ISSN : 0976 - 8688
Der Pharmacia Sinica
In-vitro Antiurolithiatic and Antioxidant Activity of Couroupita guianensis Aubl Leaves
Himanshu N. Panchal1, Sapna D. Desai1*, Maitri K. Soni1, Priyanka Mishra2 and Dhananjay B. Meshram2
1Department of Pharmacology, Pioneer Pharmacy Degree College, Sayajipura, Vadodara, Gujarat – 390019, India
2Department of Quality Assurance, Pioneer Pharmacy Degree College, Sayajipura, Vadodara – 390019, India
Abstract
Urolithiasis is a chronic disease that has affected human kind from last so many centuries. In this disease pain gets precipitated due to the calcium oxalate stone in kidney or in any part of urinary tract. Perfect causes for urolithiasis are not known, but age, sex, diet, life style, medical history, family history, etc are responsible for it. From the ancient time people have been using plants and animal sources as a medicine. There are so many plants which are having an antiurolithiatic activity. This study proves that the aqueous extract of Couroupita guianensis Aubl has a capability to dissolve calcium oxalate crystal in in vitro conditions when it is compared with cystone as a standard. The antiurolithiatic activity of Couroupita guianensis Aubl leaves is probably mediated through the inhibition of Calcium oxalate crystallization. In addition to its free radical scavenging and antioxidant activities, it acts as an excellent agent for the prevention of urolithiasis.
Keywords
Couroupita guianensis Aubl, antiurolithiatic, cystone, calcium oxalate, hydroxyl radical scavenging.
Introduction
Herbal drugs are prescribed widely because of their effectiveness, less side effects, and relatively low cost. Couroupita guianensis Aubl. (family: Lecythidaceae), commonly known as cannon ball tree is grown in Indian gardens as an ornamental tree for its beautiful flowers (Figure 1) [1,2].
Couroupita guianensis Aubl was first described by Fusse Aulbet. There are diverse pharmacological activities like antipyretic, antidepressant, antiseptic, analgesic, anti-inflammatory, antiprotozoal, anticancer, antiulcer, antimicrobial, wound healing, antidiarrheal, antioxidant and antifertility activities of different parts of this tree [3,4]. The detailed botanical study and morphology is shown in Tables 1 and 2 respectively [5]. Table 3 depicts the common names of Couroupita guianensis Aubl.
| Botanical Study | |
|---|---|
| Kingdom | Plantae |
| Sub kingdom | Tracheobionta |
| Division | Magnoliophyta |
| Class | Magnoliopsida |
| Order | Lecythidales |
| Family | Lecythidaceae |
| Genus | Couroupita |
| Species | Couroupita guianensis Aubl |
Table 1: Botanical classification of Couroupita guianensis
| Part of Plant | Description |
|---|---|
| Leaves | Simple, alternate, narrowly elliptic to 10 inches long and 4 inches wide. |
| Flowers | 6 petals, orange-red with white central disk held on racemes. |
| Fruits | Capsules, globose, brown, 4-8 inches in diameter, contains up to 300 seeds. Weight of mature fruit is approximately 1450 gm. |
Table 2: Description of Couroupita guianensis
| Common Names | |
|---|---|
| English | Cannon ball tree |
| Hindi | Shivaling, Ayahuma, kailashpati |
| Tamil | Nagalinga pushpam |
| Spanish | Coco de mono; Abrico de Macaco |
Table 3: Common names of Couroupita guianensis
Urolithiasis is the stone formation in urinary system. In this condition, urinary calculi are formed and gets located anywhere in the urinary tract [6]. The common symptoms are sudden pain, vomiting, nausea. Location of pain depends on the location of stone. There are some risk factors for the causation of kidney stone like epidemiological factors and genetic predisposition like dietary risk factors, climate, occupation, family history of stones etc. Under normal biological process aerobic cells generate Reactive Oxygen Species (ROS), oxygen-derived free radicals, superoxide anion (O.-2), hydrogen peroxide (H2O2), peroxynitrite (ONOO.), hydroxyl radicals OH. and peroxyl radical (ROO.). Under pathological conditions, the rate of formation of these partially reduced oxygen species speeds up, resulting in reduced antioxidant defenses of the renal cells, leading to oxidative renal cell injury. Renal epithelial cells upon exposure to various crystals like calcium oxalate, calcium phosphate, and uric acid produce ROS, leading to kidney stone formation by creating an imbalance between the promoters and inhibitors of lithogenesis.
Therefore, drugs with multiple targets, such as antioxidant, anti-inflammatory and antispasmodic properties are an overt choice for antiurolithiatic drug development [7-9].
Materials and Methods
Collection of plant materials: Leaves of plant Couroupita guianensis were collected from the Kamati Baug Garden, Vadodara, Gujarat, India. It was ensured that plant was healthy and disease free.
Preparation of extract: Leaves were washed to remove dust and other undesirable particles. Then leaves were dried under shade and crushed. 20 gm of fine powder of leaves was extracted with 100 ml of water, methanol, and chloroform at room temperature in conical flask for 48 hours. The results of preliminary phytochemical investigation are shown in Table 4.
| S. No. | Test | Water | Methanol | Chloroform |
|---|---|---|---|---|
| 1 | Test for Alkaloid | |||
| 1) Mayer’s reagent | + | - | - | |
| 2) Dragondorff’s reagent | - | - | - | |
| 3) Wagner’s reagent | + | + | + | |
| 4) Hager’s reagent | - | + | + | |
| 2 | Test for Glycoside | |||
| 1) Keller-killiani test | - | + | - | |
| 2) Borntrager’s test | - | - | - | |
| 3) Ledal’s test | - | - | - | |
| 3 | Test for Phenolic compounds | |||
| 1) Ferric chloride test | - | - | - | |
| 2) Lead acetate test | + | + | + | |
| 3) Gelatin test | - | - | - | |
| 4 | Test for Carbohydrates | |||
| 1) Molisch test | - | - | + | |
| 2) Fehling’s test | - | - | - | |
| 5 | Test for Flavonoids | |||
| 1) Ammonia test | - | - | - | |
| 2) Shinoda/paw test | - | - | - | |
| 6 | Test for Proteins and free amino acids | |||
| 1) Millon’s test | - | - | - | |
| 2) Xethoprotein test | + | - | - | |
| 3) Biuret test | - | - | + | |
| 7 | Test for Saponins | |||
| 1) The alcoholic extract was evaporated to dryness, residue was extracted with petroleum ether and acetone. To the insoluble residue after extraction, 5 ml of water was added and shaken well. | - | - | - | |
| 2) To the alcoholic extract, 3 drops of sodium bicarbonate was added and shaken well. | - | - | + | |
| 8 | Test for Sterols | |||
| 1) Salkowski reaction | + | - | - | |
| 2) Hersche’s Son’s reaction | - | - | - | |
| 9 | Test for Acidic compounds | |||
| 1) To the 2 ml of alcoholic extract, 1 ml NaHCO3 solution was added | - | - | - | |
| 2) 2 ml of alcoholic extract was taken in warm water and filtered. The filtrate was then tested with litmus paper and methyl orange. | - | - | - |
Table 4: Phytochemical analysis of leaf extract of Couroupita guianensis Aubl [9]
In Vitro Antioxidant Assay
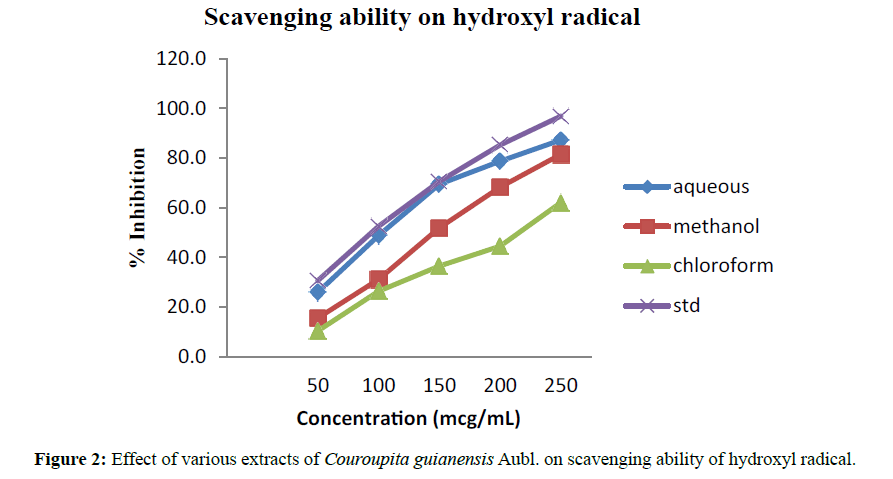
Scavenging ability on hydroxyl radicals: Hydroxyl scavenger ability was performed by generating the OH radicals from FeSO4 and hydrogen peroxide. The radical scavenging activity of Couroupita guianensis (aqueous, methanolic and chloroform extracts) was determined by their ability to hydroxylate salicylate. The reaction mixture (3 ml) contained 1 ml FeSO4 (1.5 mM), 0.7 ml hydrogen peroxide (6 mM), 0.3 ml sodium salicylate (20 mM) and varying concentrations of Couroupita guianensis extracts. After incubation for 1 h at 37°C, the absorbance of the hydroxylated salicylate complex was measured at 562 nm. The percentage scavenging effect was calculated as scavenging activity = [1-(A1-A2)/A0] X 100%, Where A0 was the absorbance of the control (without extract) and A1 was the absorbance in the presence of the extract, A2 was the absorbance without sodium salicylate [10].
Evaluation of Antiurolithiatic Activity
1. Preparation of experimental kidney stones (calcium oxalate and calcium phosphate stones) by homogenous precipitation: 1.47 gm of calcium chloride dihydrate was dissolved in 100 ml distilled water and 1.34 gm of sodium oxalate was dissolved in 100 ml of 2N H2SO4. Both were mixed equally in a beaker to precipitate out calcium oxalate with stirring. Equimolar solution of calcium chloride dihydrate (AR) in distilled water and disodium hydrogen phosphate (AR) in 10 ml of (2N H2SO4), was allowed to react in sufficient quantity of distilled water in a beaker. The resulting precipitates were of calcium phosphate. Both precipitates were freed from traces of H2SO4 by ammonia solution. The precipitates were washed with distilled water and dried at 60 °C for 4 hours [11,12].
2. Preparation of semipermeable membrane from farm eggs: The semipermeable membrane of eggs lies in between the outer calcified shell and the inner contents like albumin and yolk. Shell was removed chemically by placing the eggs in 2M HCL overnight, which caused complete decalcification. Further, it was washed with distilled water, and carefully with a sharp pointer a hole was made on the top and the contents were squeezed out completely from the decalcified egg. Then egg membrane was washed thoroughly with distilled water, and was placed in ammonia solution, in the moistened condition for a while and rinsed with distilled water. The membrane was stored in refrigerator at a pH of 7.0-7.4 [13,14].
3. Estimation of calcium oxalate by Titrimetry: The dissolution percentage of calcium oxalate was evaluated by taking exactly 2 mg of calcium oxalate and 10, 20, 30, 40 mg of the dried extract powder. These were packed together in semipermeable membrane of egg. This was allowed to suspend in a conical flask containing 100 ml of 0.1M tris buffer. First group served as blank containing only1 mg of calcium oxalate. The second group served as positive control containing 1 mg of calcium oxalate and along with the 20, 30, 40, 50 mg of standard drug, i.e. Cystone. The 3rd and 4th groups along with 1 mg of calcium oxalate containing 20, 30, 40, 50 mg aqueous, 20, 30, 40 mg methanolic, and 20, 30, 40, 50 mg chloroform dried extract powder. The conical flasks of all groups were kept in an incubator preheated to 37°C for 24 h. The contents of semipermeable membranes were removed from each group into separate test tubes, 2 ml of 1N sulphuric acid was added to each test tube, mixture was warmed at 80-90 °C and titrated with 0.9494 N KMnO4 till a light pink color end point was obtained. The amount of remaining undissolved calcium oxalate was subtracted from the total quantity used in the experiment in the beginning to know the total quantity of dissolved calcium oxalate by various solvent extracts. Each ml of 0.9494 N KMnO4 is equivalents to 0.1898 mg of calcium oxalate. The obtained results were further calculated by putting in calculation factors to get mg of calcium oxalate remaining [11,15]. The equations are as given below.
Each ml of 0.9494 N KMnO4 is equivalent to 0.1898 mg calcium oxalate.
So x ml of 0.9494 N KMnO4 is equivalent to how much mg of calcium oxalate.
That calculated amount of calcium oxalate were then converted in to the % of calcium oxalate.
Results and Discussion
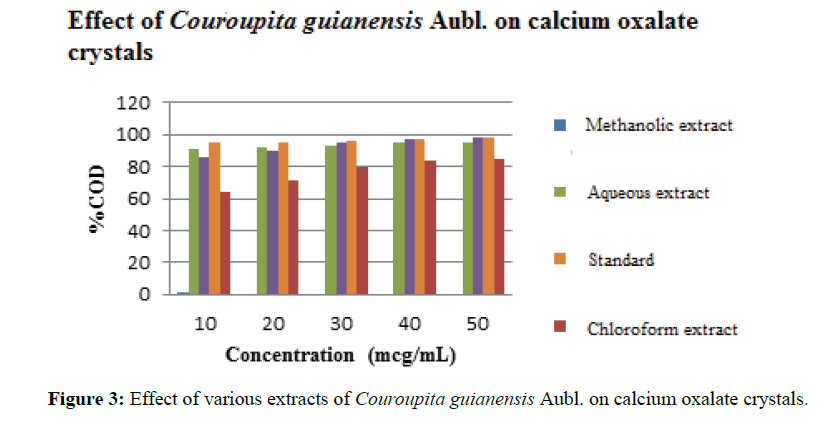
Phytochemical screening of aqueous, chloroform, and methanol extracts of Couroupita guianensis indicated the presence of alkaloids, glycosides, amino acids or protein, saponins and sterols. The results of phytochemical screening are shown in Table 4. The graphical representation of percentage inhibition in hydroxyl radical scavenging activity is depicted in Figure 2. The aqueous extract shows the highest inhibitory activity with an IC50 of 127.22 mg/mL. The order of activity for the extract were standard (cystone) (32.34 mg/mL)> Aqueous (127.22 mg/mL)> Methanolic extract (150.6 mg/mL)> Chloroform extract (208.33 mg/mL) of Couroupita guianensis. The observed results of the percent calcium oxalate dissolved by various extracts of Couroupita guianensis are shown in Table 5 and the results are depicted in Figure 3. The results showed that aqueous extract showed a good radical scavenging activity and a good antiurolithiatic activity.
| S. No. | Group | % Calcium Oxalate Dissolved |
|---|---|---|
| 1 | Blank | 0 |
| 2 | Aqueous extract | 40.8 ± 0.032 |
| 3 | Chloroform extract | 32.5 ± 0.025 |
| 4 | Methanol extract | 38.6 ± 0.035 |
| 5 | Standard (cystone) | 58 ± 0.028 |
Table 5: Summary of % COD by various extracts of Couroupita guianensis
Conclusion
The good radical scavenging activity showed that abundant antioxidant activity prevents the cells from causing injury. The rate of formation of partially reduced oxygen species is increased in urolithiasis and/or the antioxidant defences of the cells are weakened, eventually leading to oxidative cell injury. The in vitro antiurolithiatic activity of different extracts of Couroupita guianensis was compared with cystone as a standard. Different extracts were found to show the antiurolithiatic activity, but the aqueous extract showed the significant % dissolution of calcium oxalate formed due to stone forming minerals when compared with the standard cystone. This study has proved that the aqueous extract of leaves of Couroupita guianensis have a good antioxidant activity that proved them to be a successful lithotropic agent.
Acknowledgement
The authors are thankful to the management of Om Gayatri Education and Charitable Trust for providing the required resources for carrying out the research project and the Principal, Pioneer Pharmacy Degree College, Vadodara, India, for providing the necessary facilities to carry out the work.
References
- Ramalakshmi, C., Ranjitsingh, A.J.A., Kalirajan, K., et al. Elixir Appl Bio, 2013. 57: 14055-14057.
- Chavda, V.P., IIJS, 2015. 2: 6-9.
- Chandolu, S.K., Gaddala, N., Vanamala, S., et al. IRJPAS, 2011. 1: 105-108.
- Sundararajan, R., Koduru, R., Pharmacophore, 2014. 5: 147-159.
- Gousia, S.K., Kumar, A., Kumar, V., Inter J Pharmacy and Pharmaceutical Science Research, 2013. 3: 140-143.
- Rajeshwari, P., Rajeswari, G., Jabbirulla, S.K., Vardhan, V., IJPPS, 2013. 5: 599-601.
- Selvam, R., Urol Res, 2002. 30: 35-47.
- Devkar, R.A., Chaudhary, S., Adepu, S., et al. Pharm Biol, 2016. 54: 1237-1245.
- Khandelwal, K.R., Sethi, V., Nirali Prakashan, 24th Edition, 2015. 25: 1-2.
- Sasikumar, V., Menon, S.G., IJPBS, 2011. 2: B92-B103.
- Saso, L., Valentini, G., Leone, M.G., et al. Urol Int, 1998. 30: 14-17.
- Phatak, R.S., Hendre, A.S., IJPPR, 2015. 7: 275-279
- Byahatti, V., Vasantakumar, P., ASL, 2010. 30: 14-17.
- Saravanasingh, K., Ramamurthy, M., Parthiban, P., Int J Curr Res Med Sci, 2016. 2: 24-27.
- Mendham, J., Denney, R.C., Barnes, J.D., et al. Vogel’s textbook of qualitative chemical analysis, 2000. 6th edition: p. 806.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences