ISSN : 2348-9502
American Journal of Ethnomedicine
Assessment of the Cytotoxic Potential of Tecoma castanifolia (D. Don) Melch Flower Extract Against MCF-7 Cell Line
P.G. & Research Department of Advanced Zoology & Biotechnology, Loyola College, Nungambakkam, Chennai-600082, Tamil Nadu, India
Abstract
Objective: Bignoniaceae is a family having 100 genera of flowering plants. This family has many species with effective cytotoxic activity. One of its genuses, Tecoma contains 14 species and they are referred as trumpet trees. Tecoma species has various uses recorded in folk medicine and are being explored scientifically. Flowers of one of the species T.castanifolia is assessed for its potential.
Method: Fresh and healthy flowers were collected and were cold extracted with solvents viz., chloroform, ethyl acetate and methanol; filtered through Whatmann No.1 filter paper and evaporated to dryness. The extracts were subjected to MTT assay against MCF-7 cell line.
Result: The extraction yielded a gummy extract which were stored in a glass vial. The preliminary data shows that of the extracts tested the ethyl acetate extract has the highest cytotoxic activity in vitro against MCF-7 cell line. The 50% growth inhibition concentration of the ethyl acetate extract was obtained at 180μg/ml.
Concluison: The study shows that the ethyl acetate extract has fair cytotoxicity and can be tested with other cell lines. Further, studies pertaining to their composition of phytochemicals etc need to be done.
Keywords
Plant extract, Cytotoxicity, MCF-7, MTT assay, Tecoma.
Introduction
Worldwide, one in seven deaths is due to cancer. Cancer is one of the leading causes of deaths as it causes more deaths than AIDS, tuberculosis, and malaria combined. [1] Current estimates from the American Cancer Society and from the International Union Against Cancer indicate that 12 million cases of cancer were diagnosed last year, with 7 million deaths worldwide; these numbers are expected to double by 2030 (27 million cases with 17 million deaths.
Breast cancer is the most commonly diagnosed cancer among women in the vast majority (140 of 184) of countries worldwide, making it the only cancer that is common among women in all regions of the world (IARC Cancer Base). The treatments that are available slow down tumor progression and/or eradicate cancer cells. But this treatment does not always promise a complete recovery based on various factors such as time of cancer diagnosis, patient age, pathological condition of the tumor. This calls for the need for discovery of higher potent drug candidates. Majority of such research has been on the traditional medicines given in literature and on the folk medicines of various tribes.
Nature has been a source of medicinal agents for thousands of years and an impressive number of modern drugs have been isolated from natural sources [2]. India is usually referred as “Botanical garden of the world” due to its large diversity in flora. Many traditional medicines in use are derived from medicinal plants, minerals and organic matter [3]. Botanicals are less expensive and easily available because of their natural occurrence in abundance. [4]
Under the order Lamiales, the Bignoniaceae constitutes the family of flowering plant. This family contains more than 800 species spread throughout India. Many species of Bignoniaceae have some use,either commercially or ethnobotanically. Several species are known to be used as a folk medicine for diabetes, skin disorders, gastro and urinary disorders etc. [5]
Tecoma is a genus of 14 species of shrubs or small trees in the trumpet vine family. Tecoma sambucifolia H.B.K. alcoholic extract of flowers also demonstrated cytotoxicity against human hepatoma cell line [6]. One species T.stans have been known to posses anti diabetic activity and the compounds like tecomine and tecostanin have been isolated from it [7-11]. Based on such literatures, the cytotoxic potential of one of the members of Bignoniaceae, Tecoma castanifolia (D. Don) Melch was analysed in this study. The part of the plant chosen was the flowers.
MATERIALS AND METHODS
Materials
The solvents viz., chloroform, ethylacetate and methanol were of analytical grade and purchased from HiMedia Laboratories Ltd., Mumbai. 3-(4,5–dimethyl thiazol–2–yl)–5–diphenyl tetrazolium bromide (MTT), Fetal Bovine serum (FBS), Phosphate Buffered Saline (PBS), Modified Eagle’s Medium (MEM) and Trypsin were obtained from Sigma Aldrich Co, St Louis, USA. EDTA, Glucose and antibiotics from Hi-Media Laboratories Ltd., Mumbai. Dimethyl Sulfoxide (DMSO) and Propanol from E.Merck Ltd., Mumbai, India.
Collection and Extraction of Plant Material
The fresh flowers of the plant were collected in Chennai and the plant was authenticated from Botanical Survey of India, Coimbatore. The voucher number for the same is BSI/SRC/5/23/2014- 15/Tech./842. The healthy flowers were cold extracted with various solvents for a period of 7 days with continuous shaking. The solvents were changed thrice during the period and filtered through Whatmann No.1 filter paper. It was then rotary evaporated to obtain a gummy extract which were stored in a glass vial.
Cell lines and Culture medium
MCF-7 (Human breast carcinoma) cell line was procured from National Centre for Cell Sciences (NCCS), Pune, India. Stock cells were cultured in MEM supplemented with 10% inactivated Fetal Bovine Serum (FBS), penicillin (100 IU/ml), streptomycin (100 g/ml) and amphotericin B (5 g/ml) in an humidified atmosphere of 5% CO2 at 37C until confluent. The cells were dissociated with TPVG solution (0.2% trypsin, 0.02% EDTA, 0.05% glucose in PBS). The stock cultures were grown in 25 cm2 culture flasks and all experiments were carried out in 96 microtitre plates (Tarsons India Pvt. Ltd., Kolkata, India).
Preparation of Test Solutions
For Cytotoxicity studies, each weighed test drugs were separately dissolved in distilled DMSO and volume was made up with MEM supplemented with 2% inactivated FBS to obtain a stock solution of 1 mg/ml concentration and sterilized by filtration. Serial two fold dilutions were prepared from this for carrying out cytotoxic studies.
Determination of cell viability by MTT Assay
The monolayer cell culture was trypsinized and the cell count was adjusted to 1.0 x 105 cells/ml using MEM containing 10% FBS. To each well of the 96 well microtitre plate, 0.1 ml of the diluted cell suspension (approximately 10,000 cells) was added. After 24 h, when a partial monolayer was formed, the supernatant was flicked off, the monolayer was washed once with medium and 100 l of different test concentrations of test drugs were added on to the partial monolayer in microtitre plates. The plates were then incubated at 37oC for 3 days in 5% CO2 atmosphere, and microscopic examination was carried out and observations were noted every 24 h interval. After 72 h, the drug solutions in the wells were discarded and 50 l of MTT in PBS was added to each well. The plates were gently shaken and incubated for 3 h at 37o C in 5% CO2 atmosphere. The supernatant was removed and 100 l of propanol was added and the plates were gently shaken to solubilize the formed formazan. The absorbance was measured using a microplate reader at a wavelength of 540 nm [12,13]. The percentage growth inhibition was calculated using the following formula
% Growth Inhibition = 100 – 
RESULT
MTT Assay
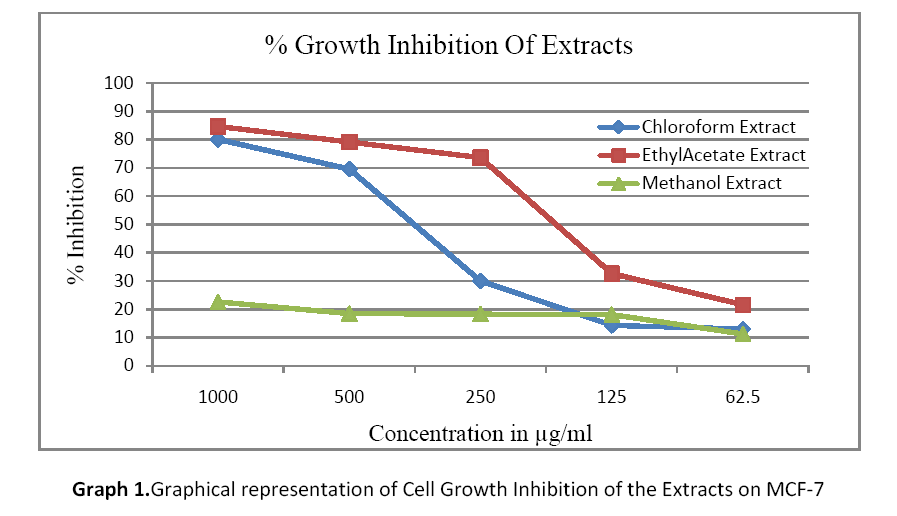
The concentration required to inhibit cell growth by 50% was assessed using MTT assay. The effectiveness of inhibition of the three extracts against MCF-7 cell line is graphically represented in Graph 1. MCF- 7 is a hormone dependant breast cancer cell line frequently used to test cytotoxic activity of various compounds and/or extracts. The GI50 values of chloroform, ethyl acetate and methanol extracts are 378.3μg/ml, 180.0μg/ml and >1000μg/ml respectively. Of the three extracts the ethyl acetate extract at 180μg/ml shows the most effective cytotoxicity at lowest concentration. The growth inhibitory concentration gives the preliminary support to further analyse the extract for cytotoxic activity.
Graph 1: Graphical representation of Cell Growth Inhibition of the Extracts on MCF-7
DISCUSSION
The genus of Tecoma has been traditionally used in various parts of the world to treat a variety of conditions and cancer is recorded to be one of it. The leaves of the plant is known to contain phenolic acid, beta-setosterol, triterpenoids etc., both phenol and flavonoids possessing antioxidant activity. Tecoma fabrisi and Tecoma impetignosa are used to treat some forms of cancer [14].
The three solvent extracts showed potent cell growth inhibitory activity. This may be due to their action on any one of the following- mitosis, biochemical processes etc. The biological activity shown by plants are mainly due to their secondary metabolites like alkaloids, phenolics, flavonoids, terpenoids, saponins etc. These are referred to as bioactive phyto compounds. Several phytochemical studies revealed that the extracts from many species of Bignoniaceae contained secondary metabolites such as saponins, tannins, flavonoids, quinones, alkaloids, anthralene derivatives, reducing sugars, glycosides, carbohydrates, querletin, kaempferol, âsitosterol, terpenes, steroids, coumarins etc., and their derivatives [15].
Out of the three crude flower leaf extracts tested here the ethyl acetate extract showed the effectiveness for further studies as an anti cancer agent. Its efficacy can be due to the fact of synergistic effects of various compounds present in the crude extract. The plants belonging to the family Bignoniaceae are one of the important sources of new bioactive compounds and as such as several new chemical entities have been isolated and tested for their efficacy as drugs [15].
CONCLUSION
The study was conducted to assess the possibility of this flower extract to have a potent cytotoxic potential. It is clear from the preliminary study that the extracts possess cytotoxic activity against MCF-7 cell line, the most cytotoxic being the ethyl acetate extract. This extract need to be further investigated for its cytotoxic potential and the compound(s) present in the extract should be analysed, which are responsible for the activity. The potential of the extracts against other types of cancer should also be analysed.
CONFLICT OF INTEREST
No conflict of interest has been raised by the authors. The project was self funded by the first author.
ACKNOWLEDGEMENT
I would like to thank my Supervisor and colleagues for their continuing support.
REFERENCES
- American Cancer Society. Global Cancer Facts & Figures 3rd Edition,2015.
- Dyamavvanahalli Lakshmanachar Shrisha et al., Bioprospecting of selected medicinal plants for antibacterial activity against some pathogenic bacteria; J. Med. Plant. Res. Vol. 5(17), pp. 4087-4093, 9 September, 2011.
- Grover, J.K., Yadav, S., and Vats, V.: Medicinal plants of India with antidiabetic potential. J. Ethnopharmacol., 81, 81–100, 2002.
- E.U Opara and R.C Wokocha., Efficacy of some plant extracts on in vitro and in vivo control of Xanthomonas campestris Pv. Vesicatoria., Agric.J., 3(3) : 163-170, 2008.
- Mohammed Rahmatullah et al., An Ethnomedicinal, Pharmacological and Phytochemical Review of Some Bignoniaceae Family Plants and a Description of Bignoniaceae Plants in Folk Medicinal Uses in Bangladesh, Adv. in Nat. Appl. Sci., 4(3): 236-253, 2010.
- L.F. Alguacil et al., Tecoma sambucifolia : anti-inflammatory and antinociceptive activities, and ‘in vitro’ toxicity of extracts of the ‘huarumo’ of Peruvian Incas., Journal of Ethnopharmacology 70 (2000) 227-233.
- S Raju et. al. Tecoma stans (L.) Juss. ex Kunth (Bignoniaceae): Ethnobotany, Phytochemistry and Pharmacology, JPBMS, 2011, 8 (07).
- Sing V et. al., Pharmacological and Phytochemical Findings of Tecoma Stans- A Review, JAPHR, 2011, Volume1, Issue 3.
- E. HERNANDEZ-GALICIA et al.,Studies on Hypoglycemic Activity of Mexican Medicinal Plants, Proc. West. Pharmacol. Soc. 45: 118-124 (2002).
- A.J. Alonso-Castro et al., The antidiabetic plants Tecoma stans (L.) Juss. ex Kunth (Bignoniaceae) and Teucrium cubense Jacq (Lamiaceae) induce the incorporation of glucose in insulin-sensitive and insulin- resistant murine and human adipocytes, Journal of Ethnopharmacology 127 (2010) 1–6.
- Thirumal et al., invitro anticancer activity of Tecoma stans (L.) Ethanolic leaf extract on human breast cancer cell line (MCF-7), IJPCBS 2012, 2(4), 488-493.
- Pavan Kumar Bellamakondi et al., In vitro Cytotoxicity of caralluma species by MTT and Trypan blue dye exclusion, Asian J Pharm Clin Res, Vol 7, Issue 2, 2014, 17-19.
- Patel Janki B et al., Anticancer and Cytotoxic Potential of Triticum aestivum Extract on Hela Cell Line, IRJP 2013, 4 (1).
- Swarna Kandakatla., K.N.V Rao., David Banji., Ethno- Medicinal Review on “Pachagotla” (Tecoma stans – (I.)Juss. Ex kunth), Herbal Tech Industry, November 2010.
- Shuvasish Choudhury et al., Phytochemistry of the Family Bignoniaceae- A review Assam University Journal of Science & Technology: Biological and Environmental Sciences, Vol. 7 Number I; 145-150, 2011.
- Abdel-Monem Ateya et al., Biological Activity of the Egyptian Medicinal Plants: Part 4 Cytotoxicity of 50 Egyptian Plants and Spices Against Hepatocellular Carcinoma., American Journal of Ethnomedicine, 2014, Vol. 1, No. 1, 056- 063
- Hayder B. Sahib et al., The Anti- proliferative Activity of Vitex agnus-castus Leaves Methanol Extract against Breast and Prostate Cancer Cell Line., AJPCT[3][02][2015] 159-166.
- Tahmasbpour et al., In vitro Antimicrobial and Cytotoxicity Assays of Satureja bakhtiarica and Zataria multiflora Essential Oils., AJPCT[3][6][2015] 502-511.
- Solomon Jeeva et al., Phytochemical evaluation of Bignoniaceae flowers., J. Chem. Pharm. Res., 2013, 5(4):106-111.
- S.Kameshwaran et al.,In vitro and in vivo anticancer activity of methanolic extract of Tecoma stans flowers., IRJP 2012, 3(3).
- SUDIPTA SAHA et al., ANTIPRO- LIFERATIVE EFFECT OF FLOWER EXTRACTS OF SPILANTHES PANI- CULATA ON HEPATIC CARCINOMA CELLS., Int J Pharm Pharm Sci, Vol 7, Issue 1, 130-134.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences