ISSN : 2634-7164
Journal of Medical Microbiology and Immunology Research
Lung Clearance of Aspergillus fumigatus Conidia Pre-opsonized with the Long Pentraxin PTX3
Mariachiara Minerva1*, Emanuele Marra1, Maria Lucrezia Pacello1, Daniela Stoppoloni1, Caterina Chiapparino1, Rita De Santis2, Giovanni Salvatori2, Flavia Novelli3 and Claudio Pioli3
1Takis srl, Via di Castel Romano, 100, 00128, Rome, Italy
2Alfasigma S.p.A. via Pontina km 30,400, 00040, Pomezia, Italy
3ENEA, Division of Health Protection Technologies, Via Anguillarese 301, 00123, Rome, Italy
- *Corresponding Author:
- Mariachiara Minerva
Takis srl, Via di Castel Romano
100, 00128, Rome, Italy
Tel: 0650576077
E-mail: minerva@takisbiotech.it
Received Date: Nov 06, 2017; Accepted Date: Nov 27, 2017; Published Date: Dec 05, 2017
Citation: Minerva M, Marra E, Pacello ML, Stoppoloni D, Chiapparino C, et al. (2017) Lung Clearance of Aspergillus fumigatus Conidia Preopsonized with the Long Pentraxin PTX3. J Med Microbiol Immunol Res 1:1.
Copyright: © 2017 Minerva M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
PTX3-preopsonized conidia of Aspergillus fumigatus were intra-tracheal administered to rats, immunosuppressed with cortisone acetate. Reduction of lung fungal burden, 24 hours after infection, confirms PTX3 ability to promote fungal clearance from the lung likely by enhancing phagocytosis and killing of conidia by resident innate immunity cells, thus indicating that the opsonic activity of the protein is exploitable also on the respiratory mucosa. Present results provide clues for the direct delivery of PTX3 to the lung.
Keywords
Phagocytosis; Opsonic activity; Fungal clearance
Introduction
Invasive pulmonary aspergillosis (IPA) represents a significant threat among subjects immunocompromised by a number of conditions including: corticosteroid treatments, ablative cancer chemotherapy or bone marrow transplantation [1,2]. Soluble and cell-associate receptors are involved in triggering a protective immune response [3]. The multimeric glycoprotein PTX3 has been recognized as a key element in the humoral harm of innate immunity against this fungal infection [4]. PTX3 has functional plasticity, it cooperates with other innate immunity proteins including complement, Ficolins, MBL, and MD2, thus reducing microbial burden [5]. In vitro experiments with phagocytes indicate that the main mechanism of action of PTX3 against Aspergillus relies on its opsonic activity [6]. The protein has shown a protective role in several pharmacological models of infection as evaluated by a reduction of the overall mortality, enhancement of the mean survival time (MST) and reduction of both blood and lung fungal burden [7-9]. The aim of this study was to dissect further the opsonic activity of the PTX3, quantifying the amount of protein effectively bound to conidia and establishing the impact of differently opsonized conidia on macrophages phagocytosis and lung clearance of the fungus. In addition, this study provides new dosage insight on the potential local therapeutic use of PTX3 on the respiratory mucosa, such as aerosol.
Methods
PTX3 binding to A. fumigatus conidia
To estimate the amount of PTX3 effectively bound to conidia, 5 × 107 conidia/ml in saline phosphate solution (PS) (150 mM NaCl, 10 mM phosphate buffer pH 7-7.2) were incubated 1 hour at room temperature with PTX3 at final concentrations in the range 1000-2 μg/ml. To remove unbound protein, conidia were collected by centrifugation and washed three times with PS. Conidia pellet was then lysed in 0.2 ml of Lamely buffer and the extracts (10 μl/line), as well as a PTX3 standard, resolved by SDS-PAGE. PTX3 was detected by western blotting with anti-PTX3 monoclonal antibody (MNB4). Densitometric analysis in comparison with PTX3 standard, using IMAGEJ software, allowed calculating the amounts of PTX3 effectively bound to conidia.
In vivo PTX3 opsonic activity
To investigate the effect of PTX3 opsonization on in vivo clearance of Aspergillus conidia, a rat model of invasive pulmonary aspergillosis (IPA) was used. Rats were immunosuppressed and infected as previously described [8,9]. Briefly, Sprague-Dawley rats (Envigo) were immunosuppressed with 150 mg of cortisone acetate (CA)/kg of body weight 6 days before and then every other day up to the day of infection and maintained with 80 mg/kg every other day until the end of the experiment. Conidia were obtained from 4- and 5-day cultures in Sabouraud agar medium at 28°C and scraped in a Sabouraud broth medium (0.05% Tween 80). Rats were intratracheally inoculated with a single administration of 5 × 107 conidia of A. fumigatus in 0.2 ml of sterile saline with or without PTX3 pre-opsonization at the indicated concentrations. This inoculum led to mortality within 12 to 13 days without proper therapy and provided an extensive lung infection.
In Vitro PTX3 opsonic activity
A. fumigatus inactivated conidia (108/ml) were labelled by incubation with the pH-sensitive Amine-Reactive pHrodo™ Green Dye 0.5 mM (Thermo Fisher Scientific P35369). Conidia labelling and pH-dependence of fluorochrome emission were assessed by suspending them in different buffers (pH range 4-8) and using the Glomax Multimode plate reader (Promega; Exc. 520 nm, Emis. 580-640 nm). RAW cells were cultured with conidia (1:5, being previously assessed as the optimal ratio) with or without PTX3 opsonization at different concentrations (0-50 μg/ml). After 6 or 18 hours, cells (2 × 104) were washed and analyzed by flow cytometry by suspending them in PBS (pH 7.4) and using a FACSCalibur (BD Biosciences) to detect fluorescence.
Results
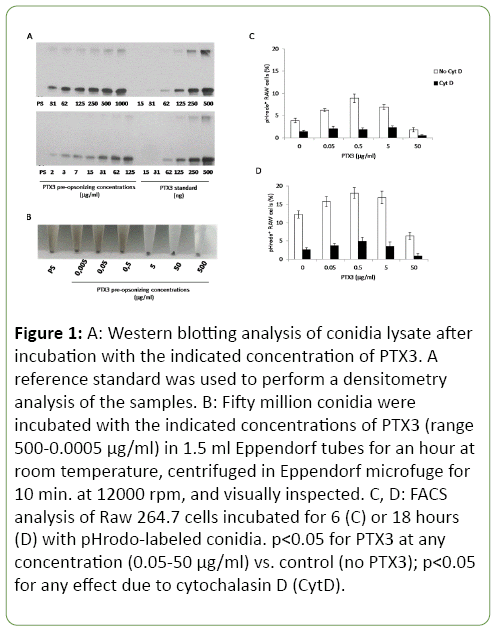
Saturation of PTX3 binding to conidia was obtained at 500 μg/ml of the pre-opsonization mix. A dose dependent binding was observed (Figure 1A). The lowest detectable binding was obtained at 2 μg/ml, that corresponds to 742 ng of bound PTX3 for 5 × 107 conidia. Assuming a molecular weight for PTX3 of 340 kDa [10,11] the number of pmoles of PTX3 bound to 5 × 107 conidia at each concentration of PTX3 in the preopsonization mix was calculated (Table 1). Multiplying PTX3 molarity with the Avogadro number we estimated that the number of molecules of PTX3 bound to single conidia, at 2 μg/ml pre-opsonization mix, was about 2.4 × 104.
Figure 1: A: Western blotting analysis of conidia lysate after incubation with the indicated concentration of PTX3. A reference standard was used to perform a densitometry analysis of the samples. B: Fifty million conidia were incubated with the indicated concentrations of PTX3 (range 500-0.0005 μg/ml) in 1.5 ml Eppendorf tubes for an hour at room temperature, centrifuged in Eppendorf microfuge for 10 min. at 12000 rpm, and visually inspected. C, D: FACS analysis of Raw 264.7 cells incubated for 6 (C) or 18 hours (D) with pHrodo-labeled conidia. p<0.05 for PTX3 at any concentration (0.05-50 μg/ml) vs. control (no PTX3); p<0.05 for any effect due to cytochalasin D (CytD).
Table 1: the amounts of PTX3 bound to conidia as calculated from the densitometry of western blotting in Figure 1A.
| PTX3 in the opsonization mix (µg/ml) | PTX3 bound (ng/5 × 107 conidia) | PTX3 bound (pmol/5 × 107 conidia) |
|---|---|---|
| 1000 | 5183 | 15 |
| 500 | 5126 | 15 |
| 250 | 4608 | 14 |
| 125 | 3556 | 10 |
| 62.5 | 2971 | 9 |
| 31.5 | 2269 | 7 |
| 15.6 | 2016 | 6 |
| 7.8 | 1732 | 5 |
| 3.9 | 1498 | 4 |
| 2 | 742 | 2 |
| 0.0 | 0.0 | 0.0 |
Noteworthy, during conidia collection by centrifugation, the amount of floating conidia (fraction resistant to sedimentation) was progressively less with the increase in PTX3 concentration (Figure 1B). This suggests that PTX3 might either work as a surfactant protein thus reducing hydrophobic interaction between conidia and plastic or that it might promote the formation of large aggregates of protein and conidia with a high sedimentation rate compared with less or non-opsonized conidia.
In order to evaluate the contribution of PTX3 opsonic activity in the clearance of the fungus from the respiratory mucosa, infection with PTX3 pre-opsonized conidia was investigated in rats. Twenty-four hours after infection, rats were sacrificed, lungs were collected and fungal CFU counted. A first experiment was performed with PTX3 at 2-500 μg/ml. Pre-opsonization of conidia with PTX3 at 500 μg/ml slightly worsened the lung CFU outcome (16552 CFU) as compared to naked conidia (13692 CFU) (p=0.7). Increased, but not significant, reduction of lung CFU was observed at PTX3 concentrations of 30 μg/ml (2145 CFU; vs. saline 13692 CFU p=0.15) and 2 μg/ml (4142 CFU; vs. saline 13692 CFU p=0.27).
In a second experiment, conidia were pre-opsonized with PTX3 at 0.5-500 ng/ml. A reduction of lung CFU was observed at all tested concentrations, being significant at 50 (p=0.04), and 0.5 (p=0.02) ng/ml (Table 2). To assess the effects of PTX3 on phagocytosis, conidia were incubated with the monocyticmacrophagic cell line RAW264. In buffers at pH 7-8, the fluorescence of pHrodo-labelled conidia was undetectable or barely detectable whereas at pH 6 it could be clearly detected and raised al lower pH values (not shown). Results in Figure 1C and 1D, show that the percentage of cells that had phagocytized naked A. fumigatus conidia and disposed them to the acidic environment of phagolysosomes was 3.9% and 12.3% after 6 and 18 hours, respectively. In the presence of PTX3, the percentage of pHrodo positive cells was higher, reaching a maximum (8.9% after 6 h, 18.1% at 18 h) at 0.5 μg/ml. The addition of cytochalasin D dropped the percentage of fluorescent cells, confirming that the observed signal was associated with phagocytosis of fluorescent conidia. Interestingly, the number of fluorescent cells was lower at higher PTX3 concentrations (1.8% and 6.4% at 50 μg/ml) at both 6 and 18 hours, respectively, suggesting that an optimal “stoichiometric” ratio between conidia and PTX3 is required for optimal phagocytosis.
Table 2: Lung fungal burden of rats intratracheally infected with A. fumigatus conidia pre-opsonized with the indicated concentrations of PTX3.
| PTX3 in the opsonization mix (ng/ml) | n | CFU (mean) | SE | T-test versus Vehicle |
|---|---|---|---|---|
| Vehicle | 12 | 299083 | 83145 | - |
| 500 | 12 | 156633 | 57063 | 0.25 |
| 50 | 12 | 85626 | 35319 | 0.04 |
| 5 | 12 | 130950 | 46126 | 0.08 |
| 0.5 | 12 | 83350 | 22385 | 0.02 |
n: Number of Rat/Group, SE: Standard Error
In previous experiments of pulmonary infection with A. fumigatus, in bone marrow transplanted mice [7], the most effective dose of PTX3 was 1 mg/kg [7]. In order to evaluate whether the lung concentration of PTX3 was consistent with our in vivo and in vitro results, 1 mg/kg of the protein was intravenously administered in PTX3 deficient mice (n=6) and lung homogenate evaluated by ELISA for PTX3, 2 hours after protein administration. PTX3 concentrations spanned between 7 and 60 ng/lung (mean 25.7 ± 20) thus in a range of concentrations consistent with the results reported herein.
Discussion
The present study provides clear evidence that the opsonic activity of PTX3 plays a relevant role in the clearance of A. fumigatus from the respiratory mucosa. The range of phagocytosis-promoting concentrations used to opsonize conidia was found consistent with the concentrations obtained in the lung after systemic administration of pharmacologically effective doses of the protein. According to the multimeric status of PTX3, previous studies suggest that the protein might work as hub, thus clustering conidia to the surface of innate immunity cells and enhancing both recognition and conidia engulfment by phagocytes [12-14]. We found that the number of molecules of PTX3 that remain bound to conidia after pre-opsonization was considerably high (up to 150.000 molecules/conidia) thus suggesting that the protein is able to assemble in large complexes of proteins and conidia. Our data indicate that high concentrations of PTX3 (500 μg/ml) in pre-opsonization experiments is ineffective or even detrimental to lung fungal clearance, suggesting that if these complexes became too large engulfment by phagocytes could be unmanageable. In agreement with this hypothesis, in vitro phagocytosis assays and in vivo infection experiments revealed that the PTX3 concentrations promoting conidia engulfment and those reducing lung fungal burdens in vivo were falling in the same range (0.05 to 5 μg/ml). In conclusion, the present study provides evidences that administration of PTX3 to the respiratory mucosa might be effective in enhancing lung fungal clearance. Direct delivery of PTX3 to the lung, for instance by aerosol, might represent a new therapeutic option and accordingly to opsonization results, PTX3 dosage should be carefully estimated to prevent detrimental effects on fungal clearance mechanisms.
Conflict of Interest
The authors Giovanni Salvatori and Rita DeSantis are inventors on patents regarding the use of PTX3 in infection diseases including those caused by A. fumigatus.
References
- Zmeili OS, Soubani AO (2007) Pulmonary aspergillosis: a clinical update. Q J Med 100: 317-334.
- Latgé JP (2001) The pathobiology of Aspergillus fumigatus. Trends Microbiol 9(8): 382-389.
- Romani L (2004) Immunity to fungal infections. Nature Review Immunology 4: 1-13.
- Garlanda C, Bottazzi B, Bastone A, Mantovani A (2005) Pentraxins at the crossroads between innate immunity, inflammation, matrix deposition, and female fertility. Annu Rev Immunol 23: 337-366.
- Moalli F, Jaillon S, Inforzato A, Sironi M, Bottazzi B, et al. (2011) Pathogen Recognition by the Long Pentraxin PTX3. J Biomed Biotechnol 2011: 830421.
- Garlanda C, Hirsch E, Bozza S, Salustri A, De Acetis M, et al. (2002) Non-redundant role of the long pentraxin PTX3 in anti-fungal innate immune response. Nature 420: 182-186.
- Gaziano R, Bozza S, Bellocchio S, Perruccio K, Montagnoli C, et al. (2004) Anti-Aspergillus fumigatus efficacy of pentraxin 3 alone and in combination with antifungals. Antimicrob Agents Chemother 48(11): 4414-4421.
- Giudice PL, Campo S, Verdoliva A, Rivieccio V, Borsini F, et al. (2010) Efficacy of PTX3 in a rat model of invasive aspergillosis. Antimicrob Agents Chemother 54(10)4513-4515.
- Giudice PL, Campo S, De Santis R, Salvatori G (2012) Effect of PTX3 and voriconazole combination in a rat model of invasive pulmonary aspergillosis. Antimicrob Agents Chemother 56(12): 6400-6402.
- Inforzato A, Rivieccio V, Morreale AP, Bastone A, Salustri A, et al. (2008) Structural characterization of PTX3 disulphide bond network and its multimeric status in cumulus matrix organization. J Biol Chem 283(15): 10147-10161.
- Inforzato A , Baldock C, Jowitt TA, Holmes DF, Lindstedt R, et al. (2010) Quaternary structure of the long pentraxin PTX3 and its functional implications in angiogenesis. J Biol Chem 285: 17681-17692.
- Moalli F, Doni A, Deban L, Zelante T, Zagarella S, et al. (2010) Role of complement and Fcg receptors in the protective activity of the long pentraxn PTX3 against Aspergillus fumigatus. Blood 116: 5170-5180.
- Bozza S, Campo S, Arseni B, Inforzato A, Lindstat R, et al. (2014) PTX3 binds to MD-2 and promotes TRIF-dependent immune protection in aspergillosis. J of Immunology 193: 2340-2348.
- Diniz SN, Nomizo R, Cisalpino PS, Teixeira MM, Brown GD, et al. (2004) PTX3 function as opsonin for the dectin-1-dependent internalization of zymosan by macrophages. J Leukocyte Biology 75: 649-665.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences