Major symptoms include fever, headache, nausea and vomiting, confusion, vision impairment, drowsiness, seizures and coma which appear mild initially, but worsen with time.
It appears that ADEM is an immune response to the infection, but instead of fighting off the infection, it causes inflammation in the CNS. Inflammation is defined as the body's complex biological response to harmful stimuli, such as infectious agents, damaged cells, or irritants. It is a defence mechanism in response to the harmful stimuli. However, in the case of ADEM, the immune response is also responsible for the demyelination which in turn destroys the white matter. As much as affects children, it affects adolescents & adults too.
Case Report
A 6-year-old Libyan boy presented to paediatric department of Tripoli medical centre with 3 days of progressed generalized weakness, and anorexia. Along with that, the patient lost his orientation and was unable to follow commands at the day of admission. Five days prior, he had Coryza-like symptoms (i.e stuffy nose, runny nose, sneezing, and post-nasal drip). His past medical history was significant for bronchial asthma; however, he is not on regular treatment or follow-up. His developmental history was going with his age without any complication. The patient received all of his routinely scheduled vaccination up to dates. Though, the patient also received a single intramuscular dose of inactivated influenza vaccination (VAXIGRIP, 2016-2017) at a primary care unit. It was his only vaccine for the session. On admission, the patient was pale, drowsy, semiconscious (Glasgow coma scale score of 11); but he was not febrile, or distressed. On neurological examination, he had neck stiffness, brisk reflexes, increased muscle tone (i.e. spastic limbs), with bilateral extensor plantar response. Fundoscopic examination revealed no signs of optic neuritis or retinal haemorrhages, nevertheless could not exclude papilledema. An initial biochemical investigation revealed leucocytosis (16.64 × 103 cells/μl; mainly granulocytes, 71.1%), thrombocytosis (522 × 103/μl), and positive CRP (24 mg/l, 1:4 titration). Other his biochemical investigations including; random blood sugar, urea and electrolytes (i.e. serum Ca+2, mg+2, Na+, and K+), Creatinine, BUN, total serum protein, complete LFT, and bleeding profile (PT, PTT, and INR) were within the normal range. Patient’s neurological deficits combined with the primary biochemical data suggested a primitive diagnosis of infectious meningoencephalitis.
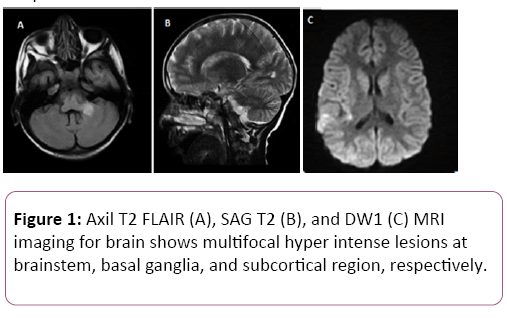
Thus, an initial empirical treatment with broad-spectrum antibiotics (Ceftazidime (50 mg/kg/dose; IV, q8 h) and Cefotaxime (150 mg/kg/24 h; IV, q8 h)), Acyclovir at 40 mg/kg/24 h; IV, q8h) was applied. Dexamethasone IV for 5 days (at 0.5 mg/kg/dose, q6 h) was also administrated, followed by steroid tapering for 4 weeks. To confirm our primary suggestion, magnetic resonance imaging (MRI) scan on brain was applied. MRI findings showed multiple lesions at subcortical, basal ganglion, and brainstem with unremarkable ventricular system (Figure 1). These findings were compatible with manifestations of ADEM. Over 7 days of therapy, dramatic clinical improvement was seen, the patient became fully conscious, and muscle tones as well as the neurological refluxes were resumed. Although, the patient was still unable to stand-up independently.
Figure 1: Axil T2 FLAIR (A), SAG T2 (B), and DW1 (C) MRI imaging for brain shows multifocal hyper intense lesions at brainstem, basal ganglia, and subcortical region, respectively.
After a week of therapy, the patient had dysuria, constipation, with spikes of fever reach to 39°C. A supplemented complete blood count (CBC) also revealed a further increase in WBCs count (23.23 × 103 cells/μl; mainly granulocyte, 88.8%) and platelets count (666 × 103/μl). any underlying haematological disorders were excluded at this point by blood film study. Hence, an enhanced therapy with Vancomycin (15 mg/kg/24 h, q8 h) was delivered. On day 21, the patient was fully recovered, and discharged with well-maintained alertness, motor function, speech, and spontaneous breathing. Outpatient follow-up at 3, 6, and 12 months reveals no relapse of the neurological symptoms.
This confirms ADEM case as the patient encountered level-1 criteria for its diagnosis by: A) - existence of multifocal neurologic deficit. B) - MRI findings of diffuse white matter lesions. C) - the monophasic course of the disease with no relapse for 3 months.
Case Definition of ADEM
ADEM is demyelinating immune-mediated inflammatory disease affecting the CNS white matter following infection or vaccination [1,2]. Sejvar et al. and the Brighton Collaboration Encephalitis Working Group have stated and structured the case definitions for ADEM in three categorized diagnostic certainty [3]. Level-1 takes in: a)-presence of focal or multifocal findings that referable to the CNS, or existence of histopathological data consistence with diagnosis of ADEM. b)-MRI findings of multifocal or diffuse hyper intense lesions of white matter. c) - monophasic course of disease (i.e. absence of relapse within a 3 months period of clinical improvement). Existence all the outlined criteria of level-1, but with insufficient follow-up time to document absence of the disease relapse, expresses the Level 2 diagnostic certainty of ADEM [3]. Setting of level 3 diagnostic certainties is based on only presence of focal or multifocal neurological manifestations that features of a demyelinating event [3].
Existence of another acute infectious or diagnosis for the illness, recurrence or relapse of the illness after a period of 3 months of clinical improvement, or inconsistency of MRI findings with the disease, excludes all levels of diagnostic certainty of ADEM [3,4]. Nevertheless, the disease could be recurrent or multiphasic. Recurrent ADEM, a term referring to relapses that happen at least 3 months after initial ADEM events or 4 weeks after finishing steroid therapy with MRI findings at the same areas of the preliminary ADEM incident [3,4]. Involving of new areas of CNS on MRI that are not existed in the initial ADEM episode refers to “Multiphasic ADEM” [3,4]. For example, if the relapse take place within the first 3 months of initial events or within 4 weeks of steroid tapering. This is known as “steroiddependent ADEM” [4].
Case definitions for ADEM develop a uniform classification for the disease and distinguish it unmistakably from other acute demyelinating disorders of CNS as optic neuropathy and multiple sclerosis [3,4].
Epidemiology of ADEM
ADEM forms 75% of cases of primary inflammatory demyelinating disorders of the central nerves system with estimated annual incidence of 0.4 to 0.8 per 100,000 [2,4,5]. ADEM can be triggered by either viral/bacterial infection or vaccination.
Post-infectious ADEM has been reported following various viral infection as influenza, herpes simplex, varicella zoster, measles, mumps, rubella, cytomegalovirus, hepatitis A, and enterovirus infections [6]. A recent prospective study has shown that 27% of patients with herpes simplex encephalitis developed neuronal antibodies within 2 months after the treatment, which suggests the risk of development of ADEM with herpes simplex infection [7]. ADEM has also linked to measles infections by incidence of 1 to 2 per 1000 infections [6,8]. Also, it has been recently reported that both seasonal and pandemic influenza H1N1 infections are associated to several rare neurological complications including ADEM [9,10]. Post-infectious ADEM is usually sessional and due to either direct myelin or neuronal damage by pathogen, or via activation of auto-immune mechanisms [4,6,11].
Post-vaccination encephalomyelitis account for less than 5% of aetiology [12,13]. ADEM is associated with a number of vaccinations such as influenza, rabies, measles, mumps, rubella, smallpox, pertussis, diphtheria-tetanus-polio (DPT), Japanese B encephalitis, pertussis, and hepatitis B vaccine [4,13,14].It is also more frequent after primary vaccination than after revaccination. Have shown that the estimated rate of postvaccination ADEM vary from 1: 4000 to 1: 80,000 after primary vaccination, and from 1: 50,000 to 1: 450,000 post revaccination. A recent study also showed a significant increase in the risk of ADEM in children after vaccination in the 31-60 day exposure interval, but not at neither 0-30 nor 61-180 day interval [15].On the other hand, the study showed no association between ADEM recurrence of and vaccine received .In view of that, ADEM condition can be considered as a neurological complication after certain vaccine. Also, a final diagnosis of post-vaccination ADEM could be supposed for our case by reasons of: a)-his recent exposure to seasonal influenza vaccine. b)-the patient encountered level 1 criteria for case definition of ADEM.c)-absence of evidences for other acute demyelinating disorders of CNS.
ADEM and influenza vaccine
Influenza (flu) vaccination is advised for some high risk groups such as elderly peoples, pregnant women, and patients with chronic disorders [16] . Only recently ADEM has been linked to influenza vaccine. It has been shown that influenza virus antigens could display a cross-reactivity with different myelin proteins that being exist in myelin sheets of CNS [1,17,18].Hence, influenza viruses of the vaccine could preserve their natural occurring antigens, which might serve as potential epitopes stimulating the autoimmunity against myelin sheets of CNS.
However, because of its low-incidence rate, still there is no large published studies for influenza vaccine-induced ADEM, or a recognised therapeutic guidelines for it [2]. In total, nearly 15 cases of ADEM following influenza vaccination has been reported [2,19] In a review of neurological side effects of influenza vaccine between 2003 and 2005 in the USA, one ADEM of 460 adverse events was reported [4,20]. An additional study that was conceded in Japan at 2007, reported 3 cases of ADEM amongst 38.02 million doses of influenza vaccine obtained between 1994 and 2004 [4,21].Recently, Clinical Immunization Safety Assessment (CISA) network reviewed reports of non-fatal serious neurological deficit of influenza (H1N1) vaccination among 212 Vaccine Adverse Event Reporting System (VAERS) and found that ADEM was deemed 3.8% of the cases [22]. The majority of the reported cases were received inactivated influenza vaccine.
According to the manufacturer leaflet, VAXIGRIP/VAXIGRIP Junior vaccine was made up in eggs and contains inactivated parts of three influenza strains. It consists of influenza A-H1N1, influenza A-H3N2, and influenza B strains. ADEM condition has been reported by Wei-Ti Chen, 42-year-old woman 3 weeks following Fluarix vaccine (i.e. inactivated influenza vaccine) [19]. Like to VAXIGRIP vaccine, Fluarix vaccine also encloses influenza A (H1N1 and H3N2) and the influenza B (Yamagata and Victorialineage) virus strains [19].This highlights the possibility of developing ADEM following VAXIGRIP vaccine. A critical care should be applied for this possibility to ensure an early right diagnosis and proper management for post-vaccination ADEM.
Diagnosis of post vaccination ADEM
Clinicians should pay further attention for the possibility of developing ADEM in patients who presented with various neurological deficits that are preceded by vaccination. Patient may presented firstly with some systemic manifestations such as fever, headache, generalized malaise, and myalgia [1].Within 30 days of primary presentation, patient may develop various neurological deficits [1].The deficits vary depending on which region of the CNS is affected and the degree of demyelination [1,23].Multifocal presentations of ADEM may perhaps a combination of pyramidal and cerebellar deficit (i.e. 76% acute hemiparesis and 59% ataxia), optic neuropathies (bilaterally), meningioma’s, depressed consciousness, and urinary dysfunction if peripheral myelitis is predominant [6,24]. Beside to the clinical presentation, MRI imaging studies of the brain should be done to confirm ADEM. It can be normal at the beginning, but at 5 to 14 days from symptom onset the characteristic lesions of ADEM are mostly developed [4]. The diagnostic imaging hallmark of ADEM is the presence of disseminated focal or multifocal regions of inflammation and demyelination within cerebral subcortical and deep cortical white matter [1,4] Lesions in the deep grey matter (i.e. the thalamus and basal ganglia) are also considered [4,6]. Corresponding to literature, MRI study of our case showed multiple scattered hyper intense lesions at subcortical, basal ganglion, and brainstem, suggesting ADEM condition.
Management and prognosis of ADEM
Once ADEM is diagnosed, appropriate therapy should be started directly to guarantee a full recovery. Steroid therapy as methylprednisolone or dexamethasone for 3 to 5 is widely used as first line therapy for ADEM [4,6,23]. This therapy regime needs to be tapered for 4 to 6 weeks as shorter tapering period has been linked with a high tendency of relapse [6,25]. In the first few days of disease, intravenous acyclovir might be supplemented if acute viral encephalitis is still suspicious. Since our case displayed a full recovery with dexamethasone treatment, we can highpoint the benefits of early steroid therapy on ADEM prognosis. Literature has also recommended a combination therapy of corticosteroids with immunoglobulin for patients who respond poorly to intravenous corticosteroids alone [4]. In severe, un-responded cases of ADEM, plasma exchange therapy is regarded a therapy of choice [4,13]. however, there is no available studies show the advantageous of its early use in the course of the disease [4] Generally, ADEM has a good prognosis with a full recovery, only a few reported cases displayed minor residual neurological deficits [4,13]. Nearly 50% to 75% of the reported cases showed a full recovery with average time ranges from 1 to 6 months [4]. There is only two reported cases of death following post-influenza vaccination- ADEM despite methylprednisolone and plasma exchange therapy [2].This could be due to the severity of the neurological deficit at the time of presentation. Brainstem dysfunction, poor response to corticosteroids, spinal cord involvement, high CSF protein levels, older age, and female gender all serve as a poor prognostic predictors for ADEM [4,6,25]. The prognosis of ADEM also significantly relays on the time of beginning of the treatment. Early treatment carries advantageous of preventing severe residual deficits and improves the disease overall outcome [19-30].
Conclusion
Although a rare incidence, physicians should be aware of developing certain neurological deficit, namely ADEM following seasonal influenza vaccination. Our case further suggests a detailed vaccination history when assessing patients with neurological manifestations. This enables a fast and effective management of ADEM, and then having a good outcome of the disease. Post-influenza vaccination ADEM risk could be coincidental, thus further separate self-controlled case series (SCCS) studies are required.
References
- Noorbakhsh F, Johnson RT, Emery D, Power C (2008) Acute Disseminated Encephalomyelitis: Clinical and Pathogenesis Features. Neurol Clin 26: 759-780.
- Shoamanesh A,Traboulsee A (2011) Acute disseminated encephalomyelitis following influenza vaccination.Vaccine 29: 8182– 8185.
- Sejvar JJ, Kohl KS, Bilynsky R, Blumberg D, Cvetkovich T, et al. (2007) Encephalitis, myelitis, and acute disseminated encephalomyelitis (ADEM): case definitions and guidelines for collection, analysis, and presentation of immunization safety data.Vaccine 25: 5771-5792.
- Huynh W, Cordato DJ, Kehdi E, Masters LT, Dedousis C (2008) Post-vaccination encephalomyelitis: Literature review and illustrative case. J Clin Neurosci 15: 1315-1322.
- Leake JA1, Albani S, Kao AS, Senac MO, Billman GF, et al. (2004) Acute disseminated encephalomyelitis in childhood:epidemiologic, clinical and laboratory features. Pediatr Infect Dis 23: 756-764.
- Farshid N (2008) Acute Disseminated Encephalomyelitis: Clinical and Pathogenesis Features. Neurol Clin 26: 759-780.
- Armangue T, Spatola M, Vlagea A, Mattozzi S, Cárceles-Cordon M, et al. (2018) Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol 17: 760 -772.
- Chowdhary J, Ashraf SM, Khajuria K Measles (2009) with acute disseminated encephalomyelitis (ADEM).Indian pediatr 46: 72-74.
- Kheiri B, Abu Sitta E, Salih A, Al Qasmi M, Bachuwa G (2018)Acute disseminated encephalomyelitis following Influenza A pneumonia. Clin Case Rep 6: 436-438.
- Ekstrand JJ (2012) Neurologic complications of influenza.Semin Pediatr Neuro19: 96-100.
- Huynh W, Cordato DJ, Kehdi E, Masters LT, Dedousis C (2008) Postvaccination encephalomyelitis: literature review and illustrative case. J Clin Neurosci 1315-1322.
- Machicado JD, Bhagya-Rao B, Davogustto G, McKelvy BJ (2013) Acute Disseminated Encephalomyelitis following Seasonal Influenza Vaccination in an Elderly Patient. Clin Vaccine Immunol1485-1486.
- Bennetto L ,Scolding N (2004) Inflammatory/post-infectious encephalomyelitis.J Neurol Neurosurg Psych 75: 22-28.
- Yoneda M (2016) Acute Disseminated Encephalomyelitis Following Immunization with Human Papillomavirus Vaccines. Intern 55: 3077-3078.
- Chen Y, Ma F, Xu Y, Chu X, Zhang J (2018) Vaccines and the risk of acute disseminated encephalomyelitis vaccine. 36: 3733-3739.
- https://www.who.int/vaccine_safety/initiative/tools/vaccinfosheets/en/
- Wucherpfennig KW, Strominger JL (1995) Molecular mimicry in T cellmediated autoimmunity: viral peptides activate human T cell clones specific for myelin basic protein. Cell 80: 695-705.
- Menge T, Kieseier BC, Nessler S, Hemmer B, Hartung HP, et al. (2007) Acute disseminated encephalomyelitis: an acute hit against the brain. Curr Opin Neurol 20: 247-254.
- Chen WT, Huang YC, Peng MC, Wang MC, Lin KP (2016)Acute Disseminated Encephalomyelitis After Influenza Vaccination: A Case Report. Crit Care Nurse 36: 1-6.
- Izurieta HS, Haber P, Wise RP, Iskander J, Pratt D, et al. (2005) Adverse events reported following live, cold adapted, intranasal influenza vaccine. JAMA 294: 2720–2725.
- Nakayama T, Onoda K (2007) Vaccine adverse events reported in postmarketing study of the Kitasato Institute from 1994 to 2004.Vaccine 25: 570-576.
- Williams SE1, Pahud BA, Vellozzi C, Donofrio PD, Dekker CL, et al. (2011) Causality assessment of serious neurologic adverse events following 2009 H1N1 vaccination.Vaccine Vol. 29: 8302–8308.
- Tenembaum S, Chitnis T, Ness J, Hahn HS ( 2007) Acute disseminated encephalomyelitis. Neurology 68: 23-36.
- Tenembaum S, Chamoles N, Fejerman N (2002) Acute disseminated encephalomyelitis: a longterm follow-up study of 84 pediatric patients 8 Neurology 59: 1224-1231.
- Anlar B, Basaran C, Kose G, Guven A, Haspolat S, Yakut A, et al. (2003) Acute disseminated encephalomyelitis in children: outcome and prognosis. Neuropediatrics 34: 194-199.
- Alexander M, Murthy JM (2011) Acute disseminated encephalomyelitis: treatment guidelines. Ann Indian Acad Neurol 14: 60-64.
- Fenichel GM (1982) Neurological complications of immunization. Ann Neurol 12: 119–128.
- Pohl D, Alper G, Van Haren K, Kornberg AJ, Lucchinetti CF, et al. (2005) Acute disseminated encephalomyelitis: an update. Arch Neurol 62: 1673-1680.
- Johnson RT (1987) The pathogenesis of acute viral encephalitis and postinfectious encephalomyitis. J Infect Dis 155: 359-364.
- Dale RC, de Sousa C, Chong WK, Cox TC, Harding B, et al. (2000) Acute disseminated encephalomyelitis, multiphasic disseminated encephalomyelitis and multiple sclerosis in children. Brain 123: 2407-2422.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences