Investigation of Blood Glucose Profile by Continuous Glucose Monitoring (CGM)
Hiroshi Bando1,2*, Koji Ebe2,3, Yoshikane Kato4, Masahiro Bando5 and Yoshikazu Yonei6
1Tokushima University/Medical Research, Tokushima, Japan
2Low Carbohydrate Diet Promotion Association, Kyoto, Japan
4Kanaiso Hospital, Tokushima, Japan
5Department of Nutrition and Metabolism, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan
6Anti-Aging Medical Research Center, Graduate School of Life and Medical Sciences, Doshisha University, Kyoto, Japan
- *Corresponding Author:
- Hiroshi Bando
Tokushima University/Medical Research
Nakashowa 1-61, Tokushima 770-0943 Japan
Tel: +81-90-3187-2485
E-mail: pianomed@bronze.ocn.ne.jp
Received Date: 21 February 2018; Accepted Date: 20 April 2018; Published Date: 27 April 2018
Citation: Bando H, Ebe K, Kato Y, Bando M, Yonei Y (2018) Investigation of Blood Glucose Profile by Continuous Glucose Monitoring (CGM). Endocrinol Res Metab Vol. 2 No 1:10.
Copyright: © 2018 Bando H, et al. This is an open-access article distributed under the terms of the creative Commons attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Diabetes has been great problem worldwide. Continuous Glucose Monitoring (CGM) has been in focus, and useful CGM, FreeStyle Libre (Abbott) has been introduced to clinical ordinary practice of medicine. Case and results: The patient was 57 year-old female with type 1 diabetes mellitus (T1DM), who showed BMI 20.6 kg/m2, HbA1c 8.6%. She was on CGM measured glucose every 15 minutes in 24 hours for 14 days. Treatment included 3 times of Insulin Aspart and once of Insulin Glargin. She showed characteristic pattern of blood glucose profile according to the working schedule of the day. Discussion and conclusion: The beneficial points of FreeStyle Libre have been convenient and accurate for clinical use for reducing hypoglycemia episodes. Treatment using CGM associated with multiple daily insulin injection (MDI) confers similar or greater benefits for glycemic profile. When evening insulin was not injected one day, blood glucose increased 320 mg/dL, suggesting 64 g of carbohydrate ingested for supposed calculation. These results suggest that CGM could recommend the timing and doses of insulin injection according to the lifestyle of the patients with clinical merit.
Keywords
Continuous glucose monitoring; FreeStyle libre; Type 1 diabetes mellitus; Type 2 diabetes mellitus; Blood glucose profile
List of abbreviations
DCCT: Diabetes Control and Complications Trial; ADA: The American Diabetes Association; MDI: Multiple Daily Insulin; rtCGM: real-time Continuous Glucose Monitoring; FDA: Food and Drug Administration
Introduction
The prevalence of diabetes has been increasing globally and a multifactorial approach is necessary to decrease several risk of complications in this situation [1]. Maintaining the adequate blood glucose has been the key to prevent microvascular and macrovascular diseases [2]. Consequently, optimizing glucose levels has been required for the patients with Type 1 Diabetes Mellitus (T1DM) and Type 2 diabetes mellitus (T2DM) [3].
There have been treatment of insulin therapy for older adults with T1DM and T2DM, and it has performed successfully with the development of continuous glucose monitoring (CGM).
Patients with T1DM has been living longer [4], and its number of older adults has been growing [5]. Consequently, insulin-treated older T1DM patients have longer diabetic history and tend to have micro- and macrovascular complications and other comorbidities [5,6].
Such patients have higher risk for severe hypoglycemia, leading to other severe states such as cerebral vascular accident (CVA), myocardial infarcts, arrhythmias, temporary or permanent cognitive impairment and death [5,7].
In order to avoid hypoglycemia, The American Diabetes Association (ADA) has recommended higher HbA1c and glycemic goals which vary according to each comorbid status in older adults [6]. CGM has been effective for avoid hypoglycemia and the The DIAMOND study with T1DM treated with multiple injection therapy, showed improved HbA1c with less hypoglycemia [8]. This study was a prospective, randomized clinical trial of CGM for adults.
There has been a useful device for CGM, which has been introduced to clinical practice. It is FreeStyle Libre, produced by Abbott, USA [9,10]. In this study, daily profile of blood glucose in patient with T1DM was measured by FreeStyle Libre and investigated detail glucose fluctuation.
Case Presentation
The patient was 57 year-old female with T1DM. She was diagnosed as T1DM 10 years ago, and has been continued insulin therapy after that. Her recent HbA1c has been around 7.5%-8.6% and blood glucose ranges from 50 mg/dL to 400 mg/dL. These results are supposed to be rather unstable and not in satisfactory diabetic controlled state.
Consequently, we tried to evaluate her current diabetic condition in detail, and we have measured the profile of blood glucose using FreeStyle Libre (Abbott).
On physical examination, her consciousness and vitals are normal, and heart, lung, abdomen and neurological findings were unremarkable. She showed 159 cm in height, 52 kg in weight, BMI 20.6 kg/m2, abdominal circumference 83 cm and thigh circumference 42 cm.
Laboratory findings on January 2018 were as follows: postprandial blood glucose on 216 mg/dL after 150 min of meal, and HbA1c was 8.5%. She showed unremarkable data about complete blood count, liver function test, renal function and lipids profile.
Current insulin treatment included Novo rapid (Novo Nordisk) 3 times a day, and Insulin Glargine (Eli Lilly and Company) of once a day. The former is Insulin Aspart given by pre–filled pen including 100 units/mL, and the latter is Insulin Glargine by BS injection kit FFP including 300 units/mL. The time and units are as follows: the former is injected 8, 10, 10 units on 0700h, 1230h, 1830h and the latter is injected 11 units on 2300h.
Continuous glucose monitoring
CGM was studied for 14 days. Using CGM device, blood profile were measured every 15 minutes in 24 hours. We used sensor-based device for blood glucose monitoring, which is the FreeStyle Libre Flash Glucose Monitoring system, produced by Abbott Diabetes Care Inc, Alameda, CA, USA. This is firstly commercial-based available sensor systems associated with several beneficial points [11,12].
It has some advantages for replacing blood glucose testing and detecting trends and tracking patterns in order to detect the episodes of hyperglycemia and hypoglycemia. Characteristic strong points are accurate, convenient and small size for practical use [10,13]. The sensor is put and wearable on the back of the upper arm during 14 days and can automatically preserve swinging glucose data every 15 min for 14 days [9,10,14,15].
Results of CGM
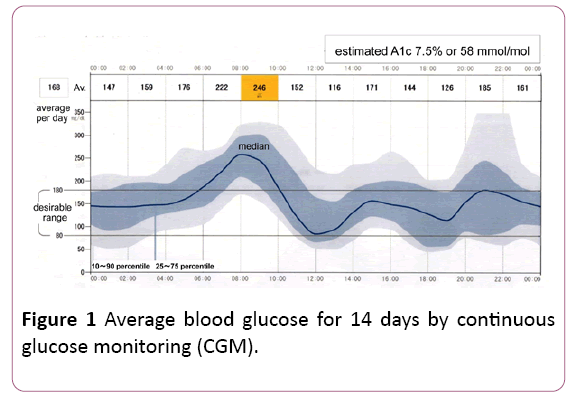
The mean value of the glycemic profile over 14 days is shown in Figure 1. In general, blood glucose tends to rise at 0800-1000 h after breakfast intake. During working, she keeps on walking all the time, then blood glucose level usually falls around 1100-1230 h. After taking light lunch, she moves again well in the second half of the afternoon, leading to blood glucose decrease in the evening. At night, she always relaxes at home, and does not move much after dinner.
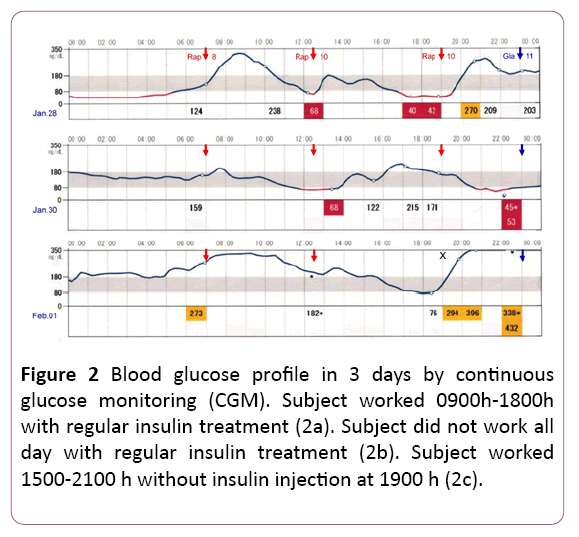
The glucose profiles in three different characteristic situations were shown in Figure 2. The lifestyle on January 28th was usual and she was working from morning until evening (Figure 2a). After taking breakfast, blood glucose increases, decreases before lunch and keeps low during 1200-1230 h. Glucose level was stable in the afternoon, and became low during 1700-1800 h because of continuation of walking. At night, glucose value was stable due to relaxed time at home.
She was off on January 30th without work, and did not walk around for the day (Figure 2b). Therefore, results with less fluctuation in blood glucose were obtained. The increase in blood glucose after 1530 h was due to intake of sugar-rich cake.
She worked during 1500-2100 h on Feb.1st (Figure 2c). Glucose fluctuation during 0900-1230h are compared with Figures 2a and 2c. When moving frequently in the morning, blood sugar falls well (Figure 2a), but when keeping still, it does not go down (Figure 2c). As she started working from 1500 h, blood sugar gradually declined. She took snacks in 1830 h, but she could not inject the insulin on the evening. After that, hyperglycemia persisted for long, although Glargine was given on 2300 h. The hyperglycemia lasted until the next morning, in which blood glucose was 220 mg/dL at 0700 h.
Discussion
Treatment using real-time CGM (rtCGM) associated with multiple daily insulin (MDI) injection confers similar or greater benefits for glycemic profile [16]. Moreover, there have been lots of studies that use of rtCGM could improve glycemic control in children and adults with T1DM [17-20]. The beneficial points of rtCGM are found in cases who wear CGM devices more frequently and respond to regulate appropriately according to the glucose data provided [17,21].
Especially, CGM is clinically important for cases with T1DM on MDI injection [22]. CGM has the potential benefits of effective use of glycemic rate of change (ROC) arrows for dose adjustments of insulin [23].
As to the insulin treatment, pre-meal glycemic slope predicts post-meal glycemic excursions and may help inform insulin dosing decisions. Rate-of-change arrows on existing devices obscure clinically actionable glycemic trend information from CGM users [24].
CGM devices have developed over the past years for the standard care for patients with T1DM [25]. Compared with the traditional blood glucose monitoring (BGM), recent CGM data represent 24-hour glycemic variability and prediction of glucose changes simultaneously. Recently, there has been broader application for patients with various states [26]. Furthermore, Food and Drug Administration (FDA) in United States approved the management decisions in 2016 that include insulin dosing from CGM values [27].
CGM devices record interstitial glucose level and have the advantage of providing detail and precise glucose readings, with recently a new generation of implantable variability [28,29]. Furthermore, CGM has shown improvement in glucose variability, measured as HbA1c, reduction of hypoglycemia and cost-effectiveness [30].
Recently, clinical practice guidelines have been generally simplified the criteria for CGM use by the endocrine society [31]. According to the data from 11 RCTs using CGM for T1DM, CGM would alters HbA1c (95% CI) by -0.28% (-0.47, -0.09), and reduces the risk of hypoglycemia, particularly with lower HbA1c levels [31].
As for T1DM case studied in this study, there seemed to be three characteristic findings. Firstly, blood glucose increased during 0800-1000 h, which is probably due to rather excess intake of carbohydrate or shortage of rapid effective insulin. Secondly, blood glucose was decreasing due to the effect of continuous physical movement. It is from her lifestyle and habit that she always moves and walks around without sitting during working.
Thirdly, when insulin injection was not done at 1900h, hyperglycemia has lasted overnight. In this point, blood glucose increased from 76 to 396 mg/dL, ie., rise of 320 mg/dL. In T1DM, there is averagely an increase of 5 mg/dL of blood glucose per 1g of carbohydrate intake. Then the ingested sugar amount was calculated to be about 64 g. Ingested carbohydrate was actually 72 g, which seemed to be roughly consistent.
Conclusion
As to this investigation, blood glucose profile can be clarified in detail by application of CGM, indicating clinical usefulness. Furthermore, it is possible to recommend the timing and dose of insulin injection according to the lifestyle of the patient, suggesting a great clinical merit. These results would be useful as basic data for better treatment and clinical research in T1DM.
Supplement
This study was conducted in compliance with the ethical principles of the Declaration of Helsinki. In addition, it was with Japan’s Act on the Protection of Personal Information along with the Ministerial Ordinance on Good Clinical Practice (GCP) for Drug (Ordinance of Ministry of Health and Welfare No. 28 of March 27, 1997). We held ethical committee meeting including physicians, nurse, pharmacist, clinical engineer and academic experts. Informed consent was obtained from the subject.
The authors would like to thank the patient and staffs for their cooperation and support. The authors declare that they have no conflicts of interest.
References
- Gregg EW, Sattar N, Ali MK (2016) The changing face of diabetes complications. Lancet Diabetes Endocrinol 4:537-547.
- The Diabetes Control and Complications (DCCT) Research Group (1995) Effect of intensive therapy on the development and progression of diabetic nephropathy in the Diabetes Control and Complications Trial. Kidney Int 47:1703-1720.
- Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, et al. (2005) Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 353:2643-2653.
- DCCT/EDIC Study Research Group (2016) Mortality in type 1 diabetes in the DCCT/EDIC versus the general population. Diabetes Care 39:1378-1383.
- Dhaliwal R, Weinstock RS (2014) Management of type 1 diabetes in older adults. Diabetes Spectr 27: 9-20.
- Chiang JL, Kirkman MS, Laffel LM, Peters AL (2014) Type 1 diabetes through the life span: a position statement of the American Diabetes Association. Diabetes Care 37: 2034-2054.
- Seaquist ER, Anderson J, Childs B, Cryer P, Dagogo-Jack S, et al. (2013) Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association. J Clin Endocrinol Metab98:1845-1859.
- Beck RW, Riddlesworth T, Ruedy K, Ahmann A, Bergenstal R, et al. (2017) Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin Injections: The DIAMOND Randomized Clinical Trial. JAMA 317:371-378.
- https://www.freestylelibre.us/
- Edge J, Acerini C, Campbell F, Hamilton-Shield J, Moudiotis C, et al. (2017) An alternative sensor-based method for glucose monitoring in children and young people with diabetes. Arch Dis Child 102: 543-549.
- Slattery D, Choudhary P (2017) Clinical use of continuous glucose monitoring in adults with type 1 diabetes. Diabetes Technol Ther 19: S55-S61.
- Hoss U, Budiman ES (2017) Factory-calibrated continuous glucose sensors: The science behind the technology. Diabetes Technol Ther 19: S44-S50.
- Sekido K, Sekido T, Kaneko A, Hosokawa M, Sato A, et al. (2017) Careful readings for a flash glucose monitoring system in nondiabetic Japanese subjects: individual differences and discrepancy in glucose concentrarion after glucose loading. Endocr J 64:827-832.
- Bailey T, Bode BW, Christiansen MP, Klaff LJ, Alva S (2015) The performance and usability of a factory-calibrated flash glucose monitoring system. Diabetes Technol Ther 17:787-794.
- Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, et al. (2017) Use of flash glucose-sensing technology for 12 months as a replacement for blood glucose monitoring in insulin-treated type 2 diabetes. Diabetes Ther 8:573-586.
- Parkin CG, Graham C, Smolskis J (2017) Continuous glucose monitoring use in type 1 diabetes: longitudinal analysis demonstrates meaningful improvements in HbA1c and reductions in health care utilization. J Diabetes Sci Technol 11: 522-528.
- Pettus J, Price DA, Edelman SV (2015) How patients with type 1 diabetes translate continuous glucose monitoring data into diabetes management decisions. Endocr Pract21:613-620.
- New JP, Ajjan R, Pfeiffer AF, Freckmann G (2015) Continuous glucose monitoring in people with diabetes: The randomized controlled Glucose Level Awareness in Diabetes Study (GLADIS). Diabet Med 32:609-617.
- Hermanns N, Schumann B, Kulzer B, Haak T (2014) The impact of continuous glucose monitoring on low interstitial glucose values and low blood glucose values assessed by point-of-care blood glucose meters: results of a crossover trial. J Diabetes Sci Technol 8:516-522.
- Hommel E, Olsen B, Battelino T, Conget I, Schütz-Fuhrmann I, et al. (2014) Impact of continuous glucose monitoring on quality of life, treatment satisfaction, and use of medical care resources: analyses from the SWITCH study. Acta Diabetol 51:845-851.
- Chamberlain JJ, Dopita D, Gilgen E, Neuman A (2015) Impact of frequent and persistent use of continuous glucose monitoring (CGM) on hypoglycemia fear, frequency of emergency medical treatment, and SMBG frequency after one year. J Diabetes Sci Technol 10:383-388.
- Maiorino M, Petrizzo M, Bellastella G, Esposito K (2017) Continuous glucose monitoring for patients with type 1 diabetes on multiple daily injections of insulin: pros and cons. Endocrine 59: 62-65.
- Giulio F, Valeria F, Clara B (2017) Comment on “The pros and cons of continuous glucose monitoring for patients with type 1 diabetes on multiple daily injections of insulin.” Endocrine 59: 230.
- Majithia AR, Wiltschko AB, Zheng H, Walford GA, Nathan DM (2018) Rate of change of premeal glucose measured by continuous glucose monitoring predicts postmeal glycemic excursions in patients with type 1 diabetes: Implications for therapy. J Diabetes Sci Technol 12:76-82.
- Forlenza GP, Argento Nb, Laffel LM (2017) Practical Considerations on the use of continuous glucose monitoring in pediatrics and older adults and nonadjunctive use. Diabetes Technol Ther 19: S13-S20.
- Rodbard D (2016) Continuous glucose monitoring: a review of successes, challenges, and opportunities. Diabetes Technol Ther 18: S3-S13.
- https://diatribe.org/fda-approves-dexcom-g5-cgm-for-insulin-dosing
- Ajjan RA (2017) How can we realize the clinical benefits of continuous glucose monitoring? Diabetes Technol Therap 19: S27-S36.
- Kropff J, Choudhary P, Neupane S, Barnard K, Bain SC, et al. (2017) Accuracy and longevity of an implantable continuous glucose sensor in the PRECISE study: A 180-day, prospective, multicenter, pivotal trial. Diabetes Care 40:63-68.
- Vigersky RA (2015) The benefits, limitations, and cost-effectiveness of advanced technologies in the management of patients with diabetes mellitus. J Diabetes Sci Technol 9:320-330.
- Peters AL, Ahmann AJ, Battelino T, Evert A, Hirsch IB, et al. (2016) Diabetes technology-continuous subcutaneous insulin infusion therapy and continuous glucose monitoring in adults: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 101:3922-3937.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences