ISSN : ISSN: 2576-1412
Journal of Applied Microbiology and Biochemistry
Influence of Galvanic Microcells on Growth of Pathogenic Bacteria and Candida Yeast
Teresa Krzysko-Lupicka*, Magdalena Krecidlo, Lukasz Krecidlo and Wojciech Spisak
1Department of Biotechnology and Molecular Biology, University of Opole, Poland
2Research & Development Centre, Poland
- *Corresponding Author:
- Teresa Krzysko-Lupicka
Department of Biotechnology and Molecular Biology
University of Opole, Opole, Poland
Tel: (077) 401 6057
E-Mail: lupickizbyszko@poczta.onet.pl
Received Date: January 27, 2017; Accepted Date: February 03, 2017; Published Date: February 10, 2017
Citation: Krzysko-Lupicka T, Krecidlo M, Krecidlo L, et al. Influence of Galvanic Microcells on Growth of Pathogenic Bacteria and Candida Yeast. J Appl Microbiol Biochem. 2017, 1:1.
Abstract
The aim of the study was to evaluate the influence of electrodes on growth of pathogenic bacteria and Candida albicans yeast. In the study, following references bacteria strains were used: Staphylococcus aureus ATTC 6538; Enterococcus faecalis ATTC 29212 (V); Enterococcus hirae ATTC 105441 (VI) and yeast strain Candida albicans ATTC 10231 (C40). Microelectrodes were prepared by ALCOR Center. Estimation of the biocidal effect of electrodes was carried out by medium poisoning method. The assessment of adhesion to the surface was performed by a modified Hawser’s method. Presence of adhesion was assessed by SEM analysis. The highest inhibition zones were obtained for Staphylococcus aureus ATTC 6538. In cases of Enterococcus faecalis ATCC 29212 and Enterococcus hirae ATTC 105441, the largest inhibition zones were noted for combination of Zn-Cu electrodes and Zn-Ag electrodes. Microelectrodes in combination with zinc and titanium (Zn-Ti) inhibited ability to surface adherence of S. aureus and E. faecalis. E. hirae ATTC 105441 had no ability to adherence to the surface.
Keywords
Staphylococcus inhibition; Enterococcus inhibition; Candida reaction; Microcells
Introduction
The pathogens surfaces contamination is one of the more important issues in medicine, especially in orthopedic surgery [1]. The adhesion of pathogenic bacteria to surfaces is a starting point of biofilm forming. Due to the increasingly better understanding the adhesion of microorganisms and their mode of interaction with the surface, methods are used, which are intended to protect the surface from this phenomenon [2-5]. These methods are based on the modification of surface being in contact with organic matter. The most common modification is to change the surface roughness, the shape, or hydrophobicity [6]. Another solution is to use metal ions, which has a centuries-old tradition. For example, vessels made of Cu and Ag was used for water disinfection and food preservation by Persian, Phoenicians, Greeks, Romans and Egyptians [7-10]. Today, we focus on creating the cover made of nanoparticles or bio-available reactive metal forms immersed in the polymer or the zeolite. Metals and their ions, in addition to the antimicrobial properties, have antiadhesive properties, which protect against biofilm formation. An example is the inhibition of proliferation of Staphylococcus aureus to the surface by means of using the zeolite shell coated with silver ions [11]. An additional advantage of the electrodes is to produce the electromagnetic field that disrupts bacterial cell interaction with the surface. There are known coats using the principle of the sacrificial electrode action. Electrochemically more active metal acts as a protector for the electrode made of metal that is less active [12]. Strengthening the anti-microbial properties using the sacrificial anode in systems such as Pt/Ag, Al/Ag, Al/Pt was patented [13,14]. The effect of electromagnetic field generated by the galvanic systems on bacteria and yeast has not been described yet. Scientific reports in this area are quite divergent [15] report that the magnetic field causes morphological changes in fungi of Aspergillus genus indicate the intensification of Staphylococcus aureus and S. epidermalis growth. On the other hand [16] argue that electromagnetic field causes a reduction in the number of bacteria and affects the shift of mid-log phase.
The aim of the study was to evaluate the effect of single electrodes and two electrode systems on the growth and adhesive ability of selected reference strains of pathogenic bacteria and Candida albicans yeast [17].
Materials and Methods
Microorganism’s strains
In the study, following bacteria strains were used: Staphylococcus aureus ATTC 6538; Enterococcus faecalis ATTC 29212 (V); Enterococcus hirae ATTC 105441 (VI) and yeast strain Candida albicans ATTC 10231 (C40).
Scheme of studies
The study was conducted in two stages. In the first one, analysis of the effect of galvanic cells placed on polystyrene Petri dishes for growth of planktonic microorganisms was performed. The second type of experiment was to create polystyrene plates measuring 1 cm2 which was a plot of microelectrodes (Table 1). The process was to prepare plates to a surface of a slide of liquid styrene dissolved in toluene/xylene. They were then applied to the microelectrode in the form of powder [18-20]. The study was carried out in two stages. In the first one, the effect of galvanic cells placed on polystyrene Petri dishes on the growth microorganisms at planktonic phase, was analyzed. The second type of experiment was based on creating the polystyrene plates of 1 cm2 are, on which microelectrodes were applied (Table 1). The process of preparing the plates consisted in application of a liquid styrene dissolved in toluene/xylene onto the Petri dish surface. Then the microelectrode in the form of powder were applied. After drying, the polymer was washed with sterile water. Because the arrangement of electrodes on the polystyrene plate was random, there were active electrodes on the surface and completely immersed in the styrene. In order to check the distance between different metals and the type of electrode, the electron micrograph coupled with mass spectroscopy was applied. In this part of the study, the influence of microelectrodes embedded on the polystyrene plate on the ability of bacteria and Candida albicans yeast to adhesion and biofilm formation was analyzed. The experiments were performed both for the two electrodes systems as for the individual electrodes (Table 1). Bacteria were incubated for 24 h in Nutrient Agar (Biocorp) and yeast strain was incubated for 48 h in Sabouraud’s Dextrose Agar (Biocorp). Next, cultures were washed with Ringer’s solution and cell suspensions were spectrophotometrically standardized (108 CFU • cm-3). The study was conducted in 4 replicates. Results were reported as the diameter of growth inhibition zone (d) ± standard deviation.
Table 1 The list of tested metals electrodes.
| Macroelectrodes | Microelectrodes |
|---|---|
| Zn | SiO2 (control) |
| Ti | Zn |
| Mg | Ti |
| Cu | Cu |
| Ag | Ti-Zn |
| Mg-Cu | Zn-Cu |
| Ti-Cu | Ti-TiO |
| Zn-Ti | |
| Zn-Cu | |
| Zn-Ag |
Antimicrobial plate test
The assessment of influence on microbial planktonic growth was carried out by modified medium poisoning method. In the study, 1 cm3 of cell suspensions was inoculated into Tryptone Soya Agar (Biocorp) or Sabouraud’s Dextrose Agar (Biocorp). Such prepared inoculum was transferred onto polystyrene Petri dishes with electrodes. The distance between electrodes was from 0.8 to 1.2 cm. Diameter of electrodes (d), depending on the metal, was from 0.8 to 2 mm. Each sample was incubated at 35°C.
Adhesion ability test
The estimation of the adhesion ability was performed by a modified Hawser’s method. Before using, the styrene plate with electrodes was washed with ethanol and then immersed in demineralized sterile water [21]. Standardized cells suspensions of tested microorganisms were poured into 5 cm3 of nutrient broth (for bacterial strains) and liquid Sabouraud’s medium (for examination of yeast). Sterile styrene plates (1 × 1 cm) with deposited microelectrodes were placed onto microbial cultures (Table 2). Undeposited sterile styrene plates and styrene plates with SiO2 were the control. Cultures were incubated at 35°C in shaker at 100 rpm for 72 h and then washed with EtOH in increasing concentration of 50%, 75% and 95% [22]. They were used as controls. Each plate was dried and samples were coated with gold and photographed using a scanning electron microscope (SEM).
Table 2 Inhibition zones of the microbial growth in the presence of electrodes.
| Electrode | Enterococcus faecalis | Enterococcus hirae | Staphylococcus aureus | Candida albicans |
|---|---|---|---|---|
| Zn | 5.0 ± 1.0 | 0.0 ± 0.0 | 28.0 ± 2.0 | 9.013 |
| Ti | 7.0 ± 1.5 | 0.0 ± 0.0 | 7.5 ± 2.0 | 0.0 ± 0.0 |
| Mg | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 | 0.0 ± 0.0 |
| Cu | 12.0 ± 1.5 | 5.0 ± 0.0 | 18.0 ± 1.0 | 35.012 |
| Ag | 0.0 ± 0.0 | 0.0 ± 0.0 | 10.0 ± 2.0 | 25.013 |
| Mg-Cu | 5.0 ± 0.0 | 0.0 ± 0.0 | 21.0 ± 1.0 | 0.0 ± 0.0 |
| Ti-Cu | 0.0 ± 0.0 | 5.0 ± 1.0 | 24.0 ± 2.0 | 0.0 ± 0.0 |
| Zn-Ti | 7.0 ± 1.0 | 9.0 ± 1.0 | 29.0 ± 1.0 | 0.0 ± 0.0 |
| Zn-Cu | 7.0 ± 1.0 | 9.0 ± 1.0 | 30.0 ± 1.0 | 35.0123 |
| Zn-Ag | 9.0 ± 0.5 | 10.0 ± 1.0 | 30.0 ± 1.0 | 0.0 ± 0.0 |
Results
Antimicrobial plate test
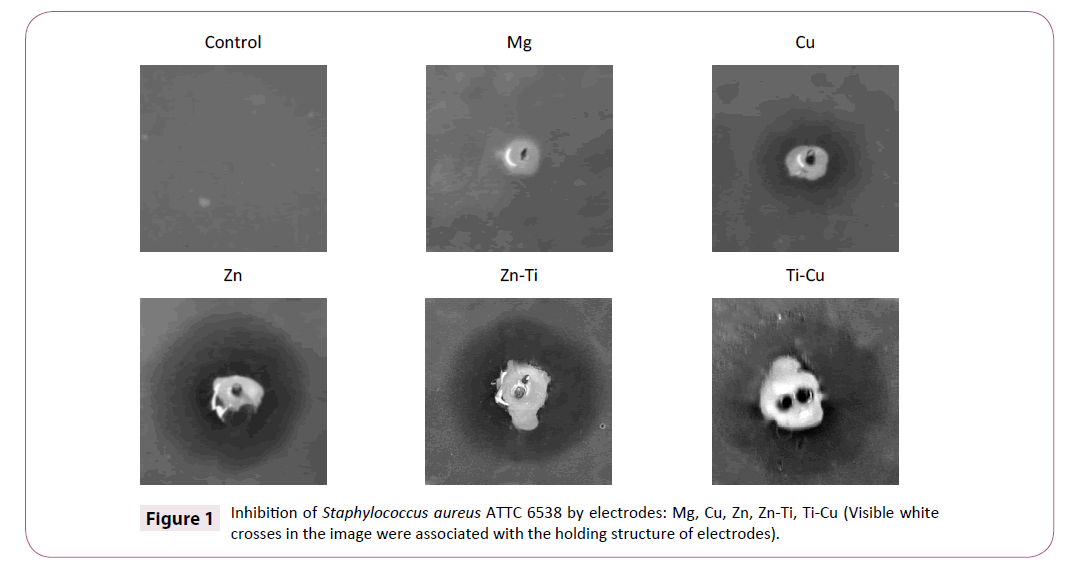
Growth of bacteria and yeast strains was analyzed in 6 cases of a single electrode and in 4 cases of combination of two electrodes. The highest antimicrobial activity of electrodes was obtained for Gram-positive reference strain of Staphylococcus aureus ATCC 6538. The largest diameters of zones were observed in cases of: Zn-Cu (30 mm), Ti-Zn (29 mm), Zn (28 mm), Ti-Cu (24 mm), Cu (18) electrodes (Table 2) [23]. Zinc electrode immersed in an electrolyte, which is the medium, causes a high inhibition of S. aureus growth. In combination with other electrode systems (Zn-Cu and Zn-Ag) slightly larger inhibition zones were obtained, but the differences between them were not significant (± 2 mm). Other dependencies were recorded in the case of copper and titanium electrodes. In experiments involving a single copper electrode, the inhibition zone was 18 mm, while for samples with titanium electrode; the zone was only 7.5 mm [24].
When the copper electrode was combined with titanium one, the inhibition zone was larger (24 mm) (Figure 1).
A similar mechanism can be observed for the copper and magnesium electrode. Only the combination results in an enhanced bactericidal activity (the zone increased by 21 mm as compared to the magnesium electrode itself and by 3 mm as compared to the copper electrode). Silver electrode caused a small inhibition zone of 10 mm, while in combination with zinc, the zone increased to 30 mm [25].
Growth of Enterococcus feacalis ATTC 29212 was compared to the control (without electrodes) on plates with: Ti, Mg, Ag electrodes and on the plate with combination of Ti and Cu electrodes. A clear line of inhibition zone for electrode Zn-Cu was not obtained. Inhibition zones for Enterococcus hirae ATTC 105441 was noted only for one electrode with Cu, but diameter of this zone was inconsiderable (5 mm). Higher antibacterial activity was shown by samples with copper electrode (12 mm) and samples with combination of electrodes containing zinc (Zn-Cu, Zn-Ag, Zn-Ti) (Table 2). No significant difference in inhibiting the growth of bacteria between individual electrodes and systems consisting of two different electrodes was observed. Tested Enterococcus strains were more resistant than S. aureus towards the electrode used.
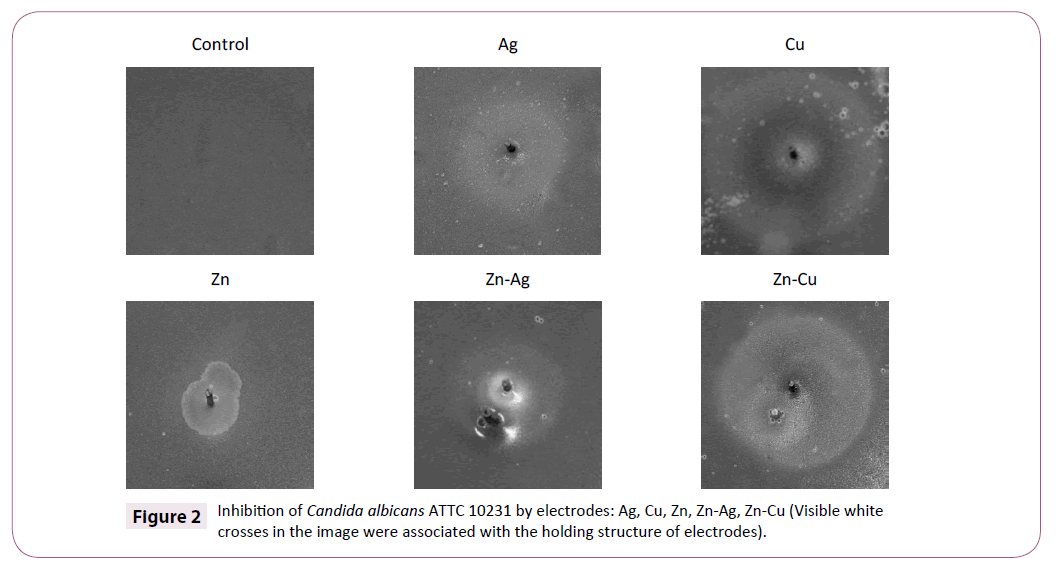
Electrodes showed no significant growth zones for Candida albicans ATTC 10231. We obtained only zones with difference in color in comparison to normal yeast growth. Copper electrodes showed zones with dark pigments, but zinc and silver electrodes have shown zones with brightest tones than normal growth (Table 2). The combination of zinc and copper electrodes indicates that dark tones around copper electrode were lesser (Figure 2).
Adhesion ability test
S. aureus was not sensitive to the presence of zinc electrode and in the presence of this electrode; it was subject to the adhesion to the surface very well. Presence of a copper electrode resulted in a partial inhibition of cell adhesion. Only a combination of copper and zinc electrodes, as well as zinc and titanium electrodes resulted in a complete inhibition of S. aureus cell adhesion (Table 3 and Figure 3).
Table 3 Inhibition zones of microbial growth in the presence of microelectrodes.
| Microelectrodes | Enterococcus faecalis | Enterococcus hirae | Staphylococcus aureus | Candida albicans |
|---|---|---|---|---|
| Styrene | +++ | – | +++ | +++ |
| SiO2 | +++ | – | +++ | +++ |
| Zn | + | – | +++ | +++ |
| Ti | ++ | – | + | – |
| Cu | + | – | + | + |
| Zn-Ti | – | – | – | – |
| Zn-Cu | + | – | – | – |
| Ti-TiO | ++ | – | ++ | – |
Adhesion ability: (+++ high) (++ medium) (+ little) (– no)
Analysis of SEM micrographs indicated that adhesion ability of E. faecalis strain was inhibited in the case of: Zn-Ti and Zn- Cu microelectrodes. E. faecalis was resistant to the titanium electrode, and only the presence of zinc electrode resulted in an inhibition of cell adhesion (Zn-Ti). The E. hirae strain did not undergo the adhesion neither to the control plates coated with styrene and silicon oxide (SiO2) nor to the polystyrene plates coated with electrodes.
C. albicans showed a small adhesive property in experiments containing zinc, copper and titanium electrodes. Complete inhibition was achieved in samples with mixed systems of electrodes (Zn-Cu, Zn-Ti, Ti-TiO) (Figure 4).
Complete adhesion inhibition of tested pathogenic strains cells was usually reported for samples, in which the plates were coated with two different metallic electrodes [25]. The best antiadhesive properties have been shown by systems containing zinc. Combination of Z n and Ti microelectrodes caused a complete inhibition of adhesion of all strains.
Discussion
This study revealed that S. aureus strain was the most sensitive to the electrodes, but sensitivity to heavy metal ions may be a strain feature. Coque et al. [4] reports that Staphylococcus bacteria belong to the group with the largest number of plasmids encoding resistance to antibiotics and heavy metals. In turn, E. hirae was the most resistant to the electrodes. It had a significant sensitivity to the presence of electrodes only in systems composed of two metals. The results are consistent with the available data, since Yasin et al. [25] isolated strains resistant to Al, Li, Ba, Cr, Fe, Ag, Ni, Zn, Sn.
On the other hand, Coque et al. isolated strains belonging to the family of Enterococcus resistant to zinc and copper. Resistance to heavy metals at gram-positive bacteria is typically associated with antibiotics resistance so-called cross resistance [4].
Larger zones of S. aureus growth inhibition were observed in the presence of mixed electrodes Ti-Cu, Zn-Ti, Zn-Cu, and Zn- Ag, which is associated with the strengthening the action of one metal by the other. This may be due to the phenomenon of the sacrificial electrode formation, which is more electrochemically reactive metal. This electrode is subject to corrosion, while corrosion of the other electrode is slower or does not occur at all. This phenomenon is utilized to produce noble metal ions on a coating covering the surface (the second electrode must then be composed of less active metal, e.g. Ag-Au). Presence of the second electrode causes that the coating has a greater antimicrobial activity than using only one kind of metal ions [5]. In this case, in the systems Ti-Zn, Ag-Zn and Cu-Zn, zinc electrode underwent faster corrosion, which increased the concentration of zinc ions in the medium and small strengthening the zone of inhibition for S. aureus. In the system Mg-Cu, magnesium ions were secreted into the medium, which have been harmless for tested strains and did not result in a clear growth inhibition. Similarly, in the case of Ti-Cu system, in which titanium was more electro-active metal.
In studies conducted on Petri dishes, C. albicans yeast showed a complete resistance against the used electrodes. This may indicate the mechanism of metal ions disposal by means of their reduction and accumulation on the cell membranes [10]. Similar correlation was found by Ronsani et al. [19] in their studies, the use of heavy metal ions, such as nickel or cadmium, increased the biomass. The authors explained such a good relationship by very good accumulation of heavy metals by Candida yeast [19]. Dark and bright tones in cultures of C. albicans with copper, zinc and silver electrodes indicated changes in metabolism of yeast cells.
The presence of heavy metal ions in a solution causes a change in the microorganism’s transcriptome and thus metabolic changes [3,11]. In study by Lee et al. [9] ZnO nanoparticles addition to the medium resulted in the expression of additional 34 genes and repression of 9 ones at Pseudomonas aeruginosa. Changes related to the operon induced by the concentration of zinc cations, which indicates that the increase in their concentration in medium affected the induction of genes regulated by that operon [9]. Dark coloring in the case of copper-containing electrodes may be associated with increased levels of Cu2+ ions in the medium. These ions are necessary for the production of cell-bound melanin by microorganisms [17-21] explain that resistance to copper in gram-positive bacteria may be related to extracellular melanin and its production. Presence of cellbound melanin was also found at C. albicans yeast [13]. These compounds, except from causing darkening of the cells, are involved in redox reactions within the environment in which the cell is present. This indicates that the presence of melanin causes the precipitation of metals in a reduced form on the cell surface [1,2]. Most of the yeast, including C. albicans, is able to sequester metal ions on the cell surface, which may be the cause of their resistance [6,14].
If in the tested samples, in which there is a system of two electrodes, one of them is a sacrificial anode, the reason of dark rings disappearance in systems Zn-Cu and Mg-Cu can be explained. Both zinc and magnesium are higher than copper in electro-activity tabled, which means that they play a function of a protector in this system. The result is that when immersed in the electrolyte, they are subject to accelerated corrosion than when alone. Electrolyte contained high concentration of zinc and magnesium ions, while low of copper. Hence the color reduction in system with zinc and complete reduction in color in system with magnesium (magnesium is the most electro-active metal of these three elements).
Studies upon the adhesive ability confirms the idea that the presence of the second electrode enhances the antimicrobial effect, but only for systems of other elements with zinc a complete inhibition of adhesion was achieved.
Conclusion
For the S. aureus strain, there were determined the largest inhibition zones for the tested electrodes; it was also characterized by the lowest adhesion capacity in the presence of microelectrodes. This shows that this strain is the most sensitive to the synergistic effect of metal ions and the electromagnetic field from the electrodes.
The tested strains of Enterococcus genus, particularly E. hirae, were resistant to the single zinc electrode.
The ability of selected electrode to inhibit the growth of bacteria and reduce their adhesion to the surface depended on the metal that built the electrode and the strain. In the case of E. faecalis and E. hirae strains, electrodes composed of copper operated most effectively, while the growth of S. aureus was inhibited by zinc-containing electrodes.
Combination of two electrodes enhanced the bactericidal effect, but only for some systems.
Tested metal systems do not inhibit the growth of C. albicans, while causing unspecified metabolic changes leading to the appearance of pigmentation. Despite the absence of the fungicidal activity, most of the electrodes effectively inhibited adhesion of C. albicans to the surface.
References
- Apte M, Girme G, Bankar A, Kumar AR, Zinjarde S, et al. (2013) 3, 4-dihydroxy-L-phenylalanine-derived melanin from Yarrowialipolytica mediates the synthesis of silver and gold nanostructures. J of Nanobiotech 11: 1-2.
- Apte M, Girme G, Nair R, Bankar A, Kumar AR, et al. (2013) Melanin mediated synthesis of gold nanoparticles by Yarrowialipolytica. Mater Lett 95: 149-152.
- Chudobova D, Dostalova S, Ruttkay-Nedecky B, Guran R, Miguel AM, et al. (2015) The effect of metal ions on Staphylococcus aureus revealed by biochemical and mass spectrometric analyses.Microbiol Res 170: 147-156.
- Coque TM, Baquero F, Martínez JL, Tedim AP, Lanza VF, et al. (2015) The Plasmidome of Firmicutes: Impact on the emergence and the spread of resistance to antimicrobials.MicrobiolSpectr3:2.
- Dowling DP, Betts AJ, Pope C, McConnell ML(2003) Anti-bacterial silver coatings exhibiting enhanced activity through the addition of platinum. Surface and Coatings Technology, pp: 637-640.
- Harrison JJ, Rabiei M, Turner RJ(2006) Metal resistance in Candida biofilms. FEMS MicrobiolEcol 55: 479-491.
- Inhan-Garip A, Aksu B, Akan Z, Akakin D, Ozaydin AN, et al. (2011) Effect of extremely low frequency electromagnetic fields on growth rate and morphology of bacteria. Intern J of Radiation Biol 87: 1155-1161.
- Köller M, Sengstock Ch, Motemani Y, Khare Ch, Buenconsejo PJS, et al. (2015) Antibacterial activity of microstructured Ag/Au sacrificial anode thin films. Materials Science and Engineering46: 276-280.
- Lee JH, Kim YG, Cho MH, Lee J(2014) ZnO nanoparticles inhibit Pseudomonasaeruginosa biofilm formation and virulence factor production.Microbiol Res169: 888-896.
- Lemire JA, Harrison JJ, Turner RJ(2013) Antimicrobial activity of metals: mechanisms, molecular targets and applications. NatRev Microbiol 11:371-384.
- Lim Ch, Kent H, Karl A, Penesyan A, Loper JE, et al. (2013) The effect of zinc limitation on the transcriptome of Pseudomonas protegens Pfâ€ÂÂ5. EnvironMicrobiol 15.
- Lopez-Serrano D, Solano F, Sanchez-Amat A (2007) Involvement of a novel copper chaperone in tyrosinase activity and melanin synthesis Marinomonasmediterranea.Microbiol 153: 7.
- Morris-Jones R, Gomez BL, Diez S, Uran M, Morris-Jones SD, et al. (2005) Synthesis of melanin pigment by Candida albicansin vitroand during Infection.InfectiImmun, p: 73.
- Noble SM(2013) Candida albicans specializations for iron homeostasis: From commensalism to virulence.CurrOpin in Microbiol 16: 708-715.
- Potenza L, Saltarelli R, Polidori E, Ceccaroli P, Amicucci A, et al. (2012) Effect of 300 mT static and 50 Hz 0.1 mT extremely low frequency magnetic fields on Tuber borchii mycelium. Can JMicrobiol 58: 1174-1182.
- Fard RMN, Heuzenroeder MW, Barton MD (2011) Antimicrobial and heavy metal resistance in commensal enterococci isolated from pigs. Vet Microbiol148: 276-282.
- Rensing Ch, Grass G (2003) Escherichia coli mechanisms of copper homeostasis in a changing environment. FEMS Microbio Rev27.
- Rizzello L, Cingolani R, Pompa PP (2013) Nanotechnology tools for antibacterial materials.Nanomedicine 8: 807-821.
- Ronsani MM, Rymovicz AUM, Meira TM, Grégio AMT, Filho OG, et al. (2011) Virulence modulation of Candida albicans biofilms by metal ions commonly released from orthodontic devices.MicrobPathog 51: 421-425.
- Rousseau C, Baraud F, Leleyter L, Gil O (2009)Cathodic protection by zinc sacrificial anodes: Impact on marine sediment metallic contamination.JHazard Mater167: 953-958.
- Solioz M, Abicht HK, Mermod M, Mancini S (2010) Response of gram-positive bacteria to copper stress. Chemistry 15: 3-14.
- Szabo S, Bakos I(2006) Cathodicprotection with sacrificial anodes. Corrosion Reviews24.
- Tessaro LWE, Murugan NJ, Persinger MA (2015) Bacterial growth rates are influenced by cellular characteristics of individual species when immersed in electromagnetic fields.Microbiol Res 172: 26-33.
- Wang J, Wang Z, Guo S, Zhang J, Song Y, et al. (2011) Antibacterial and anti-adhesive zeolite coatings on titanium alloy surface.MicroporousMesoporous Mater 146: 216-222.
- Yasin A, Bulent I, Sema T (2012) Biochemical and molecular characterization of lead-resistant environmental isolates of Enterococcus faecalis. N Biotechnol 29: S193.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences