Ultrastructural Observation of Pseudoamphisiella lacazei Reveals More Morphological Features and Provides Taxonomical Evidence
Cai X1*, Chen Y1*, Pan X1, Zhang S1*, Wang Y1, Jing X1, Langlois GA2 and Qiu Z1*
1Lab of Protozoology, College of Life Science and Technology, Harbin Normal University, Harbin, China
2College of Arts and Science, Bryant University, Smithfield, USA
- Corresponding Author:
- Zijian Qiu & Ying Chen
College of Life Science & Technology
Harbin Normal University, Harbin
Heilongjiang, China
Tel: 13836018162
E-mail: Qiuzijian48@163.com
- Co-Author:
- Cai X and Chen Y
Lab of Protozoology, College of Life Science and Technology
Harbin Normal University, Harbin, China
Received date: April 10, 2017; Accepted date: April 25, 2017; Published date: May 02, 2017
Citation: Cai X, Chen Y, Pan X, et al. Ultrastructural Observation of Pseudoamphisiella lacazei Reveals More Morphological Features and Provides Taxonomical Evidence. Insights Aquac Cult Biotechnol 2017, 1:1.
Abstract
Ultrastructure of Pseudoamphisiella lacazei was investigated by transmission electron microscopy (TEM) and scanning electron microscopy (SEM) with an improved sample preparation method. The major results included: 1) A vacuole layer which had been considered absent in this species was found, which indicated that the cortex layers should thus contained three layers: a smooth membrane, a vacuole layer, and a fiber layer. 2) Extrusomes were found to distribute among the cortex vacuoles, and they were supposed to be a type of mucocyst which were similar to the mucocyst of Urostylida based on their shape and structure. 3) Some bacteria were observed to be attached to the fiber layer as if they were endosymbionts of P. lacazei. In order to analyze the features and taxonomical position of Pseudoamphisiella, morphological features of all four species of Pseudoamphisiella were compiled and compared. Our results suggested that ultrastructure observation could supplement and correct the gross and micro-level observation and provide additional evidence to the taxonomical argument.
Keywords
Ciliate; Electron microscopy; Morphology
Introduction
Hemberger placed Pseudoamphisiella in Family Amphiellidae, but Song transferred it to a new family Pseudoamphisiellidae based on the origin of the right marginal cirri and the absence of front-o-terminal cirri, making the group sufficiently different from other hypotrichs (Order Hypotrichida) [1-3].
The main features of genus Pseudoamphisiella according to Song [2] include a long elliptical body with one left and one right marginal cirri row; frontal, buccal and caudal cirri are present; extremely well-developed transverse cirri; two parallel and widely-spaced ventral rows that derive from numerous frontmidventral transverse cirri anlagen; and conspicuous extrusomes which are commonly located in an outer alveolar layer. This genus contains four known species, P. lacazei [2] (formerly Holosticha lacazei), P. alveolata [4] Song and Warren (formerly Holosticha alveolata) [5], P. quadrinucleata Chen et al. and P. elongate Li et al. [6,7]. This new genus has been widely investigated using various methods, including living observations, protargol impregnation, and small subunit ribosomal rRNA (SSU rRNA) gene sequencing [3-9]. Pseudoamphisiella lacazei was selected to be the type species of this genus. This free-living species has been recorded in marine habitats in the northern China seas. It swims relatively rapid but with long stationary periods, and feeds on bacteria, debris, small ciliates or flagellates [2,10]. However, no ultrastructure has not yet been described.
In this paper, some new ultrastructure characteristics of P. lacazei cortex and cytoplasm observed by transmission electron microscopy (TEM) and scanning electron microscopy (SEM) were added to the diagnosis of this species. We also compared the available morphological features of living and protargol staining observations of all four species of Pseudoamphisiella.
Materials and Methods
Microorganisms
Pseudoamphisiella lacazei, provided by Prof. Weibo Song, Ocean University of China, was collected from coastal waters of the Yellow Sea. Samples were cultured in Petri dishes with boiled seawater with the salinity about 31%, water temperature 18 and pH 8.1 [2]. Some rice grains were added to promote the growth of bacteria.
Method of SEM observation
Samples were prepared according to the method described by Gao et al. [11]. The ciliates were fixed in 1% KMnO4 and 100% glacial acetic acid at room temperature for 15 min. After washing with double distilled water for several times, washing off the cytoplasm, the samples were then conventionally dehydrated, freeze dried and gold sprayed. The samples were then observed by a HITACHI S-3400 SEM.
Method of TEM observation
Samples were prepared according to the method described by Gu et al. [12]. They were fixed by 2% OsO4 and 5% glutaraldehyde at 4°C for 30 min. After washing with the cacodylate buffer, followed by conventional dehydration, the samples were embedded with Epon 812 and successively polymerized at 40°C for 20 h and at 60°C for 48 h. The embedded samples were then ultrathinsectioned and stained with uranyl acetate and citrate lead before they were observed under a FEI TECNAI G2 TEM.
Results
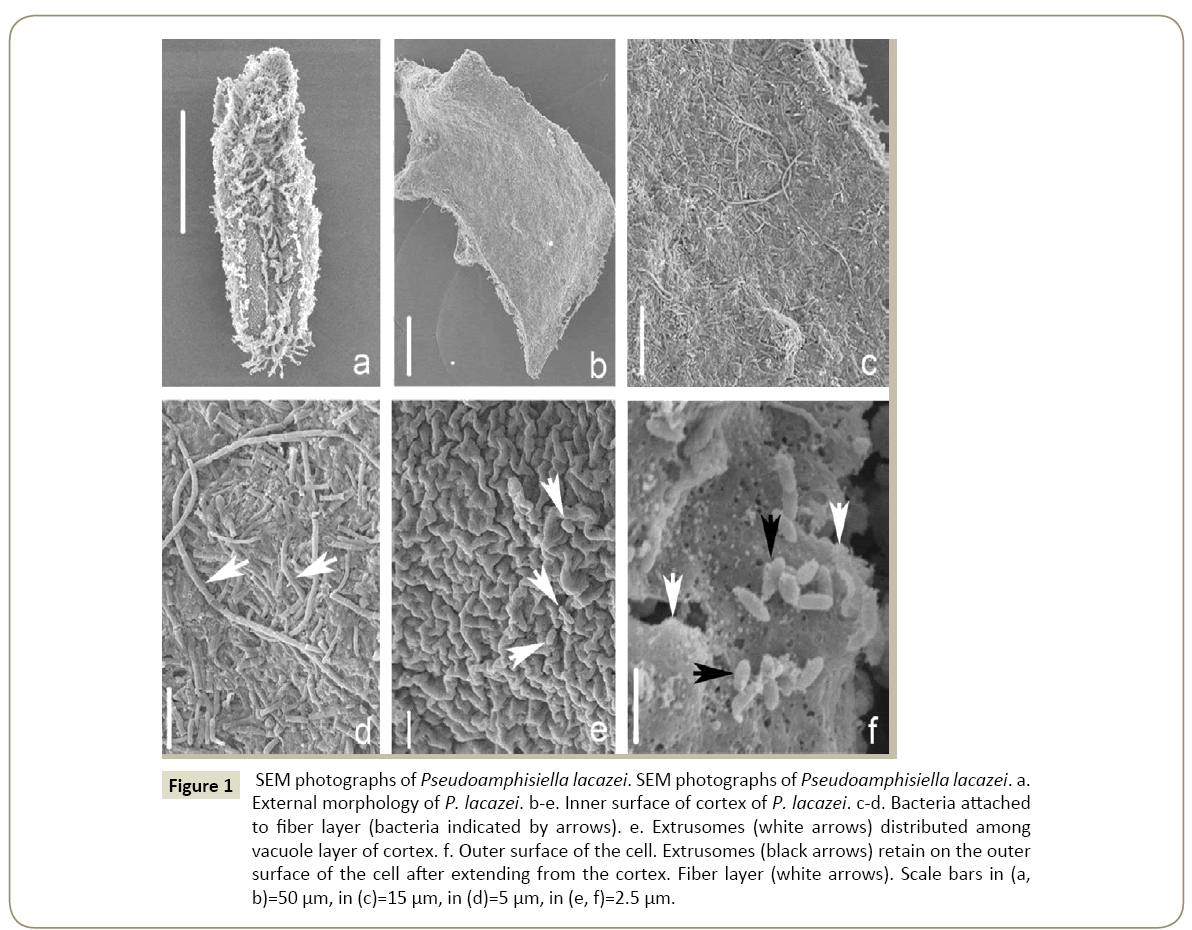
SEM ultrastructures of Pseudoamphisiella lacazei (Figure 1)
The Conventional SEM method can only illustrate the external morphology of samples (Figure 1a). But a new method allows us to peel off a piece of cell membrane to observe the inner surface of the cortex [11] (Figure 1b). By using this new method, the following new characteristics of P. lacazei are revealed by the current study: (1) The cortex is conspicuous and contains two parts: an inner fiber layer (Figures 1c, 1d and 1f) and an external vacuole membrane (Figure 1e). (2) Numerous oval extrusomes distributed among the vacuoles (Figure 1e) and retained on the outer surface of the cell after extending from the cortex (Figure 1f). (3) High numbers of bacteria are attached closely to the fiber layer. Some of them even extend into the gap of the fiber layer and are intertwined with these fibers (Figures 1c and 1d).
Figure 1: SEM photographs of Pseudoamphisiella lacazei. SEM photographs of Pseudoamphisiella lacazei. a. External morphology of P. lacazei. b-e. Inner surface of cortex of P. lacazei. c-d. Bacteria attached to fiber layer (bacteria indicated by arrows). e. Extrusomes (white arrows) distributed among vacuole layer of cortex. f. Outer surface of the cell. Extrusomes (black arrows) retain on the outer surface of the cell after extending from the cortex. Fiber layer (white arrows). Scale bars in (a, b)=50 µm, in (c)=15 µm, in (d)=5 µm, in (e, f)=2.5 µm.
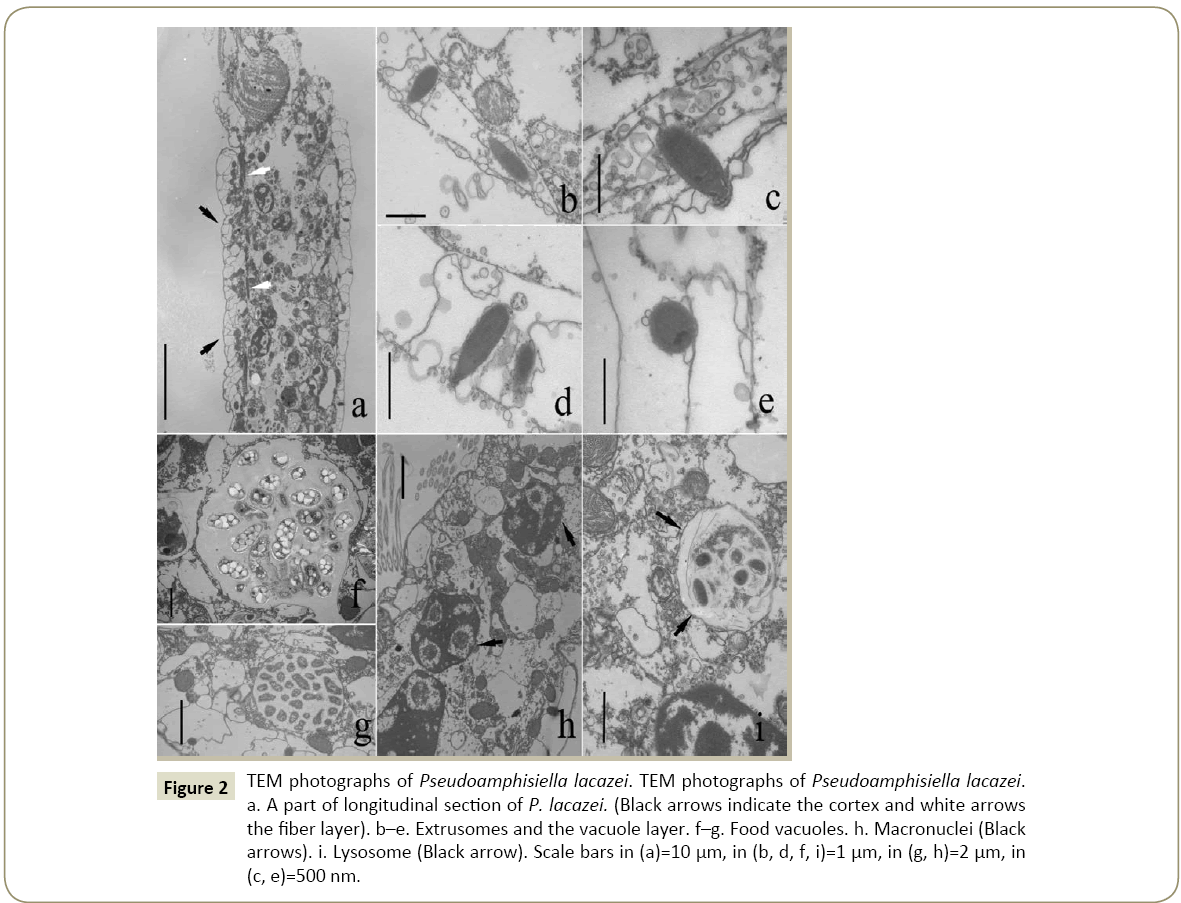
TEM ultrastructures of Pseudoamphisiella lacazei (Figure 2)
TEM observation showed that the vacuole membrane consists of two parts: a smooth membrane outside and an intensive vacuole layer beneath it (Figures 2a and 2c). The fiber layer, which appears to be only one cell-layer thick, covers the inner surface of these vacuoles (Figure 2a).
Figure 2: TEM photographs of Pseudoamphisiella lacazei. TEM photographs of Pseudoamphisiella lacazei. a. A part of longitudinal section of P. lacazei. (Black arrows indicate the cortex and white arrows the fiber layer). b–e. Extrusomes and the vacuole layer. f–g. Food vacuoles. h. Macronuclei (Black arrows). i. Lysosome (Black arrow). Scale bars in (a)=10 µm, in (b, d, f, i)=1 µm, in (g, h)=2 µm, in (c, e)=500 nm.
The extrusomes are distributed among those vacuoles (Figures 2b-2e). Their shape under TEM is long-oval, with one end slim, the other end round (Figure 2d). The average size is about 1.17 μm × 0.41 μm. These extrusomes can be observed to have two parts, which we tentatively name “head” and “body”. The length ratio of head and body is about 1:5. The head is wrapped in a dark cap-shaped structure (Figure 2c). The body part looks like a sac filled with filamentous matters (Figures 2c and 2d). A cup-shaped extrusome was observed as it had ejected the filamentous matters and the head were lost or indented into the body (Figure 2e).
The cytoplasm is not shown very dense. There were a few macronuclei and many mitochondria distributed in it (Figure 2h), but most of the space of cytoplasm was filled with various vacuoles, including different food vacuoles (Figures 2f and 2g) and some lysosomes (Figure 2i).
Discussion
Supplementary description of Pseudoamphisiella lacazei and its modified diagnosis
As the type species of Pseudoamphisiella, the morphological features of P. lacazei have been described on gross and microscopic levels [13] as follows: Body is usually 150-300 × 45- 100 um in vivo, elongate-shaped with both sides parallel and posterior end of cell broadly rounded; it’s left margin of anterior region usually ear-shaped and extrusomes bar-shaped. There are three frontal, two buccal and 8-11 caudal cirri. 20 rows transverse cirri that extends to anterior 1/2 of body, two ventral rows with 14-16 and 16-21 cirri respectively, 21-34 left and 20-31 right marginal cirri and 8-11 dorsal kineties. AZM composes of 39-56 membranelles. These extrusomes in P. lacazei are very similar in shape, structure and class to those in Urostyla grandis, the type species of Urostylida [10]. This result supports that the Family Pseudoamphisiellidae should be assigned to the order Urostylida [8,10].
The following new features are reported at the ultrastructural level in this paper, which should be added to the diagnosis of this species: Three structure layers constitute the cortex including an instant membrane, multiple vacuole structure layer and a fiber layer; only one style of extrusome is present, which should be mucocyst according to Hausmman’s description [14].
Morphological features and taxonomy of Pseudoamphisiella species
There are four species belonging to Pseudoamphisiella, P. alveolata, P. elongate, P. quadrinucleata and P. lacazei. We compiled their morphological features reported by different researchers since 1996 in Tables 1 and 2. All these features were obtained based on in vivo observation and protargol staining experiments, using a light microscope. Some morphological features observed under light microscope were used to identify species, such as: (1) The shape and size of extrusome. In P. lacazei, it is bar-like, 2 μm long compared to bar-like, 2–3 μm long in P. alveolata and P. quadrinucleata, rod-shaped in P. elongate. (2) The vacuole layer is absent in P. lacazei, but is present in the other three species. Obviously, these features are illustrated better by SEM and TEM which has been confirmed in the current study (Figures 1 and 2). However, our SEM and TEM observation confirmed that the vacuole layer does exist but is covered by a smooth membrane from outside and a fiber layer from inside (Figures 1e and 1f).
| Species | No. of AM | No. of BC | No. of FC | No. of cirri in ventral row 1 | No. of cirri in ventral row 2 | No. of LMC | No. of RMC | Pretransvers | No. of TC | No. of CC | No. of DK | No. of Mac. | No. of Mic. | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P. elongate | 39–49 | 2 | 3 | 13–17 | 15–21 | 26–45 | 28–36 | 2–3 | 16–21 | 6–7 | 6–7 | 25–44 | 6–17 | Li et al. |
| [7] | ||||||||||||||
| P. quadrinucleata | 49–75 | 2 | 3 | 11–22 | 14–20 | 15–31 | 14–20 | – | 14–23 | 13–24 | 12–17 | 4 | 2–6 | Shen et al. |
| [6] | ||||||||||||||
| P. alveolata | 37–43 | 2 | 3 | 11–15 | 10–15 | 15–30 | 12–16 | – | 13–17 | 11–16 | 9–12 | 2–3 | 1–6 | Hu and Suzuki |
| [4] | ||||||||||||||
| P. alveolata | 46–61 | 2 | 3 | 11–14 | 11–15 | 17–26 | 11–16 | – | 14–20 | 14–24 | 11–14 | 1–3 | – | Shao et al. |
| [8] | ||||||||||||||
| P. alveolata | 47–59 | 2 | 3 | 11–15 | 10–14 | 14–20 | 12–14 | – | 12–16 | 11–16 | 10–12 | 2 | 2–5 | Song and Warren |
| [5] | ||||||||||||||
| P. lacazei | 39–49 | 2 | 3 | 11–15 | 16–23 | 21–31 | 20–29 | – | 16–23 | 9–11 | 8–11 | 24–57 | 7–10 | Song et al. |
| [3] |
Note: Data based on protargol-stained specimens; –: Not mentioned; Mac: Macronuclei; Mic: Micronuclei; DK: Dorsal Kineties; TC: Transverse Cirri; CC: Caudal Cirri; RMC: Right Marginal Cirri; LMC: Left Marginal Cirri; FC: Frontal Cirri; BC: Buccal Cirri; AM: Adoral Membranelles
Table 1: Cirri features of Pseudoamphisiella species.
| Species | Extrusome | Alveolar layer(size, shape, µm) | Food vacuoles | Body size | References |
| (size, shape, µm) | (µm × µm) | ||||
| P. elongate | – | Present 3–4 | – | 160–240 × 30–40 | Li et al. [7] |
| Rod-shaped | irregular polygonal structure | ||||
| (Viewed dorsally) | |||||
| P. quadrinucleata | 2–3 | Present 5 | Difficult to recognize | 160–220 × 60–100 | Shenet al. [6] |
| Bar-like | – | ||||
| P. alveolata | 1.5–2 | Present 3 | Not observed | 120–200 × 50–70 | Hu and Suzuki [4] |
| Bar-like | Irregular polygonal structure | ||||
| (Viewed dorsally) | |||||
| P. alveolata | 2–3 | Present 3 | Difficult to recognize | 120–240 × 50–80 | Song and Warren [3] |
| Bar-like | Irregular polygonal structure | ||||
| (Viewed dorsally) | |||||
| P. lacazei | 2 | Contain small ciliates and flagellates | 120–300 × 40–80 | Songet al. [3] | |
| Absent | |||||
| Bar-like | |||||
| P. lacazei | – | Thin (inconspicuous) | Large and few | 200–300 × 70–100 | Song [3] |
| Short bar-shaped | – |
Note: Data based on in vivo observation; –: Not mentioned
Table 2: Other morphological features of Pseudoamphisiella species.
According to Berger [10] and Shao et al. [8], the Family Pseudoamphisiellidae should be treated as a peripheral group in the order Urostylida, based on its morphological and morphogenesis features. Recently some researchers found that Pseudoamphisiellidae may represent an ancestral form for both Discocephaline and Urostylida species [15] according to the small subunit ribosomal RNA sequence. We recommend the Pseudoamphisiellida should be placed in the order Urostylida.
In summary, ultrastructure observation can supplement and correct the micro-level observation and provide some evidence to the taxonomical argument.
Bacteria
A large number of bacteria were observed within P. lacazei cell. They are embedded within the fibers but not in food vacuoles. There can be two possible explanations for this phenomenon: (1) The bacteria were taken in as food but were not digested. This might have been due either to the accidental bursting of the phagosome membrane or to the inactivated or lost lysosome receptors [16]. (2) A daring speculation is that the bacteria might be the endosymbiont of P. lacazei. The phenomenon appears to support the hypothesis that the endosymbiont of the bacterium and the ciliate probably originates from phagocytosis.
Conclusion
1. According to the mucosyts of P. lacazei, the Family Pseudoamphisiellidae should be assigned to the order Urostylida.
2. According to the ultrastructure results of the cortex of P. lacazei, which consists of two layers of structures, the vacuole layer outer and a fiber layer from inner.
3. Large numbers of bacteria were attached to the fiber layer of P. lacazei, two possible explanations for this phenomenon; a daring speculation is that the bacterium might be the edosymbiont of P. lacazei.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (No. 31471950, 31101613, and No. 30970311) and Heilongjiang Postdoctoral Funding Project “Study on toxicology of intermediate-type filament system and other ultrastructures of ciliates”. Our thanks also due to Wang Xuedong and Cui Lin, Electron Microscopy Laboratory, Northeast Agriculture University, for technical assistance.
References
- Hemberger H (1982) Revision der Ordnung Hypotrichida STEIN (Ciliophora, Protozoa) an Hand von Protargolpräparaten und Morphogenesedarstellungen. Diss Math Nat Fak University of Bonn, Bonn, p: 182.
- Song WB (1996) Description of the marine ciliate Pseudoamphisiella lacazei(Maupas, 1883) nov. gen., nov. comb. (Protozoa, Ciliophora, Hypotrichida). Oceanologia Et Limnologia Sinica 27: 18-22.
- Song WB, Warren A, Hu XZ (1997) Morphology and morphogenesis of Pseudoamphisiella lacazei(Maupas, 1883) Song, 1996 with suggestion of establishment of a new family Pseudoamphisiellidae nov. fam. (Ciliophora, Hypotrichida). Arch Protistenk 147: 265-276.
- Hu XZ, Suzuki T (2006) Observation on a Japanese Population of Pseudoamphisiella alveolata (Kahl, 1932) Song et Warren, 2000 (Ciliophora: Hypotrichida): morphology and morphogenesis. Acta Protozool 45: 41-52.
- Song WB, Warren A (2000) Pseudoamphisiella alveolata (Kahl, 1932) nov. comb., a large marine hypotrichous ciliate from China (Protozoa, Ciliophora, Hypotrichida). Eur. J. Protistol 36: 451-457.
- Shen Z, Lin XF, Long HG, Miao M, Liu HB, et al. (2008). Morphology and SSU rDNA gene sequence of Pseudoamphisiella quadrinucleata n. sp. (Ciliophora, Urostylida) from the South China Sea. J Eukaryot Microbiol 55: 510-514.
- Li LQ, Song WB, Al-Rasheid KAS, Warren A, Li ZC, et al. (2010) Morphology and morphogenesis of a new marine hypotrichous ciliate (Protozoa, Ciliophora, Pseudoamphisiellidae), including a report on the small subunit rRNA gene sequence. Zool J Linn Soc 158: 231-243.
- Shao C, Song WB, Warren A, Al-Rasheid KAS, Yi ZZ, et al. (2006). Morphogenesis of the marine ciliate, Pseudoamphisiella alveolata (Kahl, 1932) Song & Warren, 2000 (Ciliophora, Stichotrichia, Urostylida) during binary fission. J Eukaryot Microbiol 53: 388-396.
- Miao M, Shao C, Chen XM, Song WB (2011). Evolution of discocephalid ciliates: Molecular, morphological and ontogenetic data support a sister group of discocephalids and pseudoamphisiellids (Protozoa, Ciliophora) with establishment of a new suborder Pseudoamphisiellina subord. n. Sci China Life Sci 54: 634-641.
- Berger H (2006). Monograph of the Urostyloidea (Ciliophora, Hypotricha). Monographiae Biologicae 85: 1-1304.
- Gao J, Zhou W, Qiao L, Chen Y, Qiu ZJ (2011). A new method of sample preparation for scanning electron microscope used to observe the structure under pellicle in ciliates. Chinese Journal of Cell Biology 33: 1004-1007.
- Gu FK, Chen L, Ni B, Zhang XM (2002). A comparative study on the electron microscopic enzyme-cytochemistry of Paramecium bursaria from light and dark cultures. Eur J Protistol 38: 267-278.
- Song WB, Warren A, Hu XZ (2009) Free-living Ciliates in the Bohai and Yellow Seas, China. Science Press, Beijing, pp: 395-396.
- Hausmann K, Hülsmann N, Radek R (2007)Protistology (Traslation of Song et al.). China Ocean University Press, QingDao, p: 203.
- Yi ZZ, Song WB, Warren A, Roberts D.Mcl, Al-Rasheid KAS, et al. (2008) A molecular phylogenetic investigation of Pseudoamphisiella and Parabirojimia (Protozoa, Ciliophora, Spirotrichea), two genera with ambiguous systematic positions. Eur J Protistol 44: 45-53.
- Cavalier-Smith T, Lee JJ (1985). Protozoa as hosts for endosymbioses and the conversion of symbionts into organelles. J Eukaryot Microbiol 32: 376-379.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences