Total and Cardiovascular Mortality Risk Associated with Allergic Rhinitis and Allergic Asthma: A Retrospective Cohort Study
Gandhi S1*, Faramawi MF2, Aryal S3, Evans E4 and Felini M5
1Department of Medical Lab Sciences and Public Health, College of Science and Technology, Tarleton State University, Stephenville, TX, USA
2Department of Epidemiology, College of Public Health, University of Arkansas for Medical Sciences, Little Rock, AR, USA
3Department Epidemiology and Biostatistics, University of North Texas Health Science Center, Fort Worth, TX, USA
4Department of Math, School of Natural Science and Math, Dallas Baptist University, Dallas, TX, USA
5Department of Obstetrics and Gynecology, Patient Care Center, University of North Texas Health Science Center, Fort Worth, TX, USA
- *Corresponding Author:
- Gandhi S
Department of Medical Lab Sciences and Public Health, College of Health Sciences and Human Services
Tarleton State University, Davis Hall #103, 1333 West Washington, 76402, Stephenville, TX, USA
Tel: 254-968-0578
E-mail: gandhi@tarleton.edu
Received date: October 30, 2016; Accepted date: December 05, 2016; Published date: December 12, 2016
Citation: Gandhi S, Faramawi MF, Aryal S, et al. Total and Cardiovascular Mortality Risk Associated with Allergic Rhinitis and Allergic Asthma: A Retrospective Cohort Study. J Clin Med Ther. 2016, 1:1.
Abstract
Title: Total and Cardiovascular Mortality Risk Associated with Allergic Rhinitis and Allergic Asthma: A Retrospective Cohort Study
Background: Asthma and allergic rhinitis are two major respiratory conditions of public health concern worldwide. These conditions are also identified as risk factors for atherosclerosis, a contributor of cardiovascular outcomes. Our objectives were to investigate the independent and concomitant role of allergic rhinitis and asthma on cardiovascular and all-cause mortality.
Methods and findings: The study participants consisted of a probability sample of US civilians from the Third National Health and Nutrition Examination Survey; vital status was ascertained through the National Death Index. Men and women ≥40 years from all racial/ethnic backgrounds were included in the study. For all-cause mortality (n=9,450) and CVD-specific mortality (n=9,410), the baseline characteristics of participants were similar except for age, education, and smoking status. For both the outcomes, hazard ratios were estimated using Cox regression models.
Risk for all-cause mortality increased for those with histories of allergic rhinitis [HR: 1.08 (95%: 0.91-1.29)], and asthma [HR: 1.36 (95%: 1.00-1.89)] whereas risk for CVD-specific mortality showed null association for those with histories of allergic rhinitis [HR: 1.00 (95%: 0.75-1.31)], with elevated risk for those with asthma [HR: 1.32 (95%: 0.85-2.06)]. The combined effect of allergic rhinitis and asthma showed increased risk for all-cause mortality [HR: 1.23 (95%: 0.91-1.29)] and CVD-specific mortality [HR: 1.66 (95%: 0.85-3.25)].
Conclusion: Our study provides preliminary evidence of the independent and concurrent association between allergic rhinitis and asthma and all-cause and CVD-specific mortality.
Keywords
Asthma; Allergic rhinitis; Bronchitis; Immunoglobulins
Introduction
Asthma and allergic rhinitis are the two most common respiratory conditions that cause significant morbidity and mortality in the United States. Asthma is a lower respiratory disease that affects approximately 23 million Americans [1], whereas allergic rhinitis is an upper respiratory condition that affects up to 60 million Americans (10-30%) [2]. These two respiratory conditions can affect the overall quality of life, cause loss of productivity, and may even lead to premature mortality [2-5]. Allergic rhinitis and asthma differ in terms of anatomical location and symptoms. However, these two conditions demonstrate similar inflammatory responses where similar effector cells are found in the upper and lower respiratory airways [6]. Some studies have highlighted the link between allergic rhinitis and asthma, with an increased prevalence and severity of asthma in the presence of allergic rhinitis and vice versa [7-9].
Inflammation appears to be a critical mechanism linking allergic rhinitis and asthma with many chronic conditions. This possible link is demonstrated by several studies where atopic patients are at increased risk for diabetes, and specific cancers such as prostate, bladder cancer, and lymphoma [10-12].
Increased susceptibility of Cardiovascular Disease (CVD) among atopic patients has been demonstrated by some studies where inflammation is suspected to play a key role in the atherosclerotic process [10-14]. For instance, histamine released by the mast cells during atopy could increase vascular permeability permitting the influx of lipoproteins and other chemicals involved in the formation of atherosclerotic plaques [13,14], and other products of mast cells such as cytokines and growth factors could increase endothelium injury and inflammatory response [13]. These observations lead us believe that if there is an association between atopic conditions with other diseases via inflammation, it may be more likely be demonstrable in CVD compared to the other diseases. Taken collectively, a combined history of having asthma and allergic rhinitis may carry more risk of CVD than having a history of only one condition due cumulative inflammatory load.
Thus far, there are no studies that have explored the risk of CVD-specific mortality resulting from a combined history of asthma and allergic rhinitis. Hence, the objectives of our study were to investigate the independent and concomitant role of allergic rhinitis and asthma on cardiovascular and all-cause mortality. This study utilized the Third National Health and Nutrition Examination Survey (NHANES III) and NHANES III-linked mortality files to investigate whether individuals with combined histories of asthma and allergic rhinitis elevate the risk for CVD-specific mortality. To our knowledge, this is the first retrospective cohort study to examine the effects that a combined history of asthma and allergic rhinitis has on allcause mortality or CVD-specific mortality using a representative sample of the U.S. population.
Methods
Study population
The study population included participants of NHANES III, from whom data was originally collected from 1988 up to 1994, and for whom mortality data was obtained until December 31, 2006 [15]. We utilized NHANES III participants that were ≥40 years of age at the time of home interview and whose mortality was ascertained based on the probabilistic match between the NHANES III records, and the National Death Index (NDI) records were eligible for this retrospective cohort study. Study eligibility was restricted to NHANES III participants who reported no prior diagnosis of chronic bronchitis or emphysema at baseline (time of the interview). Study eligibility was then restricted to participants with complete data on allergic rhinitis and asthma at baseline as well as completes data on variables of interest at baseline.
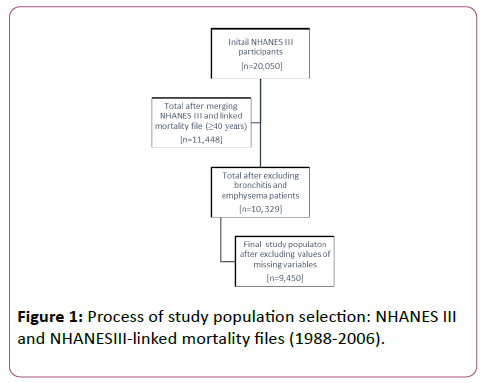
The initial NHANESIII file contained records for 20,050 participants and the NHANES III-Linked Mortality File contained records for 20,024 participants. After merging these two files and excluding participants that were <40 years (n=8,576), those with bronchitis and emphysema (n=1,119), and those with missing values of the pertinent variables (n=879), 9,450 participants remained in the dataset. Figure 1 shows the process of final study population selection after applying the inclusion/exclusion criteria.
Exposure assessment
Data for the histories of allergic rhinitis and asthma were obtained from the Household Adult File of NHANES III. Participants that answered “yes” at the time of the survey to the questions pertaining to having had a prior diagnosis of allergic rhinitis by a physician, or a prior diagnosis of asthma by a physician were categorized as exposed to the respective condition. Additionally, participants that answered “yes” to both questions were categorized as exposed to allergic rhinitis and asthma. Participants that reported having never been diagnosed with either condition were assigned to the referent category.
Covariates assessed for confounding included age, sex, race, alcohol history (yes/no), smoking history (ever/never), education (HS, college), physical activity (moderate, light), and BMI (normal, overweight, obese). All were extracted from the Household Adult File of NHANES III.
Outcome assessment
For all-cause mortality, deaths from all causes (ICD-10 codes) listed in the NHANES III linked mortality file that occurred among NHANES III participants from 1988 until were included in the analysis in December 31, 2006. For CVD-specific mortality, deaths from CVD-specific causes (ICD-10 codes: I10-I13, I20-I28, I34-I38, I42-I51, I60-I99) listed in the NHANES III Linked Mortality File that occurred among NHANES III participants from 1988 until a period after December 31st, 2006 were included in the analysis.
Statistical analysis
Univariate and bivariate analyses were performed to compare the frequency and percent distributions of was used to test the null hypothesis of independence for categorical variables (p-value of <0.05, two-tailed). The covariates assessed for confounding included age, sex, race, BMI, smoking, alcohol, education, and physical activity.
Cox proportional hazards regression models were fitted using SAS PROC SURVEYPHREG procedure (SAS 9.2, SAS Institute, Cary, NC). The follow-up (in person-years) was calculated from baseline (day of the interview) until CVD-related deaths, or deaths from other causes, or termination of the study. These models with censored observations and time at risk (years) for all-cause mortality and CVD-specific mortality were utilized to calculate HRs, along with the corresponding 95% CI. For all-cause mortality, censored individuals were those that did not die during the observation period, and those that were lost to follow up by the end of 2006. For CVDspecific mortality, censored individuals were those that did not die from CVD-related complications, and those that were lost to follow up by the end of 2006. The proportional hazards assumption was checked by using the interaction terms (variable x time); no violations were detected.
Results
Table 1 demonstrates the baseline characteristics of NHANES III participants with allergic rhinitis and asthma. The study participants with these two conditions were similar in their underlying characteristics except for age, smoking, and education (p<0.05).
| Allergic Rhinitis (n=577) | Allergic Asthma (n=185) | Allergic Rhinitis and Allergic Asthma (n=89) | No Allergic Rhinitis and No Allergic Asthma (n=8599) | P-value | |
|---|---|---|---|---|---|
| Demographic Factors Total=9450 | |||||
| Age categories | |||||
| Age 40-49 | 184 (9.15) | 45 (2.35) | 35 (1.60) | 2171 (86.89) | <0.001* |
| Age 50-59 | 122 (8.00) | 33 (1.48) | 20 (1.11) | 1579 (89.41) | |
| Age 60-69 | 106 (6.21) | 48 (2.05) | 18 (0.93) | 1956 (90.81) | |
| Age 70-79 | 95 (6.05) | 32 (1.49) | 11 (0.71) | 1608 (91.74) | |
| Age 80-90 | 70 (5.49) | 27 (1.71) | 5 (0.36) | 1285 (92.43) | |
| Gender | |||||
| Male | 239 (6.59) | 73 (1.84) | 35 (0.88) | 4245 (90.69) | 0.07 |
| Female | 338 (8.57) | 112 (2.01) | 54 (1.39) | 4354 (88.02) | |
| Race/ethnicity | |||||
| Non-Hispanic white | 341 (7.87) | 90 (1.79) | 51 (1.13) | 4507 (89.21) | 0.62 |
| Non-Hispanic black | 148 (6.80) | 56 (2.31) | 28 (1.46) | 2041 (89.43) | |
| Mexican-American | 68 (5.31) | 29 (1.62) | 9 (0.83) | 1753 (92.24) | |
| Other race/ethnicity | 20 (7.06) | 10 (3.34) | 1 (1.04) | 298 (88.56) | |
| Smoking | |||||
| Current smoker | 96 (5.42) | 40 (2.00) | 18 (0.89) | 1856 (91.69) | 0.03* |
| Former smoker | 201 (8.28) | 72 (2.28) | 28 (0.77) | 2838 (88.67) | |
| Non-smokers | 280 (8.27) | 73 (1.60) | 43 (1.59) | 3905 (88.53) | |
| Alcohol | |||||
| Non-drinkers | 321 (7.35) | 116 (2.13) | 48 (1.24) | 5072 (89.28) | 0.74 |
| Drinkers | 256 (7.93) | 69 (1.72) | 41 (1.05) | 3527 (89.29) | |
| Education | |||||
| Less than high school | 177 (4.61) | 91 (2.21) | 25 (0.54) | 3975 (92.64) | <0.001* |
| High school-GED | 174 (7.48) | 52 (2.01) | 25 (1.02) | 2428 (89.50) | |
| More than high school | 226 (9.96) | 42 (1.66) | 39 (1.70) | 2196 (86.68) | |
| Physical activity | |||||
| Active | 171 (8.19) | 43 (1.36) | 29 (1.33) | 2509 (89.12) | 0.22 |
| Insufficiently active | 287 (7.57) | 77 (1.91) | 42 (1.06) | 3931 (89.46) | |
| Inactive | 119 (6.82) | 65 (3.02) | 18 (1.10) | 2159 (89.06) | |
| Body Mass Index | |||||
| Normal (<25) | 240 (8.09) | 66 (1.61) | 29 (1.11) | 3463 (89.19) | 0.42 |
| Overweight (25-30) | 212 (7.22) | 66 (1.84) | 34 (1.00) | 3345 (89.94) | |
| Obese (>30) | 125 (7.46) | 53 (2.79) | 26 (1.53) | 1791 (88.22) | |
| % and mean (SE) are weighted (the percentages may not add to 100% due to rounding), frequencies are not weighted; Chi-square test for categorical variables: Rao-Scott; *Statistically significant | |||||
Table 1: Characteristics of the study participants by allergic status at baseline for all-cause mortality: NAHANES III and NHANESIIIlinked mortality file (1988-2006).
Table 2 details the unadjusted and adjusted hazard ratios with 95% confidence intervals for allergic conditions and all-cause and CVD-specific mortality. After adjusting for age, education, and smoking, people having allergic rhinitis [HR: 1.08 (95% CI: 0.91, 1.29)], allergic asthma [HR: 1.36 (95% CI: 1.00, 1.89)], and both allergic rhinitis and allergic asthma [HR: 1.23 (95% CI: 0.78, 1.93)] all demonstrated increased risk for all-cause mortality. Only the risk estimate associated with allergic asthma was marginally significant.
| All-cause mortality | CVD-specific mortality | |||
|---|---|---|---|---|
| Unadjusted HR (95%CI) | Adjusted HR (95%CI) | Unadjusted HR (95%CI) | Adjusted HR (95%CI) | |
| Self-reported allergic conditions | ||||
| Allergic rhinitis | 0.83 (0.67, 1.03) | 1.08 (0.91, 1.29) | 0.77 (0.56, 1.05) | 1.00 (0.75, 1.31) |
| Allergic asthma | 1.26 (0.85, 1.86) | 1.36 (1.00, 1.89)* | 1.21 (0.74, 1.98) | 1.32 (0.85, 2.06) |
| Allergic rhinitis and allergic asthma |
0.62 (0.38, 1.00) | 1.23 (0.78, 1.93) | 0.80 (0.42, 1.54) | 1.66 (0.85, 3.25) |
| No allergic rhinitis and allergic asthma |
1.00 | 1.00 | 1.00 | 1.00 |
| All-cause mortality: adjusted for age (continuous), education, and smoking; CVD-specific mortality: adjusted for age (continuous) and education. *Statistically significant. | ||||
Table 2: Hazard Ratios (HR) and 95% confidence intervals (CI) for all-cause mortality and CVD-specific mortality: NHANES III and NHANES III-linked mortality file (1988-2006).
The risk of CVD-specific mortality was null for having a history of allergic rhinitis [HR: 1.00 (95% CI: 0.75, 1.31)], but increased for those with a history of asthma [HR: 1.32 (95% CI: 0.85, 2.06)] when adjusted by age and educational status. The risk of CVD-specific mortality was higher yet for those who reported having had both conditions [HR: 1.66 (95% CI: 0.85, 3.25)]. Change in risk estimates were negligible for CVD-specific mortality upon further adjustments by common confounders noted in the literature and for the covariates that were significantly associated with CVD-specific mortality (results not shown).
Discussion
Although the majority of findings were not significant, data from this study support an increased risk of CVD-specific mortality due to combined histories of both allergic rhinitis and asthma. A follow-up study that used a cohort of 203,595 participants reported joint effects between asthma and a history of allergy, and three CVD-specific outcomes: CHD [HR: 1.39 (95% CI: 1.32, 1.45)], cerebrovascular disease [HR: 1.18 (95% CI: 1.11, 1.25)], and heart failure [HR: 1.80 (95% CI: 1.72, 1.89)] [16]. Although we could not calculate the risk estimate for heart failure due to low numbers, our sub-analysis of CVDspecific estimates replicated the findings of the study by Iribarren et al. [16]. We observed an elevation in risk for the combined effect of having allergic rhinitis and allergic asthma for cerebrovascular disease [3.75 (95% CI: 1.09, 13.00)], CHD [1.47 (95% CI: 0.35, 6.12)], and MI [2.08 (95% CI: 0.53, 8.19)]. This suggests that when different cardiovascular diseases are combined into one category, dilution of effect may occur, underestimating the total risk of CVD-specific mortality.
Another potential source of dilution of effect in previous studies has been in the broad categorization of allergic conditions. Matheson et al. [17] found the risk of stroke mortality was 50% higher among survey participants that reported previously diagnosed allergic rhinitis, asthma, or both the conditions (as one exposure) compared to the non-exposed category [HR: 1.52 (95% CI: 0.80, 2.92)] [17]. We estimated the risk of stoke mortality to be 1.16 (95% CI: 0.68, 1.98) from allergic rhinitis alone, 0.66 (95% CI: 0.26, 1.70) from asthma alone and 3.75 (95% CI: 1.09, 13.00) from a combined history of allergic rhinitis and asthma, which supports a dilution of effect when these categories were combined into one group. While the main interest of this study was in CVDspecific mortality, all-cause mortality was also examined to gain perspective in interpreting results for CVD deaths associated with allergies. Although it is possible the risk of allcause mortality could be driven mainly by CVD-related deaths, the proportion of deaths due to CVD in our data (47.8%) do not support this explanation (data not shown). Hence, we suspect that other causes of mortality associated with allergies (e.g., cancers and autoimmune diseases) may have influenced the risk estimates for all-cause mortality.
The results of this study should be interpreted cautiously considering its limitations. We did not have data regarding the severity and onset of allergic rhinitis and asthma, and were unable to determine if the duration of disease increased risk among those with persistent/severe forms compared to the ones with intermittent/mild forms with shorter duration. Internal validity of the study could have been threatened when the exclusion criteria was applied during study population selection. Participants that were excluded from the study could be much older from those who were included. However, the demographics of the study participants were similar to those who were excluded, making selection bias less likely due to subject participation.
Measurement error is also highly likely in this study, which could have influenced our study findings. Unlike asthma, allergic rhinitis is considered a mild disease and over 50% of people do not seek care [18-20]. Therefore, NHANES III participants may have severely underreported this condition, as they were not “physician diagnosed.” Similar to allergic rhinitis, many participants may have undiagnosed asthma as well. In a population-based study (n=1,149) conducted in Denmark among those with allergic rhinitis or asthma, of all the asthmatic cases, almost 50% of the participants had undiagnosed and untreated asthma [21].
Despite these limitations, there were a number of strengths associated with this study. This is the first longitudinal study to discern the combined effects of two common allergic conditions (allergic rhinitis and asthma) on CVD-specific mortality. We accounted for the key confounders that could obscure our study findings. Some of the previously conducted studies did not control for COPD, which could be a strong confounder for the explored relationships [17,22]. However, we have excluded participants having two major forms of COPD, chronic bronchitis and emphysema. Additionally, these studies controlled for factors that could have mediated the pathway (mediators), instead of acting as a confounder. For example, controlling for hypertension could have biased their results as there is evidence that allergic conditions can increase blood pressure [23,24].
Our results suggest that inflammation being the link between atopic diseases such as asthma and allergic rhinitis and CVD-specific mortality is a feasible hypothesis. This study provides new evidence and should be used to support future molecular studies measuring total inflammatory burden in the body contributed by these two allergic conditions. Additionally, findings uncovered from this study may aid health practitioners to closely monitor patients that have prior diagnoses of allergic rhinitis and asthma to reduce the burden of CVD-specific mortality. However, the need for public health interventions to decrease allergen exposures and premature mortality, assuming there is a causal relationship, cannot be recommended until absolute measures of association (e.g., PAR %) rather than relative measures of association (e.g., HR) determine public health significance of this study’s findings. Future prospective studies that incorporate data related to episodic events and duration of the disease, in order to accurately measure the long-term effects of these conditions, it will be important to further elucidate the joint effects of allergens on mortality.
Conclusion
The results of our study indicate that a combined history of asthma and allergic rhinitis may increase the risk of premature mortality from CVD, and from all-causes combined. Future molecular studies that utilize specific methods to ascertain allergic conditions (e.g., use of immunoglobulins), and rigorous population-based studies should focus on the long-term health effects of allergic rhinitis and asthma. If additional robust studies replicate our findings, public health efforts can be focused on prevention and treatment strategies that could reduce the burden of morbidity and mortality associated with allergic conditions.
Acknowledgment
This research study was not supported by any grant funding.
References
- National Heart Lung and Blood Institute (NHLBI)(2012) NHLBI Morbidity and Mortality Chart Book. NIH.
- Settipane RA,Schwindt C (2013) Chapter 15: Allergic rhinitis: Am J Rhinol Allergy 27: 52-55.
- Barnett SB,Nurmagambetov TA (2011) Costs of asthma in the United States: 2002-2007. J Allergy Clin Immunol 127: 145-152.
- Schatz M (2007) A survey of the burden of allergic rhinitis in the USA. Allergy 62: 9-16.
- Stempel DA, Woolf R (2002) The cost of treating allergic rhinitis. Curr Allergy Asthma Rep 2: 223-230.
- Hadley JA, Derebery MJ, Marple BF (2012) Comorbidities and allergic rhinitis: Not just a runny nose. J FamPract 61: S11-S15.
- Navarro A, Valero A, Julia B, Quirce S (2008) Coexistence of asthma and allergic rhinitis in adult patients attending allergy clinics: ONEAIR study. J Investig Allergol ClinImmunol 18: 233-238.
- Ryan MW (2008) Asthma and rhinitis: Comorbidities. Otolaryngologic Clinics of North America 41: 283-295.
- Valero A, Pereira, C, Loureiro C, Cocera C, Murio C, et al. (2009) Interrelationship between skin sensitization, rhinitis, and asthma in patients with allergic rhinitis: A study of spain and portugal. Journal of Investigational Allergology and Clinical Immunology: Official Organ of the International Association of Asthmology (INTERASMA) and SociedadLatinoamericana De Alergia E Inmunologia 19: 167-172.
- Juhn YJ (2012) Influence of asthma epidemiology on the risk for other diseases. Allergy Asthma Immunol Res 4: 122-131.
- Merrill RM, Isakson RT, Beck RE (2007) The association between allergies and cancer: What is currently known? Annals of Allergy, Asthma & Immunology: Official Publication of the Ann Allergy Asthma Immunol 99: 102-116.
- Yun HD, Knoebel E, Fenta Y, Gabriel SE, Leibson CL, et al. (2012) Asthma and proinflammatory conditions: A population-based retrospective matched cohort study. Mayo Clinic Proceedings. Mayo Clin 87: 953-960.
- Bergmann K, Sypniewska G (2011) Is there an association of allergy and cardiovascular disease? BiochemiaMedica: Casopis Hrvatskoga Drustva Medicinskih Biokemicara/HDMB 21: 210-218.
- Rozenberg I, Sluka SH, Rohrer L, Hofmann J, Becher B, et al. (2010) Histamine H1 receptor promotes atherosclerotic lesion formation by increasing vascular permeability for low-density lipoproteins. ArteriosclerThrombVascBiol 30: 923-930.
- National Center for Health Statistics (NCHS) (2009) Office of analysis and epidemiology, Third national health and nutrition examination survey (NHANES III) linked mortality file, Mortality follow-up through 2006: Matching methodology.
- Iribarren C, Tolstykh IV, Miller MK, Sobel E, Eisner MD (2012) Adult asthma and risk of coronary heart disease, cerebrovascular disease, and heart failure: A prospective study of 2 matched cohorts. Am J Epidem 176: 1014-1024.
- Matheson EM, Mainous III AG, Carnemolla MA (2009) The association between allergy skin testing, atopic respiratory conditions, and stroke mortality in middle-aged and elderly adults. Journal of the American Board of Family Medicine: JABFM 22: 604-609.
- Canonica GW, Bousquet J, Mullol J, Scadding GK, Virchow JC (2007) A survey of the burden of allergic rhinitis in Europe. Allergy 62: 17-25.
- Demoly P, Calderon MA, Casale T, Scadding G, Annesi-Maesano I, et al. (2012) Assessment of disease control in allergic rhinitis. ClinTransl Allergy 3: 7-7022-3-7.
- Valovirta E, Myrseth SE, Palkonen S (2008) The voice of the patients: Allergic rhinitis is not a trivial disease. CurrOpin Allergy ClinImmunol 8: 1-9.
- Nolte H, Nepper-Christensen S, Backer V (2006) Unawareness and undertreatment of asthma and allergic rhinitis in a general population Respir Med 100: 354-362.
- Knoflach M, Kiechl S, Mayr A, Willeit J, Poewe W, et al. (2015) Allergic rhinitis, asthma, and atherosclerosis in the bruneck and ARMY studies. Arch Intern Med 165: 2521-2526.
- Corbo GM, Forastiere F, Agabiti N, Baldacci S, Farchi S, et al. (2016) Rhinitis and snoring as risk factors for hypertension in post-menopausal women. Respir Med 100: 1368-1373.
- Kony S, Zureik M, Neukirch C, Leynaert B, Vervloet D, et al. (2003) Rhinitis is associated with increased systolic blood pressure in men: A population-based study. Am J Respir Crit Care Med 167: 538-543.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences