On a Case of Inflammatory Myofibroblastic Tumor of the Urinary Bladder: An Unexpected Diagnosis
Surgical Pathology Service, ASL AL, Santo Spirto Hospital, Casale Monferrato, Italy
- *Corresponding Author:
- Stefania Erra
Surgical Pathology Service, ASL AL
Santo Spirto Hospital
Casale Monferrato, Italy
Tel: 0142434716
Email: stefania.erra@virgilio.it
Received Date: 16 March, 2018; Accepted Date: 13 April 2018; Published Date: 23 April 2018
Citation: Erra S, Zambello L (2018) On a case of Inflammatory Myofibroblastic Tumor of the Urinary Bladder: An Unexpected Diagnosis. J Mol Sci. 2:8.
Abstract
Inflammatory myofibroblastic tumor is a particular mesenchymal neoplasm caused by a proliferation of fibroblastic spindle cells in a myxoid stroma with an inflammatory background. Typically, the lesion occurs in young patients with a female predominance. It may affect a wide range of anatomic sites, but the majority of cases are described in lung and intra-abdominal soft tissue. The case described in this report regards a 28-year-old woman with a polypoid urinary bladder mass, without any anamnestic pathological history.
Keywords
Inflammatory myofibroblastic tumor; Urinary bladder
Introduction
Primitive sarcomas of the urinary bladder are rare tumors, described only in a limited number of cases. Many of them represent round cell Ewing-like sarcomas [1] or spindle cell neoplasms with muscular differentiation. Myofibroblastic proliferations are difficult to classify and differential diagnosis include many malignant tumors of mesenchymal origin, as leiomyosarcoma, rhabdomyosarcoma, angiosarcoma and fibrous histiocytoma [2]. It is important to differentiate between benign and malignant lesions to choose the right therapy. Unfortunately the symptomatology and the instrumental aspect of the lesions are not useful for the differential diagnosis, as they are superimposable in different tumors. Masses of urinary bladder, in fact, mainly manifest with nonspecific symptoms, like hematuria, irritative and obstructive voiding symptoms and abdominal pain. Rarely it can also be notice loss of weight as a result of the release of cytokines.
Inflammatory myofibroblastic tumor can have three different histologic patterns: nodular fascitis-like pattern (most common), with myxoid, vascular and inflammatory areas; fibrous histiocytoma-like one, characterized by a more compact spindle cell proliferation and scattered inflammatory infiltrate and desmoid-like aspect, with dense collagen stroma and only fewer spindle and inflammatory cells [3]. There is no evidence of metastases, but the tumor may recur after surgery.
Case Report
In the present article, an exceptional case of inflammatory myofibroblastic tumor of the bladder is reported. A 28-yearold woman arrived at a peripheral hospital with a conspicuous hematuria. An ultrasound examination revealed the presence of a circular whitish mass of 4 cm in diameter. It was located in parietal site, with an intraluminal protruding. The patient undergoes to a trans-urethral resection of the mass and the fragments obtained have been investigated in surgical pathology. The specimen has been formalin fixed and paraffin embedded in its totality that is 38 grams of total weight.
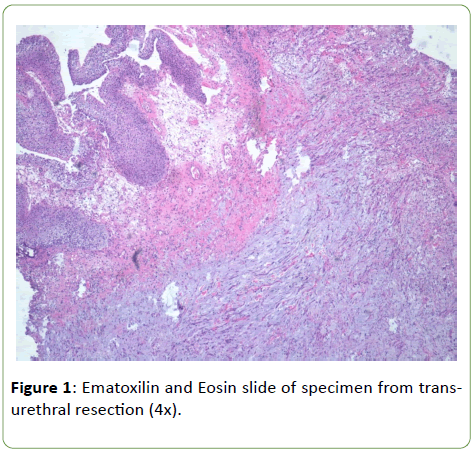
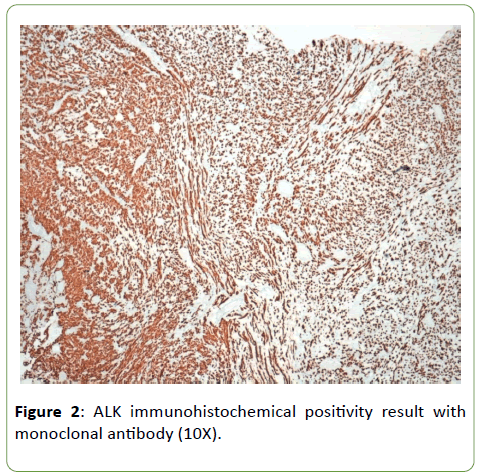
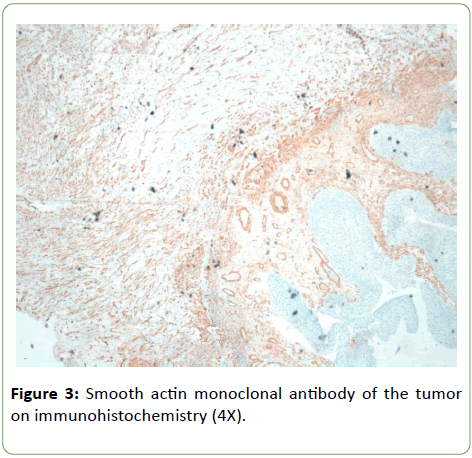
Histologically tumor is composed of spindle and circular cells with small nucleus in a myxoid background with presence of conspicuous inflammatory cells (Figure 1). Neoplastic elements invaded muscular tissue with necrosis too. Immunophenotypical definition of the neoplasm was made for a correct differential diagnosis. Neoplastic elements have resulted positive for ALK monoclonal antibody (Figure 2); negative for myogenin and S100 protein anibodies; negative were some markers of endothelial and epithelial differentiations such as CD31 and EMA, while PanK and calponin resulted positive, with a focal positivity for musclesmooth actin monoclonal antibody too (Figure 3). On the basis of morphological aspects and strong positivity for ALK monoclonal antibody, consistent with the immunophenotypical picture, the diagnosis of inflammatory myofibroblastic tumor was made.
Discussion
Differential diagnosis is important in all malignant neoplasia, but in urinary bladder it can be difficult to recognize malignancy from urothelial origin rather than tumors of mesenchymal one. This consideration is supported by the much undifferentiated morphological aspect of some urothelial lesions. For this reason, the epithelial origin of neoplasia must even consider in urinary bladder, especially in the case of lesions vegetating in the lumen. In fact, most of urinary bladder tumors are carcinomas, while primitive sarcomas are very rare.
Inflammatory myofibroblastic tumor can mimic both sarcomatoid urinary carcinomas than sarcomas with spindle pattern of growth. The first case of inflammatory myofibroblastic proliferation in urinary bladder was reported in 1980 [4]. Nowadays, forty-one studies have been identified on this pathological entity, and 182 patients were included for review and subsequent analyses for the presence of traslocation of anaplastic lymphoma kinase [5]. The most of revisited cases (65%) were ALK-positive with immunostaining method [6]. About this, it seems that immunohistochemistry for ALK mutated protein detection correlates well with the presence of ALK gene rearrangement [7].
Local tumor recurrence rate of this tumor is 4%, and no cases of distant metastases have been reported. Compared with ALK-negative IMTs, ALK-positive IMTs has a female predilection with a sex ratio (male:female) of 1:1.67 (P =0.048). ALK-positive IMTs also seem to occur in younger patients (P=0.072). No significant differences were noted in terms of their clinical presentations and histologic features [5,6]. On immunohistochemical staining, ALK-positive inflammatory myofibroblastic tumors have more positive results for desmin (P=0.042) and p53 (P=0.05), and more negative results for clusterin (P=0.003).
The significance of the presence of the mutated ALK protein in some inflammatory myofibroblastic tumors seems to have a prognostic value and a predictive response to targeted therapy. In fact, some nonresectable tumors undergone pharmacotherapy with ALK inhibitors [8,9].
The detection of ALK mutated protein in a tumor with myofibroblastic proliferation in an inflammatory background can be useful for differential diagnosis with other neoplastic entities, because many monoclonal antibodies do not have an unequivocal result. Cytocheratin antibodies are seen positive in IMT, but they are positive in urothelial carcinoma and in some other not epithelial tumor. Actine immunoreactivity may not help, because alfa-smooth muscle actin (SMA) is positive in 43% of sarcomas and in 63% of inflammatory myofibroblastic tumors and also in more than 50% of postoperative spindle cell nodule. Inflammatory myofibroblastic tumor can be confused with myxoid leiomyosarcoma because both have myxoid stroma, even if leiomyosarcoma is more uniform in its cellularity and have more cytologic atypia.
Conclusion
At last, ALK-positive IMTs of the urinary bladder has a female predilection, it occurs more frequently in younger patients. Differential diagnosis can be made on surgical specimen only, with an exhaustive sampling of the material send to surgical pathology service. Morphological appearance of the neoplastic cells, myxoid stroma and inflammatory background suggest the correct diagnosis. The presence of ALK mutated protein detected with immunohistochemistry has an important value in supporting diagnosis of inflammatory myofibroblastic tumor.
Regardless of this observation, ALK gene mutation in IMT of the urinary bladder seems to correlate with a good prognosis after surgical resection, above all for the possibility to response with molecular target therapies [10]. In fact about half of the inflammatory myofibroblastic tumors rearrange the Anaplastic Lymphoma Kinase (ALK) locus from chromosome 2p23 to chromosome 17p23, causing ALK aberrant protein expression. ALK inhibitor Crizotinib can give total response in patients with ALK-rearranged inflammatory myofibroblastic tumor [11], while no results have been observed in tumors without ALK translocation. These data emphasizes the role of ALK chimeric protein seems to employ in intracellular signaling during carcinogenesis. The statement of ALK mutated protein in IMT suggests a therapeutic strategy in these soft tissue tumors.
References
- Tonyali S, Yazici S, Yesilimark A, Ergen A (2016) The Ewing’s sarcoma family of tumors of urinary bladder: a case report and rewiew of literature. Balkan Med J 33: 462-466.
- Machioka K, Kitagawa Y, IzumiK, Kitamura S, Ikeda H, et al. (2014) Inflammatory myofibroblastic tumor of the urinary bladder with benign pelvic lymph node enlargement: a case report. Case Rep Oncol 7: 571-575.
- Rosai, Ackerman (2011) Surgical Pathology. 10th edition, page: 300-306.
- Roth J (1980) Reactive pseudosarcomatous response in urinary bladder. Urology 16: 635-637.
- Teoh JY, Chan NH, Cheung HY, Hou SS, Ng CF (2014) Infiammatory myofibroblastic tumor of urinary bladder, a sistematic review. Urology 84: 503-508.
- Freeman A, Geddes N, Munson P, Joseph J, Ramani P, et al. (2004) Anaplastic lymphoma kinase (ALK1) staining and molecolar analysis in inflammatory myofibroblastic tumor of the urinary of the bladder: a preliminary clinicopatological study of nine cases and rewiew of the literature. Mod Pathol 17: 765-771.
- Toshiki E, Taku N, Takashi N, KeitaroI, Ryosuke A, et al. (2016) Inflammatory myofibroblastic tumor of the urinary bladder: a case report. Case Rep Oncol 9: 464-469.
- Applebaum H, Kieran MW, Cripe TP, Coffin CM, Collins MH, et al. (2005) The rationale for nonsteroidal anti-inflammatory drug therapy for inflammatory myofibroblastic tumors: a children’s oncology group study. J Pediatr Surg 40: 999-1003.
- Chavez C, Hoffman MA (2013) Complete remission of ALK-negative plasma cell granuloma( inflammatory myofibroblastic tumor) of the lung induced by celecoxib: a case report and rewiew of literature. Oncol Letter 5: 1672-1676.
- Ryan CW, Desai J (2013) The past, present, and future of cytotoxic chemotherapy and pathway-directed targeted agents for soft tissue sarcoma. Am Soc Clin Oncol Educ Book.
- Butrynski JE, D’ Adamo DR, HornickJL, Dal Cin P, Antonescu CR, et al. (2010) Crizotinib in ALK-rearranged inflammatory myofibroblastic tumor. N Engl J Med 18: 1727-1733.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences