Modern treatment of Head and Neck Melanoma: Case report and Review of literature
Megan E Ballard* and Giovana R Thomas
Department of Otolaryngology, University of Miami Hospital, Miami, Florida
- Corresponding Author:
- Megan E Ballard
Department of Otolaryngology
University of Miami Hospital, Miami, Florida
E-mail: megan.ballard@jhsmiami.org
Received date: July 24, 2017; Accepted date: August 28, 2017; Published date: September 07, 2017
Citation: Ballard ME, Thomas GR (2017) Modern Treatment of Head and Neck Melanoma: Case Report and Review of Literature. Res J Ear Nose Throat. Vol. 1 No. 1:2
Abstract
Surgical excision remains standard of care for cutaneous melanoma of the head and neck. Current guidelines suggest wide local excision of melanoma with recommended margins based on depth of disease. Once disease progresses to regional or distant spread however, additional adjuvant therapies should be offered in addition to surgical excision. Radiotherapy has been a mainstay of melanoma treatment and for several decades, immunotherapy with interferon was the mainstay of immune therapy. Targeted immunotherapy options are now available and are associated with favorable survival outcomes. These therapies are currently being studied in differently combinations and sequences to render improved results and possibilities. We present a case report of a young patient with locally advanced cutaneous melanoma of the scalp, who required multidisciplinary care for treatment. This case also highlights the intricate interplay necessary between various oncologic disciplines to achieve the highest possible survival outcomes for these patients.
Keywords
Melanoma; Scalp; Excision; Immunotherapy; Otolaryngology; Head; Neck
Introduction
Melanoma is an aggressive cutaneous cancer that has carried a high mortality rate for many years. It has been widely reported that the incidence of melanoma has continued to rise despite the decrease incidence of other cancers and the plateau of their respective mortality rates.
Melanoma often develops from existing nevi and has documented associated risks related to sun exposure, fairness of skin, and genetic mutations. It is postulated that the rise in melanoma is secondary to outdoor leisure time in industrialized areas [1]. Early in presentation, melanoma can be treated with a simple surgical excision; however, often at presentation, melanoma is an advanced systemic disease requiring multiple modalities for treatment.
There are five major types of melanoma, which are superficial spreading, nodular, acral lentiginous, lentigo maligna and mucosal [2]. Certain types of melanoma have been associated with different mutations which can influence the therapy that follows. Scalp melanoma is approximately 3-5% of all cutaneous melanomas and has been shown to have a high relapse rate and worse prognosis for survival when compared to other head and neck melanomas [3].
Because of the severity of melanoma and the need to treat the cancer both surgically and medically, the necessity of cross communication among fields is crucial for patient survival. It is important to fashion patient care in a multidisciplinary approach in order to provide the patient with their best odds at survival.
This article will discuss the progress in treatment of advanced stage melanoma exemplified in our 24 year old patient with stage IIIc nodular melanoma. His case highlights an outcome that, less than two decades ago, would have been an anomaly and is now becoming the norm.
Overview of standard treatment
Surgical excision remains standard of care for cutaneous melanoma of the head and neck. Current guidelines suggest Wide local excision (WLE) of melanoma with recommended margins based on depth of disease (refer to chart). Depending on the evidence of vertical growth and metastasis, melanoma may require further surgical intervention. If there is high risk or clinical evidence of nodal metastasis, a Sentinel lymph node biopsy (SLNB) or therapeutic neck dissection is recommended. Particularly in the case of melanoma of the head and scalp, patients may require parotidectomy in addition to the cervical lymph nodal dissection. The participation of a head and neck oncologic surgeon for head and neck melanoma currently involves resection, reconstruction and lymphadenectomy when indicated. If disease is limited locally, surgical excision with 0.5 cm margins suffices for in situ melanoma, 1 cm margins for thickness of 2 mm and 2 cm margins for tumors that are thicker than 2 mm [4] However, in the head area; it is difficult to achieve these margins without sacrifice of crucial function.
Once disease progresses to regional or distant spread, additional adjuvant therapies should be offered in addition to surgical excision. Radiotherapy has been a mainstay of melanoma treatment. For several decades, immunotherapy with interferon was the mainstay of immune therapy.
Thankfully, options are no longer limited to surgery and radiation, but include a continuously elongating list of targeted immunotherapy options. These therapies are currently being studied in differently combinations and sequences to render improved results and possibilities.
Case Report
A healthy 24 year old Caucasian man presented to his dermatologist concerned about a growing right parietal scalp nodule for 6 months. The melanoma occurred in the location of a nevus the patient had since nine years of age. At the time of presentation, his lesion measured 6 × 4 cm, approximately 2.5 cm in height and 1.5 cm in thickness. Additionally, vertical growth was noted with focal pagetoid spread. On exam, there was no palpable cervical lymphadenopathy.
The initial biopsy showed melanoma mitotic rate of >1 mm3 and no desmoplasia or ulceration. Our patient had no personal history of cancer of any kind and no documented family history. Of note, the patient did recall a paternal aunt deceased from possible lower leg skin cancer, but our patient was unclear of her diagnosis. He did not use tobacco, drink alcohol or use other illicit substances. In his skin history, he could not recall any incidences of burning or blistering with sun exposure.
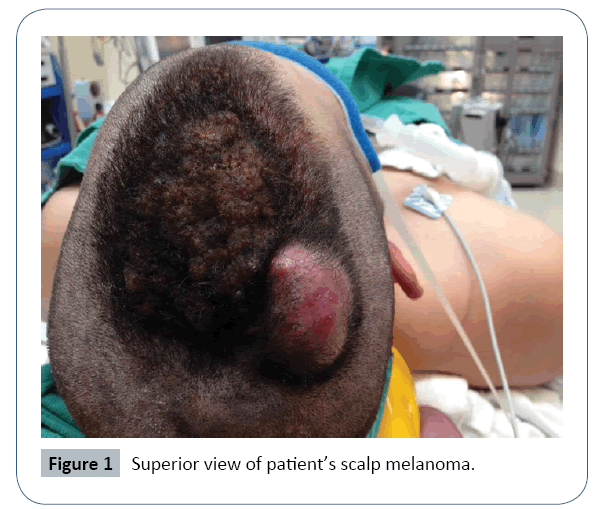
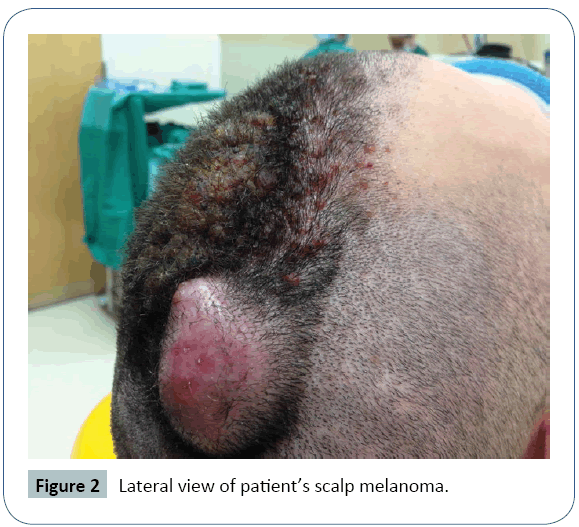
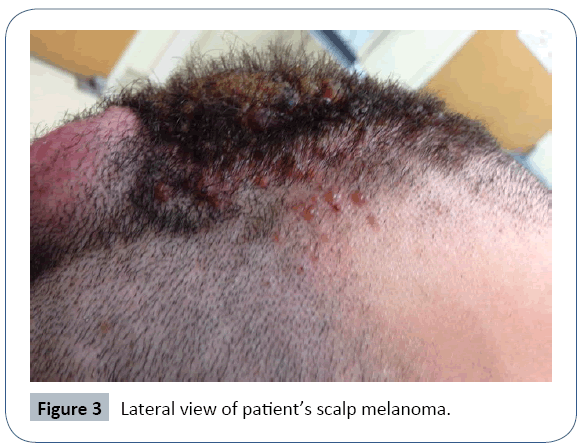
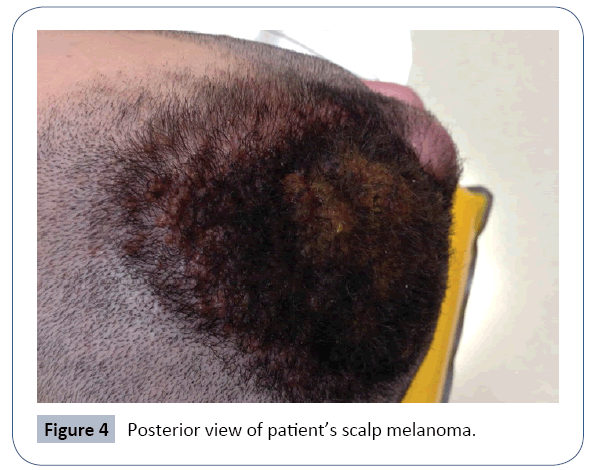
The first surgical excision for full evaluation occurred less than two weeks after his initial presentation given the rapid rate of growth. Within that time, the melanoma doubled in size to 12 × 8 cm and multiple new satellite lesions appeared. Refer to Figures 1-4 for appearance of the scalp melanoma prior to excision. Bilayer Integra grafting was placed over the large excision site until definitive reconstruction. Over the next 2 weeks, he was seen by Hematologic oncology and radiation oncology. He also obtained additional imaging for appropriate staging. Initial PET scan showed bilateral neck findings, particularly in the right level II B, as well as, multiple bilateral thyroid nodules. Less than one month after diagnosis, definitive resection with reconstruction was performed. The operation included a right parotidectomy, right neck dissection of levels II-V and revision scalp excision with additional 1 cm margins. The reconstruction of the defect was completed with a radial forearm free flap.
The pathology showed MART-1 positivity confirming melanocytic origin and close margins <1 mm despite obtaining two centimeter margins with several foci in dermal lymphatics; additionally, the melanoma was nodular type, had 16 mm thickness, ulceration, regional lymph node involvement and BRAF mutation positivity. Ultimately, the tumor was staged at pT4bN3, stage IIIc.
The initial adjuvant treatment plan was immunotherapy followed by radiotherapy. The patient was started on dabrafenib and trametinib two months after his initial presentation. Prompted by concerning imaging findings near his thyroid, our patient underwent a thyroid fine needle aspiration yielding results consistent with papillary thyroid cancer, Bethesda V-suspicious for malignancy. A thyroidectomy was planned after an appropriate response was obtained by immunotherapy. Two weeks after starting immunotherapy, rapidly growing satellite lesions were noted around the graft site. Over the next 5 months, the patient had repeated fevers, rashes and decreased cardiac ejection fraction causing the immunotherapy to be intermittently held or reduced on different occasions.
Throughout this time, the patient continued to see dermatology for routine surveillance every 3 months. The lesions on his scalp were monitored but no new lesions were found caudal to the neck. Four months after diagnosis and 2 months after immunotherapy started, steady progression of scalp disease was noted. The scheduled thyroidectomy was delayed and, the BRAF inhibitor, vemurafenib was started. The enlarging scalp lesions regressed on vemurafenib, but 3 new additional scalp lesions began growing while taking the new inhibitor. Nine months after initial presentation, immunotherapy was held and radiation started twice a week for a plan of 2 weeks. The decision was made to replace vemurafenib with ipilimumab, a monoclonal antibody, due to continued progression of lesions in the scalp and one on his ear.
Ten months after diagnosis, the patient had repeat WLE with skin graft to remove innumerable new cutaneous lesions, the larger lesions measuring 2-4 cm. The radiotherapy regimen was increased to 5 times a week and he completed a 21 day course of treatment shortly after his last excision. In total, our patient received 50 gy (20 fractions) to the scalp as post-operative adjuvant treatment for melanoma. The patient tolerated the treatments well with minor skin darkening, dryness and telangectasias which resolved over time.
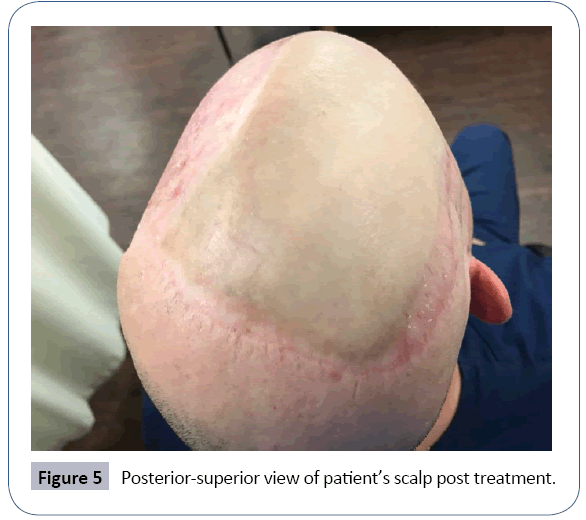
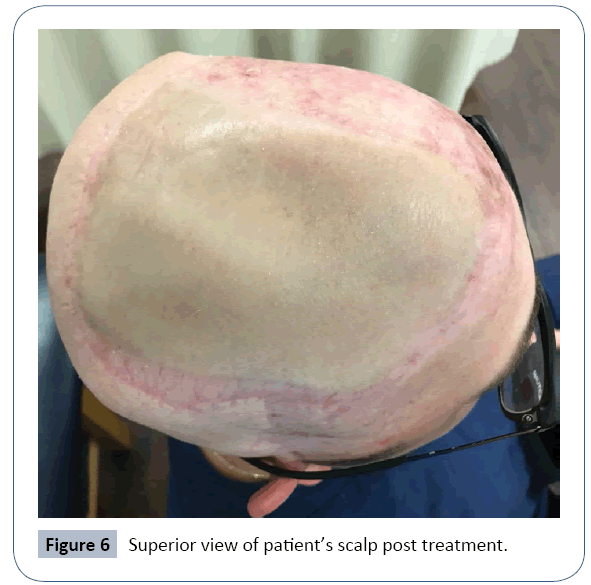
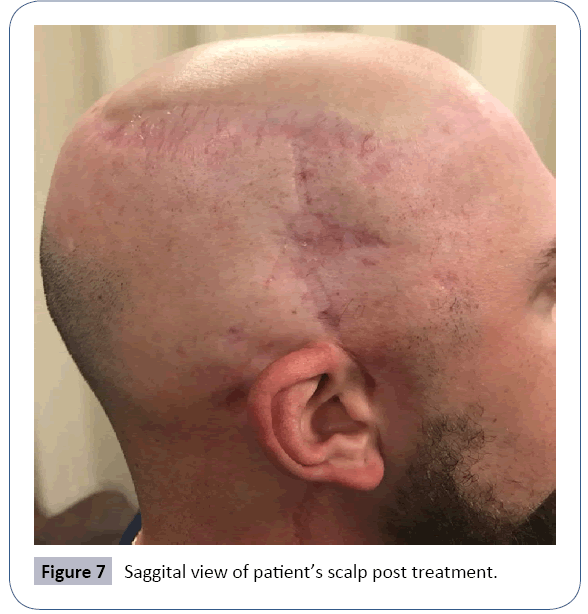
After radiation therapy completion, our patient had a total thyroidectomy with paratracheal node dissection. Pathology for the thyroid returned papillary thyroid carcinoma, follicular variant, T1N0, stage I. The patient recovered well after his thyroid surgery, which was his last operation. Figures 5-7 show the patient’s scalp 3 years after his last surgery and treatment. The patient continues to have scheduled surveillance exams by multiple specialties and scheduled imaging.
Discussion
Surgical treatment
Over the years, varying studies focus on the necessary extent of surgical dissection, specifically comparing SLNB versus the need for more invasive resection including, radical or modified radical neck dissection.
Over time, surgical management has evolved from a more radical approach to a more conservative approach. Despite surgical changes, conservative approaches, like modified neck dissection, continue to have risks, including vascular or nerve injury. For these reasons, SLNB and selective neck dissection has been studied as an alternative to detect disease and limit added morbidity. These alternatives are offered if regional evidence of metastasis is present [5-7].
The SLNB can serve a dual purpose. It is useful in detecting evidence of metastasis and also has been shown to be linked to poor prognosis when positive, thus it stands for useful general knowledge of the case and surgical planning [1] Some studies have noted that a more limited lymphadenectomy may yield the same regional control results as radical or modified radical neck dissection because the patients receive adjuvant therapy after dissection. In instances where reoccurrences arise in locations outside of a neck dissection’s contents, like our patient's auricular lesion, it can be argued that a less aggressive neck dissection could yield the same results [7].
The need for adjuvant therapies came due to the unfavorable outcomes of patients with advanced staged melanoma. Historically, surgery was offered for local control of melanoma, but once regional or distant metastasis was identified, survival outcome was considered poor [5]. Over the years, adjuvant therapies like chemotherapy and radiation therapy were added to the treatment plan of advanced melanomas.
In recent years, targeted immunotherapies have been developed to help control advanced disease. Particularly, ipilimumab has been identified as one of the main successes of immunotherapy for melanoma [8].
Adjuvant therapy
The addition of immunotherapy to existing treatment options for melanoma has revolutionized the treatment of these patients. The added benefits of radiotherapy, chemotherapy and immunotherapy are thought to be synergistic. The idea of the “abscopal effect” highlights one of the possible reasons multimodal treatment of melanomas are a success.
Barker and Postow reviewed clinical outcomes of radiation therapy and immunotherapy combination in treatment of melanoma and agree with this theory. Radiation therapy, when given locally, is thought to induce immune system recognition of the melanoma. With immunotherapy, as an immune system modulator, the identification response is heightened and the immune system becomes more effective at melanoma recognition, similar to administering a vaccine [9].
The immunotherapy medications that our patient received are the most widely studied in the literature. Immunotherapy’s popularity in research can be attributed to the discovery that when infiltrating T lymphocytes are noted in the tumor, better outcomes occurred. Research began in search of modulators to aid the immune system in recognizing the melanoma antigens. Interferon alpha was one of the first immunotherapies approved in 1995, but studies have not had consistent favorable responses to the therapy. The newer immunotherapy options have been shown to improve response rates and increase progression free survival and overall survival [10].
In a small case series, Hassel et al. demonstrated improvement in overall survival for patients with advanced melanoma when using vemurafenib and ipilimumab as combination therapy. In their study, patients with metastatic melanoma were followed after initially receiving vemurafenib as a monotherapy followed by ipilimumab. Vemurafenib was discontinued to prove ipilimumab’s role in disease control. Disease control was achieved for seven out of the 10 patients. This order of treatment and response is much like what we saw in our patient [11].
The mechanism of action of these immunotherapeutic agents has been studied. Immunotherapy is effective in targeting pathways of the melanoma tumor cell. The BRAF gene is responsible for cell growth, thus, when a BRAF mutation occurs; the cells bypass autoregulation and continue to multiply. The BRAF v600e mutated tumor can have uninhibited expansion by activating the mitogen-activated protein kinase pathway (MAPK), which occurs in approximately 40% of advanced melanoma cases [12,13]. Dabrafenib and vemurafenib are BRAF inhibitors.
Trametinib is a MEK inhibitor. It has been shown that while effective alone, the combination of BRAF and MEK inhibitors yields better results [14]. Another target is the cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) that is an inhibitory immune checkpoint receptor that inhibits T cells by binding the antigen-presenting cells (APC). This results in immune system downregulation. If an antibody blocks CTLA-4, T-cell proliferation and activation can occur and the body can fight against the tumor. Ipilimumab is a monoclonal antibody for CTLA-4 [15-18]. Immunotherapy research is also ongoing into T cell immunoglobulin and mucin-3, lymphocyte activation gene 3 (LAG3), killer inhibitory receptors (KIR) and indoleamine 2,3-dioxygenase (IDO) to aide in melanoma treatment [17]. Current ongoing studies are examining the combinations, dose and timing of these therapies together [19].
Despite these successes, it is important to note that immunotherapy responses are often limited by time because resistance develops in many patients. The developed resistance can be divided into 3 categories: a subsequent reactivating kinase, a new mutation in gene capable of circumventing the blocked kinase or, activation of a separate growth pathway. For this reason, the use of molecular targeted therapy exists as a double-edged sword providing rapid responses and regression, but short-lived victory and potentially fewer options due to the high rate of developed resistance that follows [17].
BRAF inhibitors have quick onset for results and show an impressive disease regression; however, in comparison to ipilimumab, their relative risk is higher and their period of disease free progression is limited [18]. Ipilimumab offers better longterm effects, but takes longer to build an appropriate immune response; therefore, ipilimumab will not be the best option for patients with rapidly progressive disease.
Conclusion
The advances in melanoma research are occurring rapidly and effects of new melanoma immunotherapy are surpassing previous expected survival rates.
Surgical resection remains one of the critical interventions after diagnosis, as our case presentation illuminates. However, since loco regionally advanced melanoma is a systemic disease, a multidisciplinary approach becomes more compelling. Medical (immunotherapy) and radiation oncologist should be part of the melanoma treatment group.
As research in this field advances, it is imperative for head and neck oncologic surgeons to keep abreast of developments in immunotherapy in order to guide and provide melanoma patients with the best personalized care available. A multidisciplinary team approach is essential to achieve favorable outcome in this disease.
Three years after our patient’s initial diagnosis, he has had no further treatment and he shows no evidence of local or distant disease.
References
- Kadakia S, Chan D, Mourad M, Ducic Y (2016) The prognostic value of age, sex and subsite in cutaneous head and neck melanoma: A clinical review of recent Literature. Iran J Cancer Prev 9: 5079.
- Unamuno Bustos BD, Murria Estal R, Pérez Simó G, de Juan Jimenez I (2017) Towards personalized medicine in melanoma: Implementation of a clinical next-generation sequencing panel. Sci Rep 7: 495.
- Tas F, Erturk K (2017) Scalp melanoma is associated with high mitotic rate and is a poor prognostic factor for recurrence and outcome. Melanoma Res 27: 387-390.
- Hayes AJ, Maynard L, Coombes G, Newton-Bishop J, Timmons M, et al. (2016) UK Melanoma Study Group, British Association of Plastic, Reconstructive and Aesthetic Surgeons, Scottish Cancer Therapy Network, Wide versus narrow excision margins for high-risk, primary cutaneous melanomas: Long-term follow-up of survival in a randomised trial. Lancet Oncol 17: 184-192.
- Raigani S, Cohen S, Boland GM (2017) The role of surgery for melanoma in an era of effective systemic therapy. Curr Oncol Rep 3: 17.
- Leiter U, Eigentler TK, Häfner HM, Krimmel M (2015) Sentinel lymph node dissection in head and neck melanoma has prognostic impact on disease-free and overall survival. Ann Surg Oncol 12: 4073-4080.
- Geltzeiler M, Monroe M, Givi B, Vetto J (2014) Regional control of head and neck melanoma with selective neck dissection. JAMA Otolaryngol Head Neck Surg 140: 1014-1018.
- Hodi FS, O'Day SJ, McDermott DF, Weber RW (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363: 711-723.
- Barker CA, Postow MA (2014) Combinations of radiation therapy and immunotherapy for melanoma: A review of clinical outcomes. Int J Radiat Oncol Biol Phys 88: 986-997.
- Van Zeijl MC, van den Eertwegh AJ, Haanen JB, Wouters MW (2017) (Neo)adjuvant systemic therapy for melanoma. Eur J Surg Oncol 3: 534-543.
- Hassel JC (2015) Vemurafenib and ipilimumab: A promising combination? Results of a case series. OncoImmunology 5: 4.
- Long GV, Grob JJ, Nathan P, Ribas A (2016) Factors predictive of response, disease progression and overall survival after dabrafenib and trametinib combination treatment: a pooled analysis if individual patient data from randomized trials. Lancet Oncol 17: 1743-1754.
- Schreuer M, Jansen Y, Planken S, Chevolet I (2017) Combination of dabrafenib plus trametinib for BRAF and MEK inhibitor pretreated patients with advanced BRAFV600-mutant melanoma: An open-label, single arm, dual-centre, phase 2 clinical trial. Lancet Oncol 18: 464-472.
- Simeone E (2017) Combination treatment of patients with BRAF-mutant melanoma: A new standard of care. BioDrugs 31: 51-61.
- Queirolo P, Spagnolo F (2017) BRAF plus MEK-targeted drugs: A new standard of treatment for BRAF-mutant advanced melanoma. Cancer Metastasis Rev 36: 35- 42.
- Vennepureddy A, Thumallapally N, Motilal Nehru V, Atallah JP, Terjanian T, et al. (2016) Novel drugs and combination therapies for the treatment of metastatic melanoma. J Clin Med Res 8: 63-75.
- Zhu Z, Liu W, Gotlieb V (2016) The rapidly evolving therapies for advanced melanoma--Towards immunotherapy, molecular targeted therapy and beyond. Crit Rev Oncol Hematol 99: 91-99.
- Luke JJ, Hodi FS (2013) Ipilimumab, vemurafenib, dabrafenib and trametinib: Synergistic competitors in the clinical management of BRAF mutant malignant melanoma. Oncologist 8: 717-725.
- Menzies AM, Long GV (2017) Optimum dosing of ipilimumab in melanoma: Too little, too late? Lancet Oncol 18: 558-559.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences