Does the Future Need Sutures? - Sutureless Aortic Valve Replacement: State of-the-Art
Igor Belluschi*
Department of Cardiac Surgery, San Raffaele University Hospital, Milan, Italy
- *Corresponding Author:
- Belluschi I
Department of Cardiac Surgery
San Raffaele University Hospital
Via Olgettina 60, Milan, 20132, Italy
Tel: +39-02-2643-5315
Fax: +39-02-2643-7125
E-mail: igor.belluschi@gmail.com
Received date: August 15, 2016; Accepted date: August 16, 2017; Published date: August 25, 2017
Citation: Belluschi I (2017) Does the future need sutures? - Sutureless aortic valve replacement: state-of-the-art. J Heart Health Cir 1: e103.
Copyright: © 2017 Belluschi I. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aortic valve stenosis is characterized by the narrowing of the aortic valve opening. From many years, the gold standard treatment is the surgical aortic valve replacement with biological or mechanical prosthesis. The sutureless approach has been introduced as an alternative to conventional surgery to reduce the cardiopulmonary bypass (CPB) time and enhance the minimally invasive technique. The aim of this editorial is to show the state-of-the-art of the sutureless technology, describing the benefits and the open issues that will enhance or delay the development of a surgery without sutures.
Keywords
Sutreless; Aortic; Valve; Perceval
Abbreviations
AS: Aortic Stenosis; AVR: Aortic Valve Replacement; CPB: Cardiopulmonary Bypass; EOA: Effective Orifice Area; LV: Left ventricle; LVOT: Left Ventricle Outflow Tract; PM: Pacemaker; PPM: Patient-Prosthesis Mismatch; PVL: Paravalvular Leak; RBBB: right Bundle Branch Block; RCT: randomized clinical trial; SU-AVR: Sutureless Aortic Valve Replacement; TAVI: Transcatheter Aortic Valve Implantation; THV: Transcatheter Heart Valves; ViV: Valve-in-Valve
Editorial
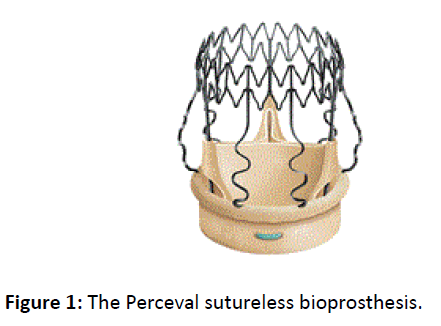
Aortic valve stenosis (AS) is characterized by the narrowing of the aortic valve opening and a consequent reduction in the anterograde blood flow generated by the left ventricle (LV) systole. It is the most frequent treated heart valve disease and it occurs in 2-7% of the population over 65 years old. The incidence increases with age. The etiology shows calcific degeneration as the most important cause in the elderly, while congenital defects (i.e. unicuspid, bicuspid and quadricuspid) and rheumatic disease, nowadays especially in the developing countries, are prevalent among the young. From many years, the gold standard treatment of AS is the surgical aortic valve replacement (AVR) with biological or mechanical prosthesis, depending on the age of the patients and their indications. High risk old patients benefit from the innovative transcatheter aortic valve implantation (TAVI) procedure. There are two types of cardiac bioprosthesis: stented or stent less valves, depending on the presence of a stent to support the leaflets, by the way they all need to be sutured to the native aortic anulus. The idea of a sutureless prosthesis was first introduced by Magovern and Cromie in 1962: the aim was to make the implantation easier and to reduce the surgical risk [1]. In 2011, the innovative sutureless bioprosthesis Perceval (Sorin S.p.A., Saluggia, Italy) was launched on the market (Figure 1).
Perceval is a surgical sutureless self-expanding valve without a sewing ring, which has been introduced as an alternative to conventional surgery to reduce the cardiopulmonary bypass (CPB) time and enhance the minimally invasive approach [2]. In the same year it obtained the CE mark after five years of three European trials (the Perceval Pilot trial, the Perceval Pivotal trial and the CAVALIER trial). Nowadays, it has been implanted in more than 15.000 patients in over 350 hospitals worldwide.
According to the literature, Perceval valve has been widely used even in off-label indications such as in concomitant mitral valve replacement [3], endocarditis [4], bicuspid aortic valve [5] and aortic regurgitation [6].
However, the advantages of the sutureless device go far beyond the reduction of the CPB time. In vitro analysis and echocardiographic follow-up is demonstrating Perceval’s great effective orifice areas (EOAs) compared to conventional sutured bioprosthesis. Its hemodynamic performance is brilliant, showing how it could be considered a good idea to reduce the risk of patient-prosthesis mismatch (PPM), which still remains an independent predictor of mortality and cardiac events. In fact, procedures like aortic root enlargement, homografts and stent less bioprosthesis are technically complex, require longer learning period and are associated with protracted aortic cross-clamp time. Therefore Perceval could become a possible solution for PPM avoidance [7].
In the past years, a few cases of Perceval degeneration have been reported [8]. Considering Perceval’s large EOA and the reduced PPM when compared to sutured valves, some Authors preferred a less invasive Valve-in-Valve (ViV) approach instead of a reoperation. The group of Fujita et al. first described an algorithm to assess the use of a Transcatheter Heart Valve (THV) into a degenerated sutureless prosthesis and then performed the insertion of a Sapien XT 23 mm in a Perceval M size [9]. Nowadays, Perceval’s 9 years follow-up, its excellent area and performance in small annuli, together with the optimal ViV feasibility could even change the strategy of the prosthesis selection in the younger population.
Three published propensity score matching showed Perceval’s good results, especially regarding pacemaker need, mortality and paravalular leaks (PVL), if compared to conventional sutured prosthesis or THVs for TAVI [10,11]. Anyway, large multicenter RCTs are still necessary to fully understand the sutureless outcome, particularly the “gray zone” patients at intermediate surgical risk.
According to the literature, pacemaker (PM) implantation rate after SU-AVR with Perceval ranges from 3,1% to 28,5% [12,13], however the largest European multicenter study series (Pilot, Pivotal, CAVALIER trials) of 731 patients showed a rate of 7,4% [2]. The exact mechanism of atrioventricular conduction disorders in SU-AVRs with Perceval remains to be clearly elucidated, even if some hypothesis concern the ballon post-dilation and the nitinol stent, similar to those of THV. Despite the surgeons’ learning curve could be an explanation of the slightly higher PM need, these results are comparable to the conventional AVRs with sutured bioprosthesis [14]. Preoperative conduction disorders could also play a role on this outcome [15].
Durability is still another open issue. Even if the manufacturer recommends this bioprosthesis in the young (the cutoff age of 65 years was removed from indications in March 2014), the published studies have a maximum of 10 years follow-up. However, a great expectation is due to the longer follow-up (almost 25 years) of the Sorin Pericarbon Freedom valve, whose biological structure represents the starting point for Perceval own biological component [16].
In conclusion, SU-AVR with the Perceval valve is rapidly developing worldwide, showing good results compared to conventional sutured bioprosthesis. However some issues remains still open, but ongoing and future RCTs will play a pivotal role to fully understand the outcome of this bioprosthesis. While a gradual but constantly higher group of intermediate-low risk patients will be destined to transcatheter procedures, the implantation of an aortic valve without suture will maybe replace the conventional sutured AVR and could represent the future standard of aortic valve surgery.
References
- Magovern GJ, Cromie HW (1963) Sutureless prosthetic heart valves. J Thorac Cardiovasc Surg 46: 726–736.
- Shrestha M, Fischlein T, Meuris B, Flameng W, Carrel T, et al. (2015) European multicentre experience with the sutureless Perceval valve: clinical and haemodynamic outcomes up to 5 years in over 700 patients. Eur J Cardiothorac Surg 1–8.
- Moriggia S, Trumello C, Buzzatti N, Iaci G, Di Giannuario, et al. (2015) Aortic sutureless Perceval valve for small root in concomitant mitral valve replacement. J Heart Valve Dis 24: 187-189.
- Lio A, Miceli A, Solinas M, Glauber M (2015) Initial Experience with Sutureless Sorin Perceval S Aortic Prosthesis for the Treatment of Prosthetic Valve Endocarditis. J Thorac Cardiovasc Surg 63: 501-503.
- Nguyen A, Fortin W, Mazine A, Bocuhard D, Carrier M, et al. (2015) Sutureless aortic valve replacement in patients who have bicuspid aortic valve. J Thorac Cardiovasc Surg 150: 851-857.
- Gilmanov DS, Solinas M, Kallushi E, Gasbarri T, Bianchi G, et al. (2015) Sutureless Aortic Valve Replacement for Aortic Incompetence. J Card Surg 30: 391-395.
- Belluschi I, Moriggia S, Giacomini A, Del Forno B, Di Sanzo S, et al. (2017) Can Perceval sutureless valve reduce the rate of patient-prosthesis mismatch? Eur J Cardiothorac Surg 51: 1093-1099.
- Di Eusanio M, Murana G, Cefarelli M, Mazzola A, Di Bartolomeo R (2014) In the era of the valve-in-valve: is TAVI in sutureless valves feasible? Ann Cardiothorac Surg 4: 214–217.
- Fujita B, Kutting M, Scholts S, Utzenrath M, Hakim-Meibodi K, et al. (2015) Development of an algorithm to plan and simulate a new interventional procedure. Interact CardioVasc Thorac Surg 21: 87-95.
- D’Onofrio A, Messina A, Lorusso R, Alfieri O, Fusari M, et al. (2012) Sutureless aortic valve replacement as an alternative treatment for patients belonging to the “gray zone” between transcatheter aortic valve implantation and conventional surgery: a propensitymatched, multicenter analysis. J Thorac Cardiovasc Surg 144: 1010-1016.
- Santarpino G, Pfeiffer S, Jessl J, Dell’Aquila AM, Pollari F, et al. (2014) Sutureless replacement versus transcatheter valve implantation in aortic valve stenosis: a propensity-matched analysis of 2 strategies in high-risk patients. H Thorac Cardiovasc Surg 147: 561-567.
- Flameng W, Herregods MC, Hermans H, Van der Mieren G, Vercalsteren M, et al. (2011) Effect of sutureless implantation of the Perceval S aortic valve bioprosthesis on intraoperative and early postoperative outcomes. J Thorac Cardiovasc Surg 142: 1453-1457.
- Konig KC, Wahlers T, Scherner M, Wippermann J (2014) Sutureless Perceval aortic valve in comparison with the stented Carpentier-Edwards Perimount aortic valve. J Heart Valve Dis 23: 253-258.
- Huynh H, Dalloui G, Ghanbari H, Burke P, David M, et al. (2009) Permanent pacemaker implantation following aortic valve replacement: current prevalence and clinical predictors. Pacing Clin Electrophysiol 32: 1520-1525.
- Belluschi I, Moriggia S, Calabrese M, Schiavi D, Alfieri O (2016) Early Predictors of Permanent Pacemaker Implantation in Sutureless Aortic Valve Replacement with Perceval Bioprosthesis: A Conduction Disorders Analysis. J Heart Health and Circ 1:3.
- Seguin JR, Grandmougin D, Folliguet T, Warembourg H, Laborde F, et al. (1998) Long-term results with the Sorin Pericarbon valve in the aortic position: a multicenter study. J Heart Valve Dis 7: 278-282.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences