Keywords
Gills histology; Tilapia; Oreochromis spilurus; Phenanthrene; PAH
Introduction
Aquatic organisms and wildlife are subjected to pollutant compounds and may cause intensive disruption in the ecosystem it may cause a decrease in biodiversity and biota in general [1]. Polycyclic aromatic hydrocarbons (PAHs) are a widespread class of environmental pollutants known to exert acutely toxic effects [2]. Phenanthrene (PHE), a three-ring PAH, is among USEPA priority PAHs because of its presence in the environment especially in aquatic ecosystems, this can be due to petrogenic and pyrogenic sources and it has been shown to exert toxicity in several marine species [3]. Bay and K region are found in PHE and they are classified as the smallest tricyclic PAH, the Bay and K regions react actively with other chemical compounds to form the main carcinogenic species [4]. PHE are used as a model substrate to carry out research on metabolism of carcinogenic PAHs. Recently, PAHs have got keen attention of researchers due to some compounds being highly carcinogenic or mutagenic [5]. These compounds can be accumulated in Fish and other aquatic organisms, basically from the aquatic environment through gills and through the process of taking inn food from the benthic sediment of the aquatic environment [6]. It is well known that, Fishes are suitable indicators of contaminants accumulation, any pollutant research on aquatic toxicity due to their high sensitivity to environment [7]. Any minimal changes may cause physiological and biochemical alteration in organ, tissues or pathways associated to the respective toxicant. In case of soluble petroleum hydrocarbons fishes are assumed to be the best bio accumulator [8].
Tilapia fish is endemic to Africa with the generic name of Tilapia is belonging to the group cichlids. This group is consisting of three main genera including Oreochromis, Sarotherodon and Tilapia. All the species of tilapia are nest builders and after fertilization, the eggs are secured in the nest by the parents. Immediately, the fertilized eggs are picked up by the parents in the mouth for incubation and hold for some days till hatching [9]. Tilapia are shaped much like sunfish or crappie but can be easily identified by an interrupted lateral line characteristic of the Cichlid family of fishes. They are laterally compressed and deep-bodied with long dorsal fins. The forward portion of the dorsal fin is heavily spined. Spines are also found in the pelvis and anal fins. There are usually wide vertical bars down the sides of fry, fingerlings and sometimes adults. Tilapias are African fish and Egyptian strains of tilapia were found the best in regard to growth rates compared to other African tilapias when introduced to many countries [10].
Tilapia fishes evolved from marine ancestors even though they are mostly freshwater water organisms. This may form or explain the basis why most of these fishes are able to withstand a wide range of water salinity. Their growth and reproduction can be achieved in brackish water. Sometimes few species carry out their reproduction and other physiological processes in a water bodies with high salinity. Although, few studies have been carried out on marine culture tiapia, relative to the large information available on their culture in freshwater habitat. Less freshwater ecosystems in different part of the world and competition for such natural resources has led to increased pressure to develop aquaculture in brackish water and sea water. Hence, the first aquatic organism that one may consider for aquaculture in brackish water and seawater is tilapia [11].
Different processes are carried out by the fish using the gills, these are gas exchange, ammonia excretion, osmoregulation and acid-base balance, but most importantly, contact of gills with pollutants can alter gills function and anatomy [12]. Resultant changes can include epithelial damage, necrosis and, the collapse of lamellae [13]. The functional results of these variations in function and structure may affect aquaculture productivity, for example, gaseous exchange, impairment of osmoregulation, reduced growth which if left unaddressed can result in the mortality of stock [14].
The nature and degree of toxicity depends on many factors, including the chemical form, the pH and temperature of the water, the length of exposure and the life stage of the fish being exposed [15]. Moreover, the long-term exposure to high doses of toxic can also have a considerable impact on fish health and physiological function with subsequent decreases in or suppression of growth, hemoglobin concentration and haematocrit values [16].
The investigation of the toxicity, histological changes potential of PAHs in marine organisms became very useful tool of risk pollution assessment as well as the environmental conditions evaluation [17]. Limited work has been done in the past on marine cultured tilapia fish (Oreochromis spilurus), exposed to phenanthrene [5]. Hence, the investigation of this study has been focused on the changes in gills structure and mucus cell distribution of marine cultured tilapia (Oreochromis spilurus) at larvae stage, were induced to different concentration of phenanthrene. On this study will reveal for the first time the possibility of Phenanthrene to penetrate histological system of marine tilapia.
Materials and Methods
Experimental fish and exposure conditions
MarineTilapia fish (Oreochromis spilurus) larvae were fed phenanthrene spiked feed for 14 days in the month of November, 2018. Oreochromis spilurus larvae were obtained from Jeddah Fisheries Research Centre (JFRC) which is located at 21°48'22.9"N, 39°02'07.7"E in the north of Jeddah. The fish was transported to the laboratory and maintained into glass aquariums (70 cm x 40 cm x 51 cm) filled with 16.8 L of aerated sea water. For each treatment, 20 larvae of marine tilapia were kept in aquarium. Water parameters such as temperature, pH and salinity were measured, using the methods of APHA (1995), the values of which were 27 ± 1ºC, 7.1 ± 0.5 and Salinity, 41±0.5%, respectively [18]. The water in each of the experimental aquarium were not changed during the 14 day trial, but only aerated. The fishes were acclimatized to the laboratory condition for two weeks before use for the experiments [19].
Test solutions and food preparations
Phenanthrene crystals (99.5% pure, Sigma) were dissolved in absolute ethanol first, then stock solution (1 mg mL-1) was prepared and stored at -4ºC in amber bottle. For making the test solutions of desired concentrations, the stock solution was further weakened with distilled water accordingly. For the preparation of phenanthrene-spiked food, the stock solution was diluted to preferred concentrations and mixed with food of the fish (TetraMin, Melle, Germany). The test dose concentrations were 0 mg L-1 (Control) and 100 mg L-1. The mixed food was stored in lined wall tubes with Teflon in order to avoid sticking of phenanthrene to the tube wall. Then, the tubes were caped and kept on roller at -4oC for 24 h to ensure mixing of food with phenanthrene properly. The tubes were opened occasionally for removing the ethanol vapors. After 24 h of mixing, the food was centrifuged, eliminated the water and then freezed [20]. Extra care was taken as phenanthrene solutions and spike food was light sensitive and hence, it was kept in the dark [21]. For treatment tilapia with phenanthrene, Spike-food mixed with different concentrations of phenanthrene (0 and 100 mg L-1) was fed to fish twice a day for 14 days [20].
Gills histology
To assess changes to gills histopathology, two fish were randomly selected from each aquarium and prepared for histology. The gills of control and experimented fish were fixed in 10% neutralbuffered formalin for 12-24 h. The samples were then subjected to common histological assessment (desiccated and rooted in paraffin). Small sections of 5 μm were excised and stained with hematoxylin and eosin [22,23].
Results
All samples were observed using the light microscope under (10x, 40x and 100x) magnification of objective lens and photographs of histological structures were captured. As a result, gills samples from tilapia fed with feed that is not treated with Phenanthrene, control group (0 mg L-1), showed that there were no changes from normal gills histology such as normal structure of lamellae and filaments, Primary and secondary lamellae, apperance of pillar cell, Normal Epithelium (NE) and Erythrocytes (E) in the lamellae (Figures 1-3).
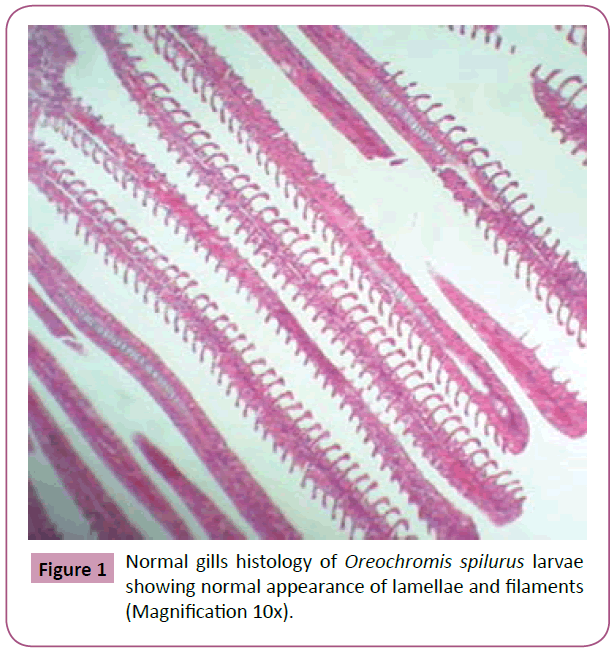
Figure 1: Normal gills histology of Oreochromis spilurus larvae showing normal appearance of lamellae and filaments (Magnification 10x).
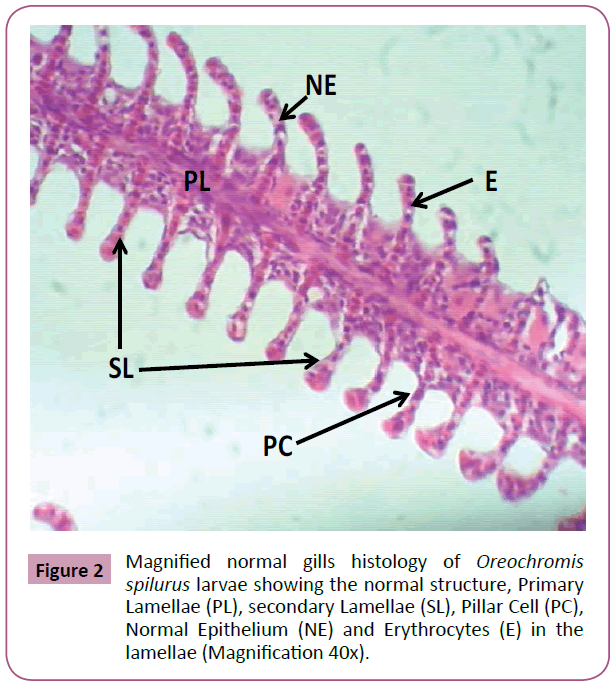
Figure 2: Magnified normal gills histology of Oreochromis spilurus larvae showing the normal structure, Primary Lamellae (PL), secondary Lamellae (SL), Pillar Cell (PC), Normal Epithelium (NE) and Erythrocytes (E) in the lamellae (Magnification 40x).
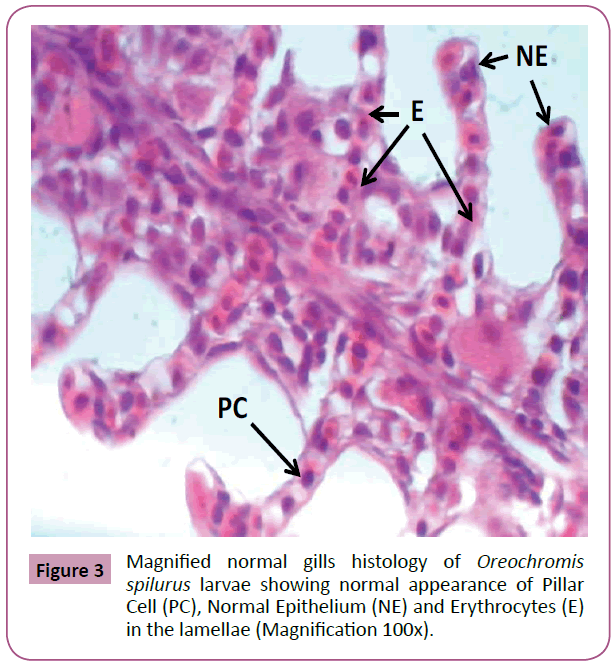
Figure 3: Magnified normal gills histology of Oreochromis spilurus larvae showing normal appearance of Pillar Cell (PC), Normal Epithelium (NE) and Erythrocytes (E) in the lamellae (Magnification 100x).
Thereafter, gills samples from fish fed with feed treated with 100 mg L-1 phenanthrene, showed a series of level of expression in epithelial hyperplasia. Changes including, small proliferations of epithelial cells, epithelial lifting and lamellar deformations as characterized by hyperplasia of the mucous and chloride cells of epithelial cells, leading to complete lamellar fusions, with rupture of blood vessel forming hematoma, fusion of the secondary lamellae and presence of goblet cells and hyperemia. Hypertrophy (hydropic degeneration) was also a frequent alteration observed in the treated fish (Figures 4-6).
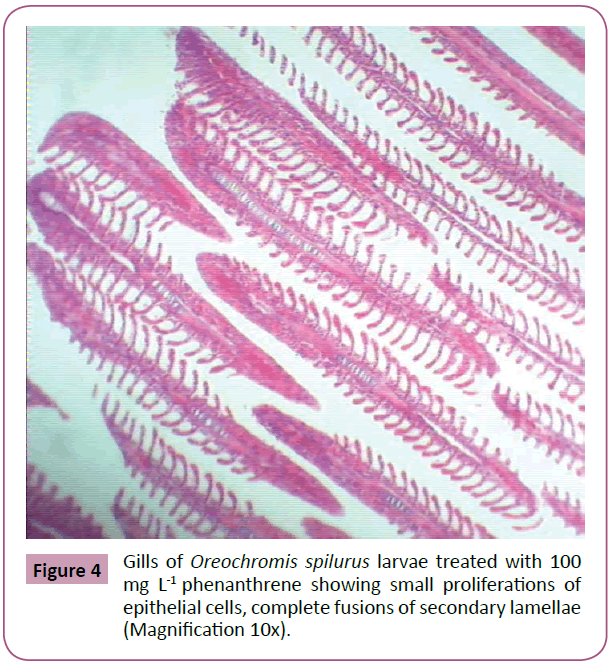
Figure 4: Gills of Oreochromis spilurus larvae treated with 100 mg L-1 phenanthrene showing small proliferations of epithelial cells, complete fusions of secondary lamellae (Magnification 10x).
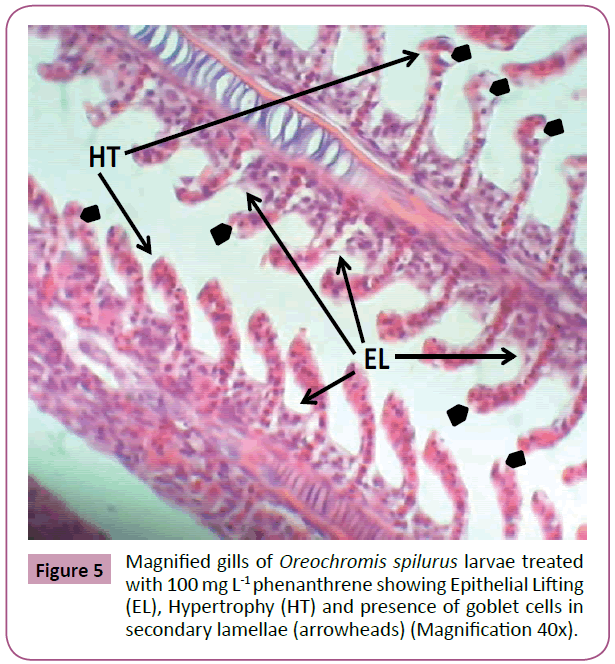
Figure 5: Magnified gills of Oreochromis spilurus larvae treated with 100 mg L-1 phenanthrene showing Epithelial Lifting (EL), Hypertrophy (HT) and presence of goblet cells in secondary lamellae (arrowheads) (Magnification 40x).
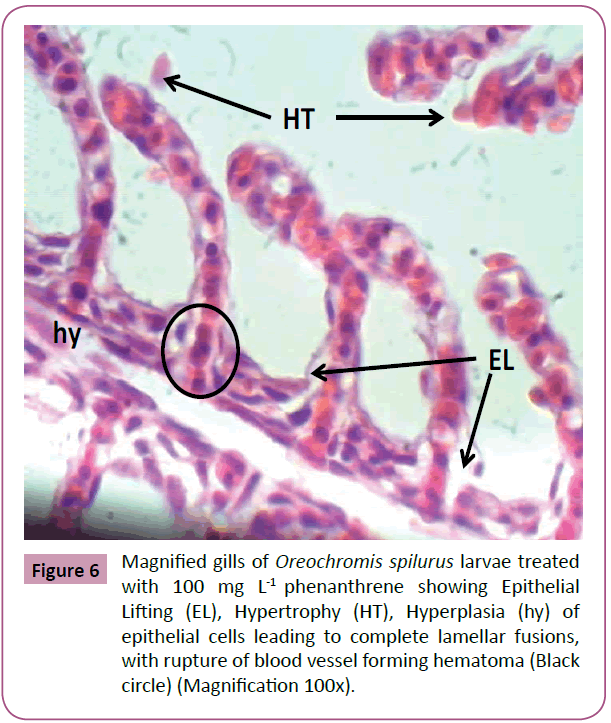
Figure 6: Magnified gills of Oreochromis spilurus larvae treated with 100 mg L-1 phenanthrene showing Epithelial Lifting (EL), Hypertrophy (HT), Hyperplasia (hy) of epithelial cells leading to complete lamellar fusions, with rupture of blood vessel forming hematoma (Black circle) (Magnification 100x).
Upon the observation under the light microscope, the changes in fish gills become more severe with obvious effect on higher level of phenanthrene. The change in cell shape, size and structure can easily be observed. Besides some noticeable deviations to the structure of the gills tissues, i.e., lifting of the lamellar epithelium were observed compared to the control. Ultimately, the impairment was more severe when the fish was subjected to highest concentration.
Discussion
In aquatic organisms, for example fish, gills are one of the most sensitive organs which gets damaged easily once the fish is exposed to broad spectrum of pollutants, even at minimal concentration levels [24]. The various function of the gills such gaseous exchange (respiration), excretion and osmoregulation, as a result of large surface area in contact with the external aquatic environment makes the gills particularly sensitive to changes in environmental chemical and physical status [25,26].
In the present study, the effect of gradual accumulation of phenanthrene in fish tissue (i.e., gills) was evaluated for 0 mg L-1 and 100 mg L-1 as control group and treated group respectively. For this research, larvae of marine tilapia fish (Oreochromis spilurus) were selected due to their wide range of adaptability in nature and higher survival in poor quality water [27].
Conclusively, the results revealed the different and obvious changes in gills structure in the group of tilapia treated with 100 mg L-1 phenanthrene. Changes such as necrosis and edemas of branchial lamellae and was confirmed that the phenanthrene has bioaccumulated in the fish. These results are in line with many previous findings that were of the same suggestions for the effect of different pollutants on fish gills [25,28]. In another study, Cerqueira and Fernandes, pointed out that as the gills are the principal site for gaseous exchange, ionic and osmotic regulation and acid-base balance, then histopathological changes to these structures will result in respiratory disturbance and electrolyte imbalance. In many studies, fish gills have been reported to react non-specifically to pollutants, which means different exposure to different pollutants can lead to similar reactions and changes in anatomical structure and physiology of gills tissue and therefore be regarded as evidence of response to stress in general [29].
Conclusion
Our investigation indicates that obvious changes were observed in gills cells of the marine cultured tilapia (Oreochromis spilurus) at larvae stage across the different concentration of Phenanthrene. From the observation under the light microscope, the changes in fish gills become more severe with clear effect on high level of phenanthrene dose. Our finding revealed that, the effect of phenanthrene can be attributed to changes in cell shape, size and structure of fish gills and confirms the possibility of phenanthrene as a pollutant with degenerative effect on cells of tilapia and possibly other organisms.
Significance Statement
This study discovered the effect of an important member of polycyclic aromatic hydrocarbon (Phenanthrene) on an aquatic organism using marine cultured tilapia fish (Oreochromis spilurus) as a model organism. To the best of our knowledge, this is the first study performed to investigate the changes in gills structure of marine cultured tilapia (Oreochromis spilurus) larvae exposed to different concentration of phenanthrene. This study will help the researchers to uncover the critical areas of marine pollutants and toxicology using Oreochromis spilurus as a model organism that many researchers are yet to explore. Thus, a new theory on phenanthrene toxicity in marine environment using Oreochromis spilurus as model organism may be arrived at.
Acknowledgments
The authors are grateful to King Abdulaziz University (KAU), Umm Al-Qura University (UQU) and Jeddah Fisheries Research Center (JFRC), Saudi Arabia for encouragement and support. Moreover, we extend our cordial thanks to King Abdulaziz City for Science and Technology (KACST) for financial support: Grant (NO. 1-18- 01-009-0182) to complete this work successfully.
References
- Srogi K (2007) Monitoring of environmental exposure to polycyclic aromatic hydrocarbons: A review. Environ Chem Lett 5: 169-195.
- Budavari S, O’Neil MJ, Smith A (1989) The merck index. Merck, USA. Pp: 1143-1144.
- Albers PH (2003) Petroleum and individual polycyclic aromatic hydrocarbons. In: Hoffman DJ, Rattner BA, Burton GA, Cairns J (Eds), Handbook of Ecotoxicology. Lewis Publishers. Pp: 1-32.
- Correia AD, Gonçalves R, Scholze M, Ferreira M, Reis-Henriques MA (2007) Biochemical and behavioural responses in gilthead sea bream (Sparus aurata) to phenanthrene. J Exp Mar Biol Ecol 347: 109–122.
- Pathiratne A, Hemachandra CK (2010) Assessment of bile fluorescence patterns in a tropical fish, Nile Tilapia (Oreochromis niloticus) exposed to naphthalene, phenanthrene, pyrene and chrysene using fixed wavelength fluorescence and synchronous fluorescence spectrometry. Bull Environ Contam Toxicol 84: 554-558.
- USEPA (1988) Drinking water criteria document for polycyclic aromatic hydrocarbons (PAHs): Prepared by the environmental criteria and assessment office, office of health and environmental assessment. US Environmental Protection Agency, Cincinnati, USA.
- Kundu D, Mondal S, Dutta D, Haque S, Ghosh A (2016) Accumulation and contamination of lead in different trophic levels of food chain in sewage-fed East Kolkata Wetland, West Bengal, India. Int J Environ Sci Technol 2: 61-68.
- Rodrigues RV, Miranda-Filho KC, Gusmao EP, Moreira CB, Romano LA, et al. (2010) Deleterious effects of water-soluble fraction of petroleum, diesel and gasoline on marine pejerrey Odontesthes argentinensis larvae. Sci Total Environ 408: 2054-2059.
- Thomas P, Michael M (1999) Tilapia life history and biology. Southern Regional Aquaculture Centre (SRAC). Publication No.283.
- Tave D, Jayaprakas V, Smitherman R (1990) Effects of intraspecific hybridization in Tilapia nilotica on survival under ambient winter temperature in Alabama. J World Aquac Soc 21: 201-204.
- Kirk RG (1972) A review of the recent development in tilapia culture with special reference to fish farming in the heated effluents of power stations. Aquac 1: 45-60.
- Weihrauch D, Wilkie MP, Walsh PJ (2009) Ammonia and urea transporters in gills of fish and aquatic crustaceans. J Exp Biol 212: 1716-1730.
- Cardoso EL, Chiarini-Garcia H, Ferreira RA, Poli CR (1996) Morphological changes in the gills of Lophiosilurus alexandri exposed to un-ionized ammonia. J Fish Bio 49: 775-787.
- Zhao JH, Lam TJ, Guo JY (1997) Acute toxicity of ammonia to the early stage-larvae and juveniles of Eriocheir sinensis H. Milne-Edwards, 1853 (Decapoda: Grapsidae) reared in the laboratory. Aquac Res 28: 517-525.
- Rebelo MF, Rodriguez EM, Santos EA, Ansaldo M (2000) Histopathological changes in gills of the estuarine crab Chasmagnathus granulata (Crustacea-Decapoda) following acute exposure to ammonia. Comp Biochem Physiol 125: 157-164.
- Voslarova E, Pistekova V, Svobodova Z, Bedanova I (2008) Nitrite toxicity to Danio rerio: Effects of sub chronic exposure on fish growth. Acta Vet Brno 77: 455-460.
- Osman AG, Abuel-Fadl KY, Kloas W (2012) In situ evaluation of the genotoxic potential of the river Nile: II. Detection of DNA strand-breakage and apoptosis in Oreochromis niloticus niloticus (Linnaeus, 1758) and Clarias gariepinus (Burchell, 1822). Mutat Res 747: 14-21.
- APHA (1995) Standard methods for the examination of water and waste-water. 19th edition, American Public Health Association, Washington, DC.
- El-Sayed MY, Abdel-Wahab AA, Nasser AA, Hossam E, Mohamed M (2013) Histological changes in the liver and intestine of Nile Tilapia (Oreochromis niloticus), exposed to sublethal concentrations of cadmium. Pakistan J Zool 5: 833-841.
- Ching-Yi H, Hui-Chen L, Wenjau L (2010) Reproductive toxicology study of phenanthrene in medaka (Oryzias latipes). Arch Environ Contam Toxicol 58:131-139.
- Velario F, Bottino P, Ugolini D, Cimberle MR, Tozzi GA, et al. (1984) Chemical and photochemical degradation of polyaromatic hydrocarbons in the atmosphere. Sci Total Environ 40: 169-188.
- Luna GL (1968) Manual of histopathological staining methods of the armed force institute of pathology. 3rd edition, McGraw-Hill Co., New York, USA.
- Bernet D, Schmidt H, Meier W, Burkhardt-holm P, Wahli T (1999) Histopathology in fish: Proposal for a protocol to assess aquatic pollution. J Fish Dis 22: 25-34.
- Karlsson L (1983) Gills morphology in the zebra fish, Brachydanio rerio (Hamilton-Buchanan). J Fish Bio 23: 511-524.
- Mallatt J (1985) Fish gills structural changes induced by toxicants and other irritants: A statistical review. Can J Fish Aquat Sci 42: 630-648.
- Mazon AF, Monteiro EAS, Pinheiro GHD, Fernandes MN (2002) Hematological and physiological changes induced by short-term exposure to copper in the freshwater fish, Prochilodus scrofa. Braz J Bio 62: 621-631.
- Cerqueira CC, Fernandes MN (2002) Gills tissue recovery after copper exposure and blood parameter responses in the tropical fish Prochilodus scrofa. Ecotoxicol Environ Saf 52: 83-91.
- Redner BD, Stickney RR (1979) Acclimation to ammonia by Tilapia aurea. Trans Am Fish Soc 108: 383-388.
- Speare DJ, Ferguson HW (2006) Systemic pathology of fish: A text and atlas of normal tissues in teleosts and their responses in diseases. In: Ferguson HW editor. Chapter 2 Gills and Pseudobranchs. Scotian Press, London. Pp: 25-63.
- Koca YB, Koca S, Yildiz S, Gürcü B, Osanc E, et al. (2005) Investigation of histopathological and cytogenetic effects on Lepomis gibbosus (Pisces: Perciformes) in the cine stream (Aydin/Turkey) with determination of water pollution. Environ Toxicol 20: 560-571.