A review of pharmacotherapy updates in the AIDSinfo guidelines for adults and adolescents with HIV infection
Ayse Elif Ozdener*, Malgorzata Slugocki and Julie Kalabalik
School of Pharmacy and Health Sciences, Fairleigh Dickinson University, USA
- *Corresponding Author:
- Ayse Elif Ozdener
Fairleigh Dickinson University, School of Pharmacy and Health Sciences, USA
Tel: 973-443-8211
E-mail: eozdener@fdu.edu
Received date: September 14, 2018; Accepted date: September 25, 2018; Published date: October 04, 2018
Citation: Ozdener AE, Slugocki M, Kalabalik J (2018) A Review of Pharmacotherapy Updates in the AIDSinfo Guidelines for Adults and Adolescents with HIV Infection. J Pharm Prac Edu Vol 1:12
Abstract
Guidelines for the management of HIV infection in adults and adolescents have recently been updated by the Department of Health and Human Services. The authors have switched to people-first language. In addition, terminology used to describe antiretrovirals’ (ARV) place in therapy has changed from “recommended regimen options” and “alternative regimen options” to “recommended initial regimens for most people with HIV” and “recommended initial regimens in certain clinical situations”, respectively. The most significant change that will impact patient care is the removal of boosted darunavir (DRV) from the “recommended initial regimens for most people with HIV” category due to its association with increased cardiovascular risk. All integrase strand transfer inhibitors (INSTI), including newly approved bictegravir (BIC) combined with tenofovir alafenamide/ emtricitabine (TAF/FTC), are recommended as initial regimens for most people with HIV infection due to their proven efficacy, safety, and high threshold for resistance development. This review article will discuss major modifications to the Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents Living with HIV with a focus on pharmacotherapy recommendations for treatment-naïve, treatment-experienced, and co-infected patients, including clinical trial results that influenced the recommendations. Adverse effects and drug interactions will also be reviewed.
Keywords
HIV, Treatment guidelines, Pharmacotherapy, AIDS; Antiretroviral
Introduction
The Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents Living with Human Immunodeficiency Virus (HIV) were updated March 2018. Changes to the guidelines that are most relevant to healthcare providers who care for persons with HIV are discussed in this review article. The updated guidelines adopted people-first language in referring to individuals who are living with HIV. Information on immediate antiretroviral therapy (ART) initiation on the day of HIV diagnosis was added. Initial ART regimens are presented as “recommended initial regimens for most people with HIV” and “recommended initial regimens in certain clinical situations.” Regarding specific drug classes, integrase strand transfer inhibitor (INSTI)-based regimens are included in most initial regimens with non-nucleoside reverse transcriptase inhibitor (NNRTI)- and protease inhibitor (PI)- based regimens for certain patients. New safety data surrounding tenofovir alafenamide (TAF) compared to tenofovir disoproxil fumarate (TDF) are discussed. Data on ongoing clinical trials and drug safety are included as well. The risks of monotherapy with any antiretroviral drug, the use of efavirenz in the first trimester of pregnancy, and recommendations for patients with hepatitis B virus (HBV)/HIV coinfection and chronic HBV and hepatitis C virus (HCV) co-infection are discussed. Investigational agents that may be considered in the case of virologic failure are explored. Pharmacologic options for persons with HIV who have sustained viral suppression and no drug resistance are reviewed. Important drug-drug interactions between new HCV drugs and ARV drugs are discussed. Updated guidelines include information on barriers to adherence to the HIV care continuum and recommendations on how to improve adherence [1].
Antiretroviral Therapy for Adults and Adolescents with HIV Infection
Protease Inhibitors for initial therapy
The current guidelines recommend INSTI-based regimens only as an initial treatment option for most people living with HIV. Boosted darunavir with ritonavir (DRV/r) was the only PI-based regimen that was recommended as initial therapy in the 2016 guidelines [2]. This regimen is now recommended in certain clinical situations. Several trials demonstrated an association between PI use and increased risk for myocardial infarctions [3,4]. It is important to note that these trials did not study DRV or DRV/r. An observational study compared cardiovascular disease (CVD) risk (MI, stroke, sudden cardiac death, invasive cardiovascular procedures) in patients receiving DRV/r vs. atazanavir/ritonavir (ATV/r). Investigators concluded that there was a small but gradual increase in CVD risk associated with DRV/r use. This association did not exist in patients taking ATV/r [5]. In fact, results from a separate study showed that ATV/r reduced atherosclerosis risk as measured by carotid artery intima medial thickness which was seen to a lesser degree in the DRV/r arm [6,7]. The clinical implication of these results is unknown and need further evaluation. Despite this association, DRV/r may be an appropriate option in patients with INSTI gene mutations where nonadherence is a concern. Protease inhibitors have a higher threshold for resistance development and may be advantageous in this situation.
Results from a phase 3 study comparing elvitegravir/cobicistat (EVG/c) plus TDF/FTC versus ATV/r plus TDF/FTC demonstrated that the rate of virologic suppression was greater with EVG/c than ATV/r [8]. Atazanavir/r was already recommended as an alternate regimen in the 2016 guidelines; thus, these results did not have an impact on the level of recommendation for EVG/c or ATV/r [2]. When comparing ATV/r plus TDF/FTC to dolutegravir (DTG) plus abacavir/lamivudine (ABC/3TC), the two regimens were noninferior in viral load suppression; however, there were fewer drug-related adverse effects associated with the DTG arm [9]. These findings had no bearing on the level of recommendation for the mentioned treatment regimens [1].
Nucleoside reverse transcriptase inhibitors (NRTI) for initial therapy
The NRTI updates focused on studies that evaluated TAF and TDF. A study investigated darunavir co- formulated with cobicistat (DRV/c) plus TAF/emtricitabine (FTC) and compared it to DRV/c plus TDF/FTC in a phase 2 trial aimed to look at viral load suppression in treatment-naïve patients. Results showed noninferiority between DRV/c plus TAF/FTC and DRV/c plus TDF/FTC. There was less proteinuria and bone mineral density reduction observed in the DRV/C plus TAF/FTC arm [10]. A head-to- head trial comparing TDF/FTC to TAF/FTC (TAF/FTC) showed that TAF/FTC was associated with higher rates of virologic suppression. It is important to note that there were more patients that discontinued treatment in the TDF arm due to kidney-related adverse effects which could have skewed the results in favor of the TAF arm. Tenofovir alafenamide was associated with higher rates of lipid increases compared to the TDF arm which may not be a favorable adverse effect in patients with pre- existing CVD [10,11].
In the new guidelines, there is no distinction between TDF and TAF in the tables that contain medication regimen recommendations. Instead of stating the entire name, including the salt form of tenofovir, the authors only include the word ‘tenofovir’ with the same level of recommendation for regimens containing DTG or EVG/c (AI). The level of recommendation for the two salt forms for tenofovir differs with raltegravir (RAL) (RAL plus TDF/FTC [AI]; RAL plus TAF/FTC [AII]) [1].
Integrase strand transfer inhibitors for initial therapy
All of the available INSTIs, including newly approved singletablet regimen containing TAF/FTC and novel bictegravir (BIC), are recommended as initial therapy in most patients with HIV infection [1]. The push for INSTI-based regimens is due to their proven efficacy and greater tolerability compared to other antiretroviral in other classes. A recent study comparing the efficacy of DTG/ABC/3TC and ATV/r plus TDF/FTC showed higher viral load suppression rates in the DTG arm. This difference was likely due to lower rates of virologic nonresponse and fewer discontinuation rates due to adverse effects in the DTG arm [9].
In May 2017, once-daily RAL was approved by the FDA. This approval came after results from a phase 3 trial that compared RAL 400 mg twice daily to RAL 1200 mg once daily (two 600 mg tablets) showed noninferiority in viral load suppression among the two drugs [12]. The new guidelines recommend the use of RAL 1200 mg once daily as an initial regimen for most people with HIV with the same level of recommendation as RAL 400 mg twice daily (AI) which is a change from the 2016 guidelines [1,2]. This once-daily option may be more suitable for patients who are having difficulty adhering to the twice-daily dosing of RAL 400 mg.
Dual-therapy strategies under investigation
The strategies discussed under this section are currently being investigated and therefore, not recommended by the authors. Combination DTG with 3TC has been studied in two, small, single-arm studies and showed positive results. The PADDLE trial included 20 treatment-naïve patients. Ninety percent of participants had undetectable viral loads defined as HIV RNA<50 copies/mL at 48 weeks of therapy. Approximately 83% of patients who reached viral load suppression at week 48 maintained it at 96 weeks. Although these are promising results, there are some limitations to the applicability of this data. All of the patients included in this study had baseline viral loads<100,000 copies/mL. Furthermore, rapid viral load suppression is an important factor in treating patients with HIV infection. In most cases, viral load suppression is expected after 4-6 weeks of treatment initiation. This study only evaluated week 48 and 96 results, therefore, it is impossible to assess how quickly viral load levels fell below 50 copies/mL with just dual-therapy. There was an average increase of 271 cells/mm3 in CD4 cell count [13]. Another single-arm trial included patients with baseline viral loads>100,000 copies/mL and had similar results to the PADDLE trial. There were 3/120 patients who developed integrase gene-associated resistance in this trial which was related to nonadherence to dual therapy [14].
Another dual therapy regimen under investigation is DRV/r plus 3TC. The ANDES trial was an open-label, randomized trial that compared dual therapy DRV/r plus 3TC to triple therapy DRV/r plus 3TC/TDF. This trial is ongoing; however, interim results show promising results: 71/75 participants in the dual-therapy arm and 68/70 participants in the triple-therapy arm achieved viral load levels<400 copies/mL [15].
Antiretroviral not recommended for people with HIV infection
Delavirdine (DLV), didanosine (ddI), nelfinavir (NFV), indinavir (IDV), and stavudine (d4T) are ARTs no longer recommended by the 2017 guidelines due to inferior virologic efficacy, increased pill burden, and serious adverse effects compared to other ARV. Some of these adverse include lactic acidosis, hepatomegaly with steatosis and pancreatitis seen with ddl and d4T, treatment-emergent rash with DLV, and nephrolithiasis with IDV [1,16-19].
There have been several studies that assess the efficacy of PI monotherapy with either DRV/r, LPV/r, or ATV/r in maintaining viral load suppression in patients infected with HIV already virally suppressed on triple ARV therapy (tART) [11,20-28]. Results have been conflicting, but the European AIDS Clinical Society guidelines do recommend PI monotherapy as a class-sparing strategy in certain situations where the patient does not have any PI resistance, has suppressed viral load<50 copies/mL for at least 6 months, and is HBV negative [29]. The NIH guidelines however, have a different stance and do not recommend PI monotherapy because of results of several studies such as the PIVOT trial, which show that PI monotherapy is inferior to tART when using an intention-to- treat analysis [20-22,26,30]. Integrase strand transfer inhibitor monotherapy is also not recommended as it increases risk of virologic rebound and INSTI resistance [31,32]. The limitation to this recommendation is that only DTG monotherapy has been studied. In addition, the studies that evaluated DTG-monotherapy where small, cohort, or case reports.
Combination ritonavir and cobicistat or TDF with TAF are not recommended because both drug combinations have the same indication and similar mechanisms of action [1]. The use of these two drug combinations would constitute duplication of therapy.
Management of treatment-experienced patient in the setting of virologic failure
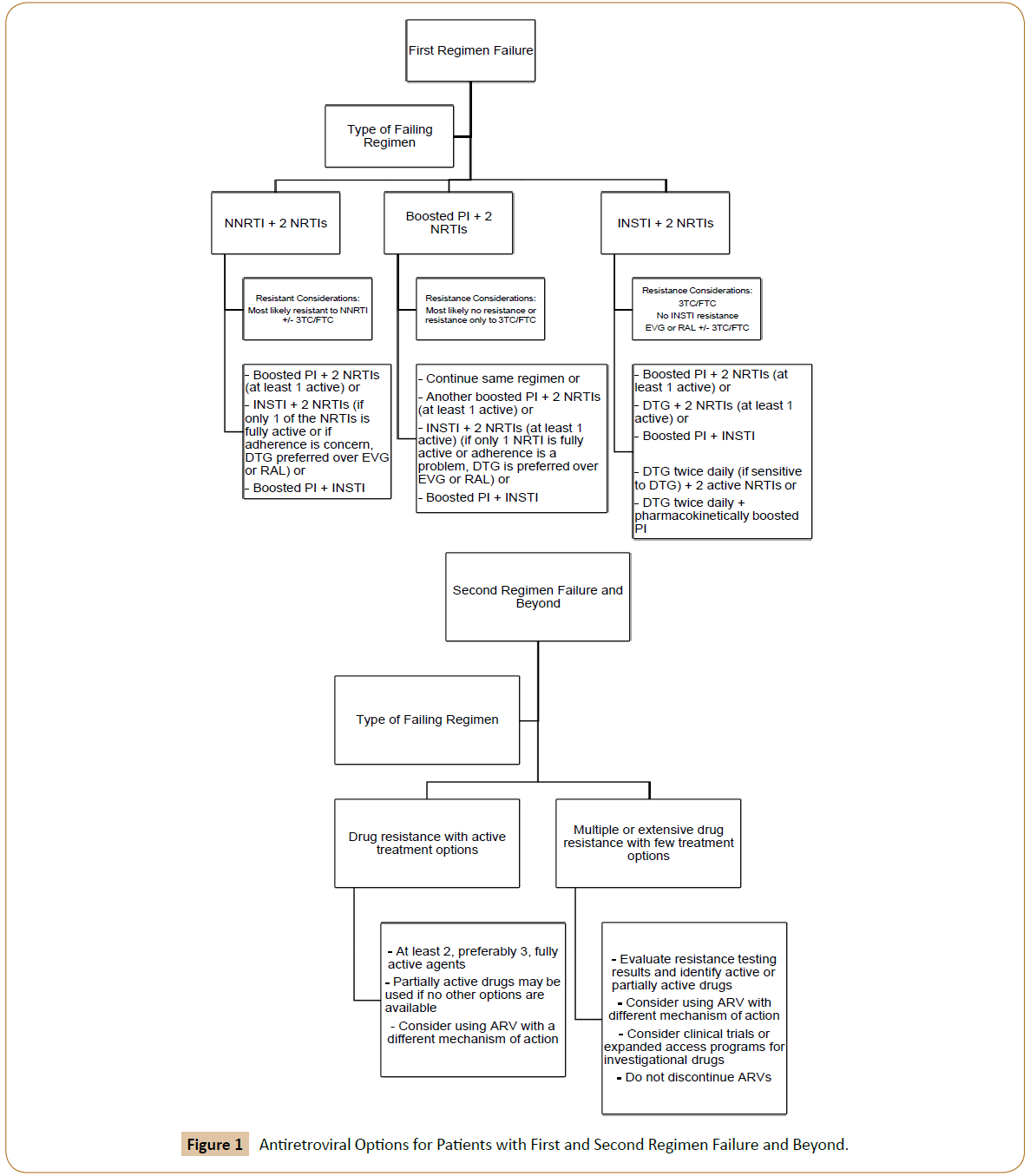
The updated guidelines define “low-level viremia” as confirmed detectable HIV RNA<200 copies/mL. The updated guidelines include recommendations on how to manage virologic failure in different clinical scenarios including failure with first antiretroviral regimen and failure with second-line regimen and beyond. The DAWNING study compared DTG plus 2 NRTIs versus lopinovir boosted with ritonavir (LPV/RTV) plus 2 NRTIs in HIV-1 infected individuals who failed first-line therapy with 2 NRTIs plus 1 NNRTI. According to reports of the interim data, at 24 weeks, 78% of patients in the DTG group versus 69% in the LPV/RTV group had HIV-1 RNA< 50 copies/mL (adjusted difference 9.6%, 95%CI 2.7% to 16.4%, p=0.006 for superiority). There were more drug- related adverse events in the LPV/RTV group mostly due to gastrointestinal disorders [33]. The SELECT study was a randomized, open-label, phase 3, non-inferiority study investigating if LPV/r plus RAL is non-inferior to LPV/r plus NRTIs based on probability of virological failure by 48 weeks. The cumulative probability of virological failure was 10.3% in the RAL group versus 12.4% in the NRTI group (weighted difference -3.4% 95% CI -8.4 to 1.5) showing non-inferiority of RAL to NRTIs as second-line antiretroviral therapy and LPV/r plus RAL as an alternative option [34]. The guidelines state that an INSTI plus 2 NRTIs is an acceptable option if patients have failed first-line NNRTI therapy. Dolutegravir is preferred over EVG or RAL if adherence is an issue and only one NRTI is fully active. If resistance to RAL and EVG is present but susceptibility to DTG is observed, a boosted PI plus 2 NRTIs, twice-daily DTG plus 2 active NRTIs, or twice-daily DTG plus boosted PI are options. Under the section of secondline regimen failure and beyond, the guidelines include a link to information on the AIDSinfo website on investigational drugs in late-stage clinical studies. Fostemsavir is a gp120 attachment inhibitor currently in Phase II development. Although ibalizumab is listed as an investigational drug in the guidelines, this CD4- directed post-attachment inhibitor was recently approved by the FDA on March 6, 2018, and indicated for HIV-1 infected, treatment experienced patients with multidrug resistance failing their current antiretroviral regimen [35].
Deciding on a treatment regimen for a patient who was previously treated with suspected drug resistance but for whom we have limited treatment information is a challenge. Updated guidelines recommend possibly selecting drugs with high barrier to resistance (DTG and/or boosted DRV) for patients with no ARV history available.
Patients should be closely monitored and assessed for HIV RNA and resistance 2–4 weeks after initiating therapy. The updated guidelines list ARV options for patients with virologic failure; these options are illustrated in Figure 1. The reader is directed to the full text in the guidelines for details.
Management of treatment-experienced patients: regimen switching in the setting of virologic suppression
The guidelines recommend against monotherapy with a boosted PI or an INSTI as a switching strategy. Evidence demonstrates virologic failure and development of resistance with this strategy. Dolutegravir monotherapy is associated with virologic failure and resistance. Wijting et al. performed a multicenter randomized trial comparing DTG with continued combination ART (con-cART) in patients on combination ART (cART) with viral load <50 copies/ mL for greater than 6 months, CD4 nadir >200 cells/uL, pre-cART peak viral load <100,000 copies/mL and no virological failure history. At week 24, DTG monotherapy was non-inferior to concART; however, at week 48, virological failure developed in 8 DTG monotherapy patients with 3 patients who developed DTG resistance leading to premature study discontinuation. Virological failure was significantly less in the cART group (p=0.03) [36]. In an international, multi-cohort, retrospective study of 178 ART experienced patients with no history of INSTI virological failure who were switched from ART regimens to DTG monotherapy, 11 developed virological failure and 7 INSTI resistance-associated mutations [37].
Switching patients to ATV/r plus RAL was associated with virologic failure and treatment discontinuations at a higher rate compared to switching patients to ATV/r plus TDF/FTC according to one study [38]. Switching patients with virologic suppression to a regimen of boosted PI and maraviroc (MVC) is not recommended based on evidence suggesting greater risk of virologic failure and treatment discontinuations compared to other regimens. The Maraviroc Switch Study, a randomized, multicenter, 96-week, open-label switch study, evaluated MVC as an alternative for HIV- 1 infected patients with R5-tropic virus and who are virologically suppressed. Patients were randomized to current combination ART, replacing PI (MVC+2 NRTI) or NRTI backbone (MVC+PI/r) with twice daily MVC. The primary endpoint was difference in proportion with plasma viral load <200 copies/mL after 48 weeks. Maraviroc plus PI/r was found to be significantly inferior to the control arm (84.1% (95% CI, -19.8% to–5.8%) and 77.7% (95% CI, -24.9% to -8.4%) with viral load <200 and <50 copies/ mL, respectively) [39].
Virologic relapse was observed in patients switched to MVC plus RAL therefore this combination should be avoided. In one singlearm study of lipohypertrophic HIV-infected patients with virologic suppression and R5 tropic virus who were switched to MVC plus RAL, 7 patients failed MVC plus RAL therapy. Five patients had virological failure and 2 discontinued treatment due to adverse events. Raltegravir resistance mutations were observed in 3 out of 5 patients and CXCR4 tropic virus in 2 out of 5 [40].
Viral load suppression will likely be maintained in the setting of between-class switches as long as there is no resistance to the other drugs in the regimen. The updated guidelines now list the option of replacing a patient on 2 NRTIs plus a boosted PI to 2 NRTIs plus MVC. In the Maraviroc Switch Study, virologically suppressed subjects were randomized to either continue the current PI/r-based regimen or to switch to MVC plus 2 NRTIs. After 96 weeks, 89% in the PI/r group and 90.4% in the MVC group had plasma viral load <50 copies/mL (95% CI, -6.6, 10.2) indicating that MVC is effective at maintaining virological suppression [41]. If switching a patient to MVC, it is recommended to assess co-receptor usage using proviral DNA from peripheral blood mononuclear cells.
The guidelines include additional studies that have been published regarding the success of boosted PI- based regimen plus 3TC (dual therapy) for maintaining virologic suppression in ART-naïve patients without resistance mutations or patients with sustained virologic suppression. In a multicenter, open-label, non-inferiority trial, virologically suppressed patients were randomized to either continue triple therapy with DRV/r plus 2 NRTIs or switch to dual therapy with DRV/r plus 3TC. Patients with viral load <50 copies/ mL after 48 weeks was 88.9% in the dual therapy arm and 92.7% in the triple therapy arm (95% CI, -11 to 3.4) with 4 patients in the dual therapy and 2 patients in the triple therapy arm developing virological failure [42]. In a multicenter, randomized, parallel, open-label, superiority trial, patients with multiple mutations including M184V at first-line failure and taking boosted PI plus 2 NRTIs with 2 viral load measurements <200 copies/mL were randomized to either monotherapy with boosted PI or boosted PI plus once daily lamivudine. At week 48, treatment failure was observed in 4 patients on dual therapy versus 33 patients on monotherapy (relative risk 8.2, 95% CI 3–22.5; odds ratio 10.6, 95% CI 3.6–42.1) with 21.8% difference between groups (95% CI 13.9–29.7; p<0.001) demonstrating superiority of dual therapy versus monotherapy [43].
Dolutegravir plus RPV may be considered for patients who should avoid NRTIs or in patients in whom DTG or RPV resistance is not expected. This recommendation was based on two phase III, multicenter, non- inferiority studies evaluating switching from a 3 or 4 drug antiretroviral regimen to DTG plus RPV in HIV-1 infected patients with viral load <50 copies/mL and no history of virologic failure. Switching patients to dual therapy was non-inferior in maintaining viral load suppression to continuing 3 or 4 drug ART at week 48 according to both the intent-to-treat [95% vs. 95%; difference: -0.4% (95% CI: -3.1%, 2.3%)] and per-protocol analysis [96% vs. 96%; difference: 0.7% (95% CI: -3.3%, 1.8%)]. Efficacy was comparable in both trials [44].
In virologically suppressed patients with history of treatment failure, new evidence suggests EVG/c/TAF/FTC plus DRV is an option to simplify treatment. Huhn et al. evaluated HIV-infected patients who were virologically suppressed with 2-3 class drug resistance and 2 or more previous regimen failures. After 24 weeks, simplification of drug regimen to EVG/c/TAF/FTC plus DRV was found to be non-inferior to continuation of baseline regimen in maintaining viral load suppression (96.6% vs. 91.3%, difference 5.3%, 95% CI, -3.4% - 17.4%). Elvitegravir/c/TAF/FTC plus DRV met noninferiority and superiority criteria by 48 weeks. Patients switched to EVG/c/TAF/FTC plus DRV regimen reported greater satisfaction and less missed doses [45].
There is currently insufficient evidence to recommend DTG plus 3TC as a maintenance strategy in virologically suppressed patients. ANRS 167 LAMIDOL trial is an ongoing open-label, single-arm, multicenter trial evaluating DTG plus 3TC in virologically suppressed patients on combination ART with 2 NRTIs and either PI, NNRTI, or INSTI. During Phase 1, the third drug was switched to DTG. During Phase 2, patients received daily combination of DTG plus 3TC. At week 24, 103 out of 104 patients maintained virological suppression [46]. In a pilot study of 20 treatment-naïve patients with no resistance and HIV RNA <100,000 copies/ mL at baseline and negative HBsAg evaluated DTG plus 3TC. Ninety percent of patients had plasma viral load <50 copies/mL by week 48 [13]. However, due to the study designs and short-term outcomes of these studies, DTG plus 3TC for maintenance therapy cannot be recommended at this time.
Special populations
Acute and recent (Early) HIV-1 infection: This section was updated to reflect findings of a recent study that showed that patients with early HIV-1 infection have non-specific and mild signs and symptoms [47]. Providers suspecting acute HIV infection even in the absence of reported high-risk behaviors should consider testing for HIV infection, especially in high prevalence areas. Because a positive result on a quantitative or qualitative plasma HIV-1 RNA test in the absence of a positive antibody test result, is highly indicative of an acute HIV infection, the diagnosis of HIV-1 infection should be later confirmed by subsequent documentation of HIV antibody seroconversion.
Antiretroviral therapy should be initiated before drug resistance test results are available. Acceptable regimens include a boosted DRV plus TDF/FTC or TAF/FTC. This is based on the rationale that resistance to boosted PIs emerges slowly. Dolutegravir pus either TDF or TAF are also acceptable options. No updated information was found in the sections addressing adolescents and young adults living with HIV, illicit drug users, women with HIV, patients with HIV-2 infection, pregnant women with HIV, or older adults.
Considerations for antiretroviral use in patients with coinfections
Hepatitis B/HIV virus coinfection: Recommendations for patients with HCV co-infection, HIV, and active hepatitis B virus infection (HBV) have been updated to include two agents with anti-HBV activity prior to initiating HCV therapy. This recommendation is based on a review of cases reported to the FDA Adverse Event Reporting System. The FDA identified 29 reports of HBV/HCV co-infected patients who received direct-acting antiviral drugs for HCV infection and experienced HBV reactivation. Two of these patients died and one required liver transplantation [48]. Wang et al. conducted an observational study of 327 patients receiving pan-oral direct-acting antiviral drugs for chronic HCV infection to evaluate the incidence of HBV reactivation in an area of China endemic for HBV. Ten patients developed hepatitis with three cases of HBV reactivation observed [49].
Acceptable agents include [TDF or TAF] plus [FTC or 3TC] prior to initiating HCV therapy. Patients with suspected HBV reactivation are those that have a current HBV infection and experience elevated liver enzymes during or immediately after HCV therapy.
Due to rapidly evolving treatment options for HCV infection, patients with HCV/HIV co-infection treated with all oral, direct-acting antiviral HCV regimen can achieve sustained HCV cure rates comparable to patients with HCV mono-infection [50-52]. The guidelines recommend that patients with chronic HCV/HIV infection should be screened for active and prior HBV infection by testing for the presence of hepatitis B surface antigen (HBsAg) and hepatitis B surface antibodies (HBsAb) and hepatitis B core antibodies (HBcAb) or IgG. Patients not immune to HBV infection (HBsAb-negative) should receive anti-HBV vaccination.
Drug interactions between antiretrovirals for HIV infection and direct-acting antivirals for HCV infection can be found in the drug interactions subsection.
Adverse effects
INSTI: The relationship between INSTIs, specifically DTG, and neuropsychiatric events is not clearly understood due to conflicting data. There are case reports and observational cohort studies discussing DTG and RAL-related neuropsychiatric adverse effects such as depression, anxiety, suicidal ideation and sleep disturbances [53-56]. A review of randomized controlled trials, however, did not find a significant difference in neuropsychiatric events including insomnia, depression, anxiety and suicidality between patients taking DTG and other ARTs including ATV/r, DRV/v, EFV, and RAL [57]. A report published by the World Health Organization stated that neuropsychiatric events were an INSTIclass effect and not specifically associated with DTG therapy [58]. These conflicting studies will need to be further investigated in order to draw conclusive recommendations. For now, it may be more appropriate to choose a non-DTG INSTI in patients with uncontrolled neuropsychiatric conditions to err on the side of caution.
Elvitegravir boosted with cobicistat has been added to the list of medications that may increase risk of cardiovascular events such as MI and ischemic stroke. Elvitegravir used by itself yields favorable lipid levels, however when combined with PK booster cobicistat, may result in increased total cholesterol and low-density lipoproteins levels. In a study comparing EVG/c/TDF/ FTC to ATV/r plus TDF/FTC, higher levels of fasting triglycerides and total cholesterol were observed in the EVG/c arm at weeks 48 and 96 [59]. However, a phase 2 study comparing ATV/r to ATV/c did not show a statistically significant difference in lipid levels between the two treatment arms [60]. Compared to EFV, however, EVG/c has less unfavorable effects on lipid levels [61]. It may be safer to initiate or switch patients to RAL, DTG, or RPV-containing regimens if there is baseline CV risk as these drugs have less lipid adverse effects.
NNRTI: The updated guidelines provide further clarification of adverse effects associated with EFV therapy. A retrospective cohort study published in 2016 did not find a correlation between EFV use and increased risk of suicidality [62]. Studies also show an increased risk of QTc prolongation in patients who are CYP2B6*6*6 allele carriers taking EFV-containing regimens [63,64]. Alternative treatment options should be initiated in patients taking other medications that increase QTc interval.
Efavirenz has also been associated with birth defects and was not recommended in women in the first trimester of pregnancy. A systematic review and meta-analysis assessed the prevalence of birth defects in infants exposed to EFV in the first-trimester of pregnancy. The study concluded that the prevalence ranged from 0-22.6% among the 19 studies identified with a 2% pooled prevalence (95% CI 0.82-3.18) with low heterogeneity between the analyzed studies. The pooled prevalence of birth defects among women taking non-EFV-based regimens in the first trimester of pregnancy was 2.9%. There was one case of neural tube defect among the studies with an incidence of 0.07% (95% CI 0.002-0.39). The authors of the meta-analysis concluded that there was no increased risk of birth defects among women taking EFV in their first trimester of pregnancy. For this reason, the new guidelines no longer recommend switching a pregnant woman’s EFV-containing regimen; however, the guidelines do recommend screening antenatal and postpartum women with HIV who are taking EFV due to risk of depression and suicidality [1,65].
Drug interactions
Recommendations for managing a particular interaction is influenced by whether a new antiretroviral drug is initiated in a patient who is on a stabilized concomitant medication or if a new concomitant medication is being initiated in a patient on a stable ART. The magnitude and significance of drug interactions are difficult to predict when several drugs with competing metabolic pathways are prescribed concomitantly [1]. The drug interaction updates for each class of ART, used in the first-line regimen, are summarized in Tables 1 and 2.
| Drug | Result of the interaction | Management | |||
| Anticonvulsants | |||||
| Carbamazepine with TAF |
↓ TAF AUC | Consider an alternative anticonvulsant | |||
| PIs | |||||
| Unboosted and boosted PIs plus TAF |
↑ AUC of TAF 10mg | No dose adjustment | |||
Table 1: Drug Interactions between Nucleoside Reverse Transcriptase Inhibitors and Other Drugs [1].
| Drug | Result of the interaction | Management | |
| Acid reducers | |||
| Ca-Containing Antacids plus RAL 1200mg |
↓ Cmax of RAL | RAL 1200mg once daily: do not co-administer | |
| Anticoagulants and Antiplatelets | |||
| Betrixaban plus EVG/c | ↑ betrixaban | Coadministration is not recommended. | |
| Consider alternative antiretroviral (e.g., an unboosted INSTI) or warfarin. |
|||
| Apixaban plus EVG/c | ↑ | apixaban | Coadministration is not recommended. Consider alternative antiretroviral (e.g., an unboosted INSTI) or warfarin. If coadministration is necessary, reduce apixaban dose by 50% and monitor for apixaban toxicity. |
| Dabigatran plus EVG/c | ↑ | dabigatran | Coadministration is not recommended. Consider alternative antiretroviral (e.g., another INSTI) or warfarin. |
| Edoxaban plus EVG/c | ↑ | edoxaban | |
| Rivaroxaban plus EVG/c | ↑ rivaroxaban | ||
| Anticonvulsants | |||
| Carbamazepine plus DTG | ↓ AUC of DTG | Increase DTG dose to 50 mg BID in treatment- naive or treatment-experienced, INSTI-naive patients. Use alternative anticonvulsant for INSTI- experienced patients with known or suspected INSTI resistance. |
|
| Carbamazepine plus RAL | ↓ RAL possible | Coadministration is not recommended. | |
| Phenobarbital Phenytoin plus RAL |
↓RAL possible | Coadministration is not recommended. | |
| Oxcarbazepine plus EVG/c | ↓ cobicistat possible | Consider alternative anticonvulsant. | |
| Antidepressants/Anxiolytics/Antipsychotics | |||
| Lurasidone plus EVG/c | ↑ | lurasidone | Contraindicated |
| Pimozide plus EVG/c | ↑ | pimozide | Contraindicated |
| Sertraline plus EVG/c | No change in levels but ↑ levels of other SSRIs possible |
No adjustment necessary for sertraline but other SSRIs should be initiated with lowest dose titrated carefully based on antidepressant response. |
|
| Antihyperglycemics | |||
| Saxagliptin plus EVG/c | ↑ saxagliptin | Limit saxagliptin dose to 2.5 mg once daily. | |
| Dapagliflozin/ Saxagliptin plus EVG/c | ↑saxagliptin | Do not co-administer, as this co-formulated drug contains 5 mg of saxagliptin | |
| Antimycobacterials | |||
| Rifampin plus RAL | ↓ AUC of RAL 400mg | RAL 800 mg BID, instead of 400 mg BID Do not co-administer RAL 1200 mg once daily with rifampin. |
|
| Rifepentin plus RAL | ↑ AUC with rifapentin once- weekly dose; ↓ RAL with once-daily rifapentin |
For once-weekly rifapentin, use standard RAL 400 mg BID doses. Do not co-administer with once-daily rifapentin. |
|
| Cardiac Medications | |||
| Ranolazine plus EVG/c | ↑ ranolazine | Contraindicated | |
| Corticosteroids | |||
| Beclomethasone Inhaled or intranasal plus EVG/c |
no change | No adjustment necessary | |
| Budesonide, Ciclesonide, Fluticasone, Mometasone; Inhaled or intranasal plus EVG/c | ↑glucocorticoid level | Coadministration can result in adrenal insufficiency and Cushing’s syndrome. Do not co-administer unless potential benefits of inhaled or intranasal corticosteroid outweigh the risks of systemic corticosteroid adverse effects. Consider |
|
| an alternative corticosteroid (e.g., beclomethasone) |
|||
| Betamethasone, Budesonide Systemic plus EVG/c |
↑ glucocorticoids ↓ PI |
Coadministration can result in adrenal insufficiency and Cushing’s syndrome. Do not co-administer unless potential benefits outweigh the risks | |
| Prednisone, Prednisolone Systemic plus EVG/c |
↑ prednisolone | ||
| Betamethasone Local injections, including intra- articular, epidural, or intra- orbital plus EVG/c |
↑ glucocorticoid | Do not co-administer. Coadministration may result in adrenal insufficiency and Cushing’s syndrome. | |
| Hepatitis C Direct Acting Antivirals | |||
| Glecaprevir/ Pibrentasvir plus DTG, RAL |
No significant effect | No dose adjustment necessary. | |
| Glecaprevir/ Pibrentasvir plus EVG/c | ↑ AUC of Glecaprevir, Pibrentasvir, and EVG |
No dose adjustment necessary | |
| Sofosbuvir/Velpatasvir plus all INSTIs |
No change | No dose adjustment necessary | |
| Sofosbuvir/ Velpatasvir/ Voxilaprevir plus EVG/c, DTG, or RAL | When given with Sofosbuvir/Velpatasvir/ Voxilaprevir (400/100/100 mg) + Voxilaprevir 100 mg: ↑Sofosbuvir AUC; no change in velpatasvir, and ↑ AUC of Voxilaprevir No change in levels of DTG, or RAL |
No dose adjustment necessary | |
| Hormonal Therapies | |||
| Hormonal Contraceptives plus DTG | No change in ethinyl estradiol, norgestimate, and DTG or RAL | No dose adjustment necessary. | |
| Hormonal Contraceptives plus EVG/c | ↑ drospirenone possible | Clinical monitoring is recommended, due to the potential for hyperkalemia. | |
| Menopausal Hormone Replacement Therapy plus DTG or RAL | With estradiol or conjugated estrogen (equine and synthetic): ↓ estrogen possible. No change in drospirenone, medroxyprogesterone, or micronized progesterone |
No dose adjustment necessary. | |
| Menopausal Hormone Replacement Therapy plus EVG/c | ↓estrogen ↑ drospirenone ↑ oral medroxyprogesterone ↑ oral micronized progesterone possible |
Adjust estrogen and progestin dose as needed based on clinical effects. | |
| Gender-Affirming Hormone Therapy plus DTG, RAL |
No change in estrogen | No dose adjustment necessary. | |
| Gender-Affirming Hormone Therapy plus DTG, EVG/c, RAL | No change in nasteride, goserelin, leuprolide acetate, spironolactone expected | No dose adjustment necessary. | |
| Gender-Affirming Hormone Therapy plus EVG/c | ↓ estradiol, increased dutasteride | Adjust dutasteride dosage as needed based on clinical effects and endogenous hormone concentrations. |
|
| Gender-Affirming Hormone Therapy plus EVG/c | ↑ testosterone possible | Monitor masculinizing effects of testosterone and for adverse effects and adjust testosterone dose as necessary. |
|
| Gender-Affirming Hormone Therapy plus DTG, RAL |
No change in testosterone level | No dose adjustment necessary. | |
| HMG-CoA Reductase Inhibitors | |||
| Atorvastatin plus EVG/c | ↑ AUC of atorvastatin | Do not exceed 20 mg of atorvastatin daily | |
| Immunosuppressants | |||
| Everolimus plus EVG/c | ↑ everolimus possible | Initiate with an adjusted immunosuppressant dose to account for potential increased concentration and monitor for toxicities. Therapeutic drug monitoring of immunosuppressant is recommended. Consult with specialist as necessary. |
|
| Miscellaneous Drugs | |||
| Alfuzosin plus EVG/c | ↑ alfuzosin | Contraindicated | |
| Calcifediol plus EVG/c | ↑ calcifediol | Dose adjustment of calcifediol may be required, and serum 25-hydroxyvitamin D, intact PTH, and serum Ca concentrations should be closely monitored. |
|
| Cisapride plus EVG/c | ↑ cisapride | Contraindicated | |
| Ergot derivatives plus EVG/c | ↑ dihydroergotamine, ergotamine, methylergonovine | Contraindicated | |
| Dronabinol plus EVG/c | ↑ dronabinol | Monitor for dronabinol-related adverse effects. | |
| Eluxadoline plus EVG/c | ↑ eluxadoline | Monitor for eluxadoline-related adverse effects. | |
| Metformin plus DTG | ↑ AUC, based on DTG dose | Start metformin at lowest dose and titrate based on glycemic control. Monitor for metformin adverse effects. |
|
Table 2: Drug Interactions between Integrase Strand Transfer Inhibitors and Other Drugs [1] *EVG is always coadministered with COBI.
Discussion and Conclusion
Integrase strand transfer inhibitors are in the forefront of HIV infection management for adults and adolescents.
This is mostly due to their better efficacy, high barrier to resistance development, and better tolerability profile compared to other classes of antiretrovirals. The co-formulation of DTG and boosted EVG with 2 NRTI backbones as a single-tablet regimen simplifies drug therapy for patients and boosts adherence rates. Uncertainty regarding the risk of neuropsychiatric events caused by DTG and EVG/c may be disconcerting for some prescribers. Psychiatric history is commonly seen in patients with HIV infection. Although it is not contraindicated to use DTG or EVG/c in the setting of psychiatric illness, they may not be the best initial option if there are other INSTIs available to the patient. If DTG or EVG/c is used in patients with psychiatric illness, closer monitoring of the illness is recommended in order to prevent worsening of condition.
Baseline CV risk should also be assessed in patients with HIV. Certain ARV should be avoided in patients with CV risk due to the uncertainty of these medications’ safety profile. These medications include some PIs such ad DRV, LPV/r, IDV, and fosamprenavir (FPV). Darunavir is more frequently used in in clinical practice compared to the other PI regimens and should be prescribed with caution in patients with CVD history. Although DRV is preferred over ATV because it has lesser risk of causing hyperbilirubinemia, increased CV events have not been observed in patients taking ATV-based regimens. For this reason, ATV may be a better option than DRV in patients with baseline CV risk, if a PI-based regimen is necessary. There is limited data that suggests that boosted EVG may increase risk of MI and ischemic stroke.
This may be due to increased total cholesterol and triglyceride levels associated with EVG/c use. If EVG/c is used in a patient with baseline CV risk and no kidney or bone disease, it would be advantageous to choose TDF/FTC over TAF/FTC as the backbone because TAF is also associated with lipid abnormalities.
Many medications increase the risk of QT prolongation to varying degrees and many times, the use of these medications is inevitable. The use of NNRTIs EFV and RPV should be avoided in patients already taking other QT- prolonging medications such as clarithromycin, if possible.
Monotherapy is not a recommended option for patients with HIV infection. However, patients with sustained virologic suppression on stable triple therapy for at least 6 months and no resistance mutations to DTG and RPV can de-escalate to maintenance, dual-therapy per the guidelines [1]. The first single-tablet, dual-therapy regimen containing DTG/RPV (Juluca®) was recently FDA approved as maintenance therapy. Dual-therapy is class-sparing which reduces the risk of resistance development. Taking two medications instead of three will also reduce risk of adverse effects for the patient.
Advancements in HIV pharmacotherapy is continuous with novel drugs coming down the pipeline such as investigational doravirine (DOR), a NNRTI expected to retain activity in the presence of common NNRTI mutations such as K103N [66]. To what extent DOR will impact HIV management and where its place will be within the guidelines is unknown. However, the increase in number of treatment options improves the management of treatment-experienced patients. Table 3 contains a summary of the current updates in guidelines reviewed in this article.
| Summary | |
| What to start | •All INSTI-based regimens are recommended for most people with HIV infection. •Boosted DRV is now recommended in certain clinical situations due to its risk of CV events. •TAF and TDF are recommended for most people with HIV infection with the same level of recommendation (AI) except RAL plus TAF/FTC (AII). •Once-daily RAL is recommended as initial therapy for most people with HIV infection and is given the same level of recommendation as twice- daily RAL (AI). |
| What not to use | •Delavirdine, ddI, NFV, IDV, d4T are no longer recommended to use in patients with HIV infection due to risk of serious adverse effects. •PI-monotherapy and INST-monotherapy is not recommended. •Combination TAF plus TDF and ritonavir plus cobicistat is not recommended as this would be duplication of therapy. |
| Management of Treatment-Experienced Patient: virologic failure | •In patients with HBV/HIV coinfection whose ARV regimen is being switched, drugs active against HBV should be continued to prevent reactivation of HBV. •An INSTI + 2 NRTIs is an option following failure of 1st line NNRTI- based therapy •ARV options for patients with virologic failure (first and second regimen failure) are described in the figures in this article. |
| Management of Treatment-Experienced Patient: regimen switching in the setting of virologic suppression | •Monotherapy with a boosted PI or INSTI as a switching strategy is not recommended. •In switching regimens for patients with HBV/HIV coinfection, drugs active against HBV infection should be continued to avoid reactivation of HBV. |
| Special populations | Acute and Recent (Early) HIV-1 Infection •Providers suspecting an acute HIV infection even in the absence of reported high-risk behaviors should consider a potential HIV diagnosis, especially in high prevalence areas. •Antiretroviral therapy (ART) should be initiated before drug resistance test results are available; acceptable regimens include boosted darunavir (DRV) and emtricitabine (FTC) plus either tenofovir disoproxil fumarate (TDF) or tenofovir alafenamide (TAF) |
| Co-infections | Hepatitis B/HIV virus coinfection •Include two agents with anti-HBV activity prior to initiating HCV therapy. |
Table 3: Summary of updates in HIV guidelines [1].
References
- Department of Health and Human Services. 2018 Adults and adolescents living with HIV guidelines for the use of antiretroviral agents in adults and adolescents living with HIV developed by the DHHS panel on antiretroviral guidelines for adults. Aidsinfo.
- Department of Health and Human Services. (2016) Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Aidsinfo.
- Lang S, Mary-Krause M, Cotte L, Gilquin J, Partisani M, et al. (2010) Impact of individual antiretroviral drugs on the risk of myocardial infarction in human immunodeficiency virus-infected patients. Arch Intern Med 170: 1228-1238.
- Worm SW, Sabin C, Weber R, Reiss P, El-Sadr W, et al. (2010) Risk of myocardial infarction in patients with HIV infection exposed to specific individual antiretroviral drugs from the 3 major drug classes: the data collection on adverse events of anti‐ HIV drugs (D:A:D) study. J Infect Dis 201: 318-330.
- Ryom L, Lundgren JD, El-Sadr WM, Reiss P, Phillips A, et al. (2017) Association between cardiovascular disease and contemporarily used protease inhibitors.
- Stein JH, Ribaudo HJ, Hodis HN, Brown TT, Tran TT, et al. (2015) A prospective, randomized clinical trial of antiretroviral therapies on carotid wall thickness. AIDS 29: 1775-1783.
- De Saint-Martin L, Bressollette L, Perfezou P, Bellein V, Ansart S, et al. (2010) Impact of atazanavir-based HAART regimen on the carotid intima-media thickness of HIV-infected persons: A comparative prospective cohort. AIDS 24: 2797-2801.
- Squires K, Kityo C, Hodder S, Johnson M, Voronin E, et al. (2016) Integrase inhibitor versus protease inhibitor based regimen for HIV-1 infected women (WAVES): a randomised, controlled, double-blind, phase 3 study. Lancet HIV 3: e410-e420.
- Orrell C, Hagins DP, Belonosova E, Porteiro N, Walmsley S, et al. (2017) Fixed-dose combination dolutegravir, abacavir, and lamivudine versus ritonavir-boosted atazanavir plus tenofovir disoproxil fumarate and emtricitabine in previously untreated women with HIV-1 infection (ARIA): week 48 results from a randomised, open-label. Lancet HIV 4: e536-e546.
- Mills A, Crofoot G, McDonald C, Shalit P, Flamm JA, et al. (2015) Tenofovir alafenamide vs. tenofovir disoproxil fumarate in the first protease inhibitor-based single tablet regimen for initial HIV-1 therapy: a randomized phase 2 study. J Acquir Immune Defic Syndr 69: 439-445.
- Arribas JR, Thompson M, Sax PE, Haas B, McDonald C, et al. (2017) Randomized, double-blind comparison of tenofovir alafenamide (TAF) vs tenofovir disoproxil fumarate (TDF), each coformulated with elvitegravir, cobicistat, and emtricitabine (E/C/F) for initial HIV-1 treatment: week 144 results. J Acquir Immune Defic Syndr 75: 211-218.
- Cahn P, Kaplan R, Sax PE, Squires K, Molina JM, et al. (2017) Raltegravir 1200 mg once daily versus raltegravir 400 mg twice daily, with tenofovir disoproxil fumarate and emtricitabine, for previously untreated HIV-1 infection: a randomised, double-blind, parallel-group, phase 3, non-inferiority trial. Lancet HIV 4: e486-e494.
- Cahn P, Rolon MJ, Figueroa MI, Gun A, Patterson P, et al. (2017) Dolutegravir-lamivudine as initial therapy in HIV-1 infected, ARV-naive patients, 48-week results of the PADDLE (Pilot Antiretroviral Design with Dolutegravir LamivudinE) study. J Int AIDS Soc 20: 1-7.
- Taiwo BO, Zheng L, Stefanescu A, Nyaku A, Bezins B, et al. (2017) ACTG A5353: A pilot study of dolutegravir plus lamivudine for initial treatment of HIV-1-infected participants with HIV-1 RNA <500,000 copies/mL. Clin Infect Dis 66: 1639-1697.
- Sued O, Figueroa MI, Gun A, A Gun, W Belloso, D Cecchin, et al. (2017) Dual therapy with darunavir/ritonavir plus lamivudine for HIV-1 treatment initiation: week 24 results of the randomized ANDES study. In: Paris, France: IAS Conference on HIV Science 561.
- Bristol-Myers Squibb (2017) Zerit package insert.
- Bristol-Myers Squibb (2018) Videx package insert.
- Merck. Crixivan package insert (2018).
- ViiV Healthcare. Rescriptor package insert (2012).
- Girard PM, Antinori A, Arribas JR, Ripamonti D, Bicer C, et al. (2017) Week 96 efficacy and safety of darunavir/ritonavir monotherapy vs. darunavir/ritonavir with two nucleoside reverse transcriptase inhibitors in the PROTEA trial. HIV Med 18:5-12.
- Antinori A, Clarke A, Svedhem-Johansson V, Arribas JR, Arenas-Pinto A, et al. (2015) Week 48 efficacy and central nervous system analysis of darunavir/ritonavir monotherapy versus darunavir/ ritonavir with two nucleoside analogues. AIDS 29: 1811-1820.
- JR Arribas, N Clumeck, M Nelson, A Hill, Y van Delft, et al. (2012) 144 analysis of the efficacy of darunavir/ritonavir (DRV/r) monotherapy versus DRV/r plus two nucleoside reverse transcriptase inhibitors, for patients with viral load <50 HIV-1 RNA copies/mL at baseline. HIV Med 13: 398-405.
- Gianotti N, Poli A, Galli M, Pan A, Rizzardini G, et al. (2014) Monotherapy with lopinavir/ritonavir versus standard of care in HIV- infected patients virologically suppressed while on treatment with protease inhibitor-based regimens: Results from the MoLo Study. New Microbiol 37: 439-448.
- Gianotti N, Galli L, Maserati R, Sighinolfi L, Ripamonti D, et al. (2016) Monotherapy with darunavir/ritonavir or lopinavir/ritonavir versus standard antiretroviral therapy: A randomized clinical trial (2pm study). New Microbiol 39: 290-294.
- Galli L, Spagnuolo V, Bigoloni A, D'Arminio Monforte A, Montella F, et al. (2016) Atazanavir/ritonavir monotherapy: 96 week efficacy, safety and bone mineral density from the MODAt randomized trial. J Antimicrob Chemother. 71: 1637-1642.
- Castagna A, Spagnuolo V, Galli L, Vinci C, Nozza S, et al. (2014) Simplification to atazanavir/ritonavir monotherapy for HIV-1 treated individuals on virological suppression: 48-week efficacy and safety results. AIDS 28: 2269-2279.
- Gilks CF, Walker AS, Dunn DT, Gibb DM, Kikaire B, et al. (2012) Lopinavir/ritonavir monotherapy after 24 weeks of second-line antiretroviral therapy in Africa: A randomized controlled trial (SARA). Antivir Ther 17: 1363-1373.
- Paton N, Stohr W, Arenas-Pinto A, Fisher M3, Williams I, et al. (2015) Protease inhibitor monotherapy for long-term management of HIV infection: A randomised, controlled, open-label, non-inferiority trial. Lancet HIV 2: 417-426.
- European AIDS Clinical Society. (2017) European AIDS Clinical Society Guidelines. European AIDS Clinical Society.
- Stöhr W, Dunn DT, Arenas-Pinto A, Orkin C, Clarke A, et al. (2016) Factors associated with virological rebound in HIV-infected patients receiving protease inhibitor monotherapy. AIDS 30: 2617-2624.
- Brenner BG, Thomas R, Blanco JL, Ibanescu RI, Oliveira M, et al. (2016) Development of a G118R mutation in HIV-1 integrase following a switch to dolutegravir monotherapy leading to cross-resistance to integrase inhibitors. J Antimicrob Chemother 71: 1948-1953.
- Oldenbuettel C, Wolf E, Ritter A, Noe S, Heldwein S, et al. (2017) Dolutegravir monotherapy as treatment de-escalation in HIV- infected adults with virological control: DoluMono cohort results. Antivir Ther 22: 169-172.
- Aboud M, Kaplan R, Lombaard J, Zhang F, Hidalgo J, et al. (2017) Superior efficacy of dolutegravir (DTG) plus 2 nucleoside reverse transcriptase inhibitors (NRTIs) compared with lopinovir/ritonavir (LPV/RTV) plus 2 NRTIs in second-line treatment: interim data from the DAWNING study.
- La Rosa AM, Harrison LJ, Taiwo B, Wallis CL, Zheng L, et al. (2016) Raltegravir in second-line antiretroviral therapy in resource- limited settings (SELECT): a randomised, phase 3, non-inferiority study. Lancet HIV 3: 247-258.
- U.S. Food and Drug Administration. (2018) FDA approves new HIV treatment for patients who have limited treatment options.
- Wijting I, Rokx C, Boucher C, Pas S, de Vries-Sluijs T, et al. (2017) Dolutegravir as maintenance monotherapy for HIV (DOMONO): a phase 2, randomised non-inferiority trial. Lancet HIV 4: 547-554.
- Blanco JL, Oldenbuettel C, Thomas R, Josep Mallolas, Eva Wolf, et al. (2017) Pathways of resistance in subjects failing dolutegravir monotherapy. In: Conference on Retroviruses and Opportunistic Infections.
- Van Lunzen J, Pozniak A, Gatell J, Antinori A, Klauck I, et al. (2016) Switch to ritonavir-boosted atazanavir plus raltegravir in virologically suppressed patients with HIV-1 infection: A randomized pilot study. J Acquir Immune Defic Syndr 71: 538-543.
- Pett SL, Amin J, Horban A, Andrade-Villanueva J, Losso M, et al. (2016) Maraviroc, as a switch option, in HIV-1-infected individuals with stable, well-controlled HIV replication and R5-tropic virus on their first Nucleoside/Nucleotide reverse transcriptase inhibitor plus ritonavir-boosted protease inhibitor regimen: Week 48 R. Clin Infect Dis 63: 122-132.
- Katlama C, Assoumou L, Valantin MA, Soulié C, Duvivier C, et al. (2014) Maraviroc plus raltegravir failed to maintain virological suppression in HIV-infected patients with lipohypertrophy: results from the ROCnRAL ANRS 157 study. J Antimicrob Chemother 69: 1648-1652.
- Pett SL, Amin J, Horban A, Andrade-Villanueva J, Losso M, et al. (2018) Week 96 results of the randomized, multicentre Maraviroc Switch (MARCH) study. HIV Med 19: 65-71. doi:10.1111/hiv.12532.
- Pulido F, Ribera E, Lagarde M, Pérez-Valero I, Palacios R, et al. (2017) Dual therapy with darunavir and ritonavir plus lamivudine vs triple therapy with darunavir and ritonavir plus tenofovir disoproxil fumarate and emtricitabine or abacavir and lamivudine for maintenance of human immunodeficiency virus type 1 viral suppression: Randomized, Open-Label, Noninferiority DUAL-GESIDA 8014-RIS-EST45 Trial. Clin Infect Dis 65: 2112-2118.
- Ciaffi L, Koulla-Shiro S, Sawadogo AB, Ndour CT, Eymard-Duvernay S, et al. (2017) Boosted protease inhibitor monotherapy versus boosted protease inhibitor plus lamivudine dual therapy as second-line maintenance treatment for HIV-1-infected patients in sub-Saharan Africa (ANRS12 286/MOBIDIP): a multicentre, randomised, parallel, open-la. Lancet HIV 4: 384-392.
- Llibre JM, Chien-Ching H, Brinson C, Francesco Castelli, Pierre-Marie Girard, et al. (2017) Phase III sword 1 & 2: switch to DTG + RPV maintains virologic suppression through 48 weeks. In: Conference on Retroviruses and Opportunistic Infections.
- Huhn GD, Tebas P, Gallant J, Timothy Wilkin, Andrew Cheng, et al. (2017) A Randomized, open-label trial to evaluate switching to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide plus darunavir in treatment-experienced HIV-1- infected adults. J Acquir Immune Defic Syndr 74: 193-200.
- Joly V, Burdet C, Landman R, François Raffi, Christine Katlama ,et al. (2017) Promising results of dolutegravir + lamivudine maintenance in ANRS 167 LAMIDOL trial. In: Conference on Retroviruses and Opportunistic Infections.
- Robb ML, Eller LA, Kibuuka H, Rono K, Maganga L, et al. (2016) Prospective study of acute HIV-1 infection in adults in East Africa and Thailand. N Engl J Med 374: 2120-2130.
- Bersoff-Matcha SJ, Cao K, Jason M, Ajao A, Jones SC, et al. (2017) Hepatitis B virus reactivation associated with direct-acting antiviral therapy for chronic hepatitis C virus: a review of cases reported to the U.S. Food and Drug Administration Adverse Event Reporting System. Ann Intern Med 166: 792-798.
- Wang C, Ji D, Shao Q, Shao Q, Li B, et al. (2017) Hepatitis due to reactivation of hepatitis B virus in endemic areas among patients with hepatitis C treated with direct-acting antiviral agents. Clin Gastroenterol Hepatol 15: 132-136.
- Naggie S, Cooper C, Saag M, Workowski K, Ruane P, et al. (2015) Ledipasvir and sofosbuvir for HCV in patients coinfected with HIV-1. N Engl J Med 373: 705-713.
- Sogni P, Gilbert C, Lacombe K, Piroth L, Rosenthal E, et al. (2016) All-oral direct-acting antiviral regimens in HIV/Hepatitis C virus- coinfected patients with cirrhosis are efficient and safe: real-life results from the prospective ANRS CO13- HEPAVIH cohort. Clin Infect Dis 63: 763-770.
- Rockstroh JK, Nelson M, Katlama C, Lalezari J, Mallolas J, et al. (2015) Efficacy and safety of grazoprevir (MK-5172) and elbasvir (MK- 8742) in patients with hepatitis C virus and HIV co-infection (C-EDGE CO-INFECTION): a non- randomised, open-label trial. Lancet HIV 2: 319-327.
- Kheloufi F, Allemand J, Mokhtari S, Default A (2015) Psychiatric disorders after starting dolutegravir: report of four cases. AIDS 29: 1723-1725.
- Harris M, Larsen G, Montaner JS (2008) Exacerbation of depression associated with starting raltegravir: a report of four cases. AIDS 22: 1890-1892.
- Peñafiel J, De Lazzari E, Padilla M, Rojas J, Gonzalez-Cordon A, et al. (2017) Tolerability of integrase inhibitors in a real-life setting. J Antimicrob Chemother 72: 1752-1759.
- Hoffmann C, Welz T, Sabranski M, Kolb M,Wolf E, et al. (2017) Higher rates of neuropsychiatric adverse events leading to dolutegravir discontinuation in women and older patients. HIV Med 18: 56-63.
- Fettiplace A, Stainsby C, Winston A, Givens N, Puccini S, et al. (2017) Psychiatric symptoms in patients receiving dolutegravir. J Acquir Immune Defic Syndr 74: 423-431.
- Kheloufi F, Boucherie Q, Blin O, Micallel J (2017) Neuropsychiatric events and dolutegravir in HIV patients: a worldwide issue involving a class effect. AIDS 31: 1775-1777.
- Rockstroh JK, Dejesus E, Henry K, Molina JM, Gathe J, et al. (2013) A randomized, double-blind comparison of coformulated elvitegravir/ cobicistat/emtricitabine/tenofovir DF vs ritonavir-boosted atazanavir plus coformulated emtricitabine and tenofovir DF for initial treatment of HIV-1 infection: Analysis of week 96 result. J Acquir Immune Defic Syndr 62: 483-486.
- Elion R, Cohen C, Gathe J, Shalit P, Hawkins T, et al. (2011) Phase 2 study of cobicistat versus ritonavir each with once-daily atazanavir and fixed-dose emtricitabine/tenofovir df in the initial treatment of HIV infection. AIDS 25: 1881- 1886.
- Zolopa A, Sax PE, Dejesus E, Mills A, Cohen C, et al. (2013) A randomized double-blind comparison of coformulated elvitegravir/ cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: Analysis of week 96 results. J Acquir Immune Defic Syndr 63: 96-100.
- Nkhoma ET, Coumbis J, Farr AM, Johnston SS, Chu BC, et al. (2016) No evidence of an association between efavirenz exposure and suicidality among HIV patients initiating antiretroviral therapy in a retrospective cohort study of real-world data. Medicine (Baltimore) 95: 2480.
- Squibb B-M. (2017) Sustiva package insert.
- Abdelhady AM, Shugg T, Thong N, Lu JBL, Kreutz Y, et al. (2016) Efavirenz inhibits the human ether-a-go-go related current (hERG) and induces QT interval prolongation in CYP2B6*6*6 allele carriers. J Cardiovasc Electrophysiol 27: 1206-1213.
- Ford N, Calmy A, Mofenson L (2011) Safety of efavirenz in the first trimester of pregnancy: An updated systematic review and meta-analysis. AIDS 25: 2301-2304.
- Molina JM, Squires K, Sax PE, Cahn P, Lombaard J, et al. (2018) Doravirine versus ritonavir-boosted darunavir in antiretroviral-naive adults with HIV-1 (DRIVE-FORWARD): 48-week results of a randomised, double-blinded, phase 3, non- inferiority trial. Lancet HIV 5: 211-220.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences