Keywords
Essential oil, Ocimum gratissimum, Antifungal activity
Introduction
The severity of infectious skin diseases is often overlooked in Africa by public health organizations; they tend to be ignored because they are not fatal [1]. Among these infectious skin diseases, fungal infections now occupy a very prominent place, their frequency and severity have increased steadily in recent years [2]. These are mainly caused by fungi of the dermatophyte and yeast types [3]. These fungi induce clinical signs which result, in fact, in a symptomatological procession which is most often painful (pruritus, burns, bacterial superinfection) and represent a source of permanent discomfort [4]. The high prevalence of superficial mycotic infections shows that 20 to 25% of the world population suffers from skin fungus [5]. In Africa, dermatophytes are responsible for 52.5% of dermatophytosis, Trichophyton rubrum involved at 44.2% and Trichophyton mentagrophytes 21.2% [6]. Candidiasis is caused by yeasts of the genus Candida, 54.3% of which incriminate the albicans species [7]. The treatment of fungal infections includes oral and topical formulations derived mainly from two families of antifungal drugs: Azoles and allylamines. However, most of the active molecules currently used have very unpleasant side effects for the patient in addition to the high cost of these. In addition to these limitations, there is the emergence of resistance in some strains to available drugs. Essential oils could be an alternative. Essential oils are complex mixtures of aromatic or odourless volatile compounds obtained respectively by distillation or maceration of the plant material [8]. They have many pharmacological properties [9]. Indeed, many research studies report the cytotoxic properties of Essential Oils (EO) towards dermatophytes and yeasts. Moreover, on an often empirical basis, the aromatherapy community has successfully used essential oils for decades to treat dermatological infections [10]. It is therefore essential to develop formulations to make the most of the antimicrobial properties of essential oils of African flora plants. Numerous physicochemical toxicological and pharmacological studies have been conducted on Ocimum gratissimum [11-14]. It is an aromatic plant of the lamiaceae family, belonging to the genus Ocimum.
Our work aims to develop an antifungal cream based on Ocimum gratissimum essential oil and to evaluate its activity on a few strains of fungi.
Materials and Methods
The plant material used was Ocimum gratissimum (lamiaceae) leaves (Figure 1). The harvest was made in the village of Ebimpé-Ivoiry Coast. The identification was made at the National Floristic Centre of Ivoiry Coast under code UCJ00888879.
Figure 1. The Ocimum gratissimum plant harvested in Ebimpte- Ivoiry Coast.
The fungal strains used were provided by the Institut Pasteur of Ivoiry Coast, yeast strains (Candida albicans N° 18-855) and dermatophyte strains (Triphophyton mentagrophytes, N° 19-108).
The animal material, albino rabbits weighing between 1500 g-1800 g, was provided by the pharmacology laboratory of the Pharmaceutical and Biological Sciences Teaching and Research Unit (SPB UFR) of the Université Félix Houphouet Boigny in Abidjan-Côte d'Ivoire. They were used for the skin tolerance test.
For the formulation, several raw materials of analytical grade were used, shea butter (local origin), tween 60 (Sigma®), stearic acid (Prolabo®), cetyl alcohol (Prolabo®), sodium alginate (Carl Roth®), Guar gum (Carl Roth®), carrageenan (Carl Roth®), and sodium benzoate (Prolabo®).
Essential Oil Extraction
The extraction method used was hydrodistillation [15]. The previously dried, ground with a GM 300 grinder knife Grindomix mixer and weighed plant material (100 g) was immersed in 500 ml of water inside a flask. The whole thing was then brought to a boil for about 1 hour and 10 minutes on a heated cap. At the end, the essential oil was collected by decanting and frozen, then weighed and stored in an amber bottle at about 12°C away from light. The extraction yield was calculated based on the weight of the dried plant material before extraction.
Physico-chemical Analysis of EO
The determination of the chemical composition of EO was performed by Gas Phase Chromatography coupled with Mass Spectrometry. The GPC/MS-MS coupling combines the high separating power of the GPC with the high analytical power of the MS [16]. The CPG was performed on a Varian CP 3380 chromatograph, equipped with an apolar column characterized by a stationary methyl silicone phase (DB5, 50 m × 0.22 mm, film thickness 0.25 μm). A 10% essential oil solution was prepared in pentane, and 0.5 μL was introduced at the column head where it was sprayed at the SPLIT injector. The detector was flame ionization. The operating conditions were as follows: injector and detector temperature (200°C), furnace temperature from 50°C to 200°C with a gradient of 5°C/min and carrier gas, nitrogen with a flow rate of 1 ml/min. The recorder was Star Chromatography Workstation software. The quantitative analysis was done by integrating the measurements of peak areas and the results expressed as relative percentages.
The CPG/MS-MS coupling device used was a HEWLETTPACKARD type, type 5970, equipped with a fused silica capillary column with a stationary DB1 grafted phase. The mass detector was a quadrupole type and the ionization energy used was 70 eV. Handling conditions were as follows: temperature programming 70-200° at 10°C/min, injection temperature 220°C, carrier gas flow rate (helium) at 0.6 mL/min and injection volume of 0.1 μL of an essential oil solution diluted 10% in hexane.
Identification was made by comparing the mass spectra and retentions indices obtained with those in the literature.
Formulation and Preparation of the Cream
The preparation method was direct emulsification [17]. After weighing the various raw materials and classifying them into hydrophilic excipients (osmosed water and tween 60) and lipophilic excipients (stearic acid shea butter, cetyl alcohol), they were introduced into two different beakers and subjected to a temperature of 60° in a water bath. Under agitation (1800 rpm) using a marine propeller mixer, and at a temperature of 60°, the emulsification was carried out by reversing the oily phase in the aqueous phase. At the end of the addition of the oil phase, the preparation was removed from the water bath and left to shake at 25°C to allow for gradual cooling for 10 minutes.
Physico-chemical Tests of the Preparation
The organoleptic characteristics of the colour, odour and appearance of the different formulae were noted. The direction of the emulsion was determined by the dye method. To the emulsion, a drop of Sudan red dye (lipophilic dye) was added. The propagation or not of this dye made it possible to determine the direction of the emulsion. The particle size analysis was consisted in depositing a drop of the emulsion on a slide and covered by a lamella, then the observation was made with objective 10 of the computer-assisted optical microscope. The pH determination was performed three times, by dipping the pH meter electrode into the preparations previously diluted to 1/10th. The rheological study was carried out using a flat cone rheometer. At fixed temperatures the shear rate was varied. The measurement parameters were as follows: Geometry of 2° PL61ST and PU20SR3642SS and PL61STS8488SS, Gap: 1000 nm, Force: 0.1 N, minimum shear rate: 0.01 s-1, maximum shear rate: 100s-1, and temperatures at 20°C, 25°C and 37°C.
Centrifuge and Heat Stability Tests
The centrifugation stability test consisted of introducing 2 g of each emulsion into test tubes and subjecting them to three successive centrifugations of 05 minutes each (2000, 3000, 5000 rpm). For temperature stability, 2 g of each emulsion was introduced into test tubes and subjected to a temperature of 40°C in an oven for 30 minutes.
Evaluation of the Acute Irritant and Corrosive Effect on the Skin and Eyes
The agreement for the tests was obtained from the Inter- UFR Ethics Committee of the Félix Houphouët Boigny University under number 2019-45. The test was performed on 3 albino rabbits. The rabbits were put in separate pens for a week to feed and acclimatize them. Then they were marked and weighed. Subsequently, the dorsal region of the trunk was shaved with a lawnmower while taking care not to injure them. The test substance (0.5 g of our cream 10% EO) was applied to a small area (about 6 cm2) of the skin, eyes and covered with a gauze pad attached with a non-irritating adhesive tape. Signs of erythema, edema, conjunctival and corneal irritation were observed in all animals and reactions were rated after 60 minutes and then 24, 48 and 72 hours after patch removal. Skin and eye reactions were rated and recorded according to the scale in the table in OECD Guidelines 404 and 405.
Microbiological Controls
In order to evaluate the microbial load of our preparation, a microbiological control was carried out. For this purpose, a series of two dilutions were made. Two techniques were used for seeding, namely deep or mass seeding for the enumeration of Mesophilic Aerobic Germs (MAG) and thermotolerant coliforms; and surface seeding for the detection of E. coli, Staphylococcus aureus, yeasts and moulds and pseudomonas.
In Vitro Evaluation of Antifungal Activity
On yeasts of the genus Candida albicans
The incorporation of the substances (EO, Cream 10% EO) into the Sabouraud-chloramphenicol medium was done using the double dilution method in inclined tubes [18]. For EO, a series of 12 test tubes were prepared with a decreasing concentration range from 20 mg/ml to 36.06 μg/ml according to a geometric link of reason ½ The same operation was repeated with the cream 10% with concentrations ranging from 400 mg/ml to 20 mg/ml. Antifungal parameters (CMI/ CMF) were determined as well as inhibition diameters by the solid media diffusion method.
On Trichophyton mentagrophyte strains
The evaluation of antifungal activity on T. mentagrophytes was performed using the agar incorporation technique according to Grover et al. and included 1% EO and cream 10% EO [19]. This technique consists of monitoring the mycelial growth of the microbial culture discs, seeded in the center of the Petri dish containing the culture medium supplemented with the substance to be evaluated at different concentrations. The medium used is SDA (Sabouraud Dextrose Agar), made selective to fungi by the introduction of an antibiotic, chloramphenicol. The concentrations tested ranged from 20 mg/ml to 3.06 μg/ml. The inhibition percentage was calculated according to the formula:
Percentage of inhibition (%)=(Dt - De)/Dt × 100
Dt (mm) = diameter of fungal growth in the control box.
De (mm) = diameter of the fungal growth in the test box.
The IMC and CMF were evaluated as before on yeasts.
In Vivo Evaluation of Antifungal Activity
It is under the same authorization number 2019-45 of the Inter-UFR Ethics Committee of the Félix Houphouët Boigny University that this evaluation was carried out. The selected rabbits were weighed and then grouped by weight. The hairs were shaved to a surface area of at least 4 cm2 on the right side. The skin has been irritated by some scarifications with vaccinostyle. On this irritated skin, for each batch a fungal paste comprising a recent culture of T. mentagrophytes and Candida albicans mixed with 2 ml of saline solution was applied using a wooden tongue depressor. After the plating, a bandage with compresses and perforated plaster was applied. Five days later, the dressing was removed to detect the infestation, which manifested itself as severe erythema with the formation of light pressure ulcers invading the skin. The preparations and the reference antifungal agent were applied to the lesions of each respective rabbit. Treatment was repeated twice daily until the observed signs disappeared definitely. The results were expressed as the average healing time, i.e. the time required for the lesions to completely disappear.
Results
The essential oil was extracted from the dry leaves with a yield of 0, 92 ± 0, 14. It had light yollow color, an apparence of oily liquid, a characteristic odour and a relative density of 0,88 at 22°C. Its chemical composition is dominated by thymol (41.31%) and p-cymene, (22.26%) followed by -terpinene (9.25%).
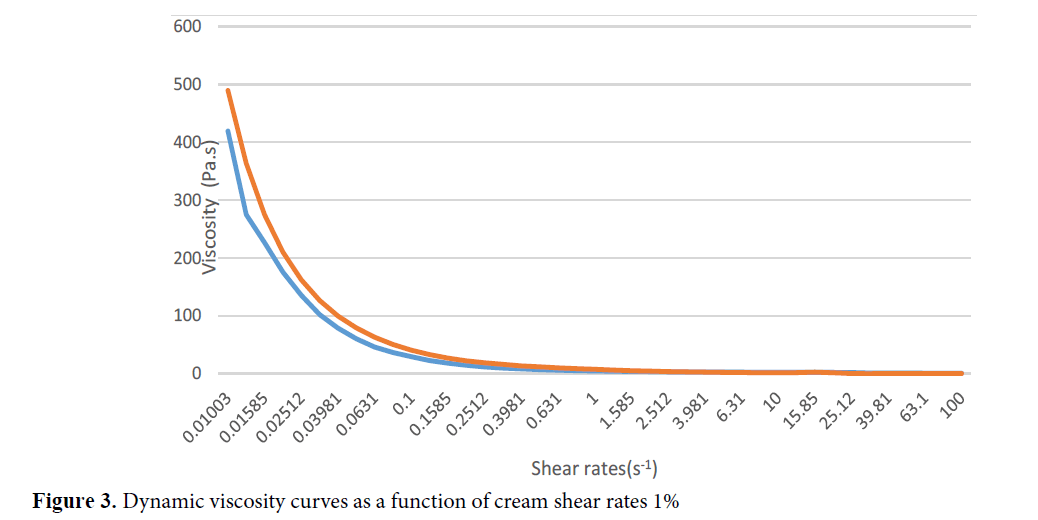
The various basic cream formulae made are presented in Table 1. The essential oil has been added to the most stable formula (Table 2). The cream formulated with Ocimum gratissimum EO was an L/H emulsion because it did not homogenize with Sudanese red [17]. Shea butter has been used as a major component (10%) of the fat phase. The final formula of the cream at 10% EO was white in colour, soft in consistency, homogeneous in appearance and with a pH of 5.4. (Figure 2). It exhibited rheofluidifying and thixotropic behaviour at temperatures of 20°C, 25°C, and 37°C for viscosity values between 100 and 500 Pa.s and at shear rates ranging from 0.01 to 100 s-1 (Figure 3).
EmulsionsEmulsion 1Emulsion 2Emulsion 3Emulsion 4SurfactantTween 60 5%Lipophlic phaseShea butter 10%Thickening agentGuar gumCarrageenanSodium alginateCetyl alcohol 5%/ Stearic acid 10%4%1%5%Osmosed waterOsmosed water Qsp 100%
Table 1: Basic cream formulas.
Ocimum gratissimum EO10%Tween 605%Shea butter10%Cetyl alcohol /5%Stearic acid10%Sodium benzoate0.10%Osmosed waterQsp 100%
Table 2: The final formula use.
GermsRabbitsTreatmentsObservations at the end of the treatmentTricophyton mentagrophytesRabbit 1Cream with 10%EOComplete healing after 4 ± 2 daysRabbit 2Econazole creamFull healing after 4 ± 1 daysRabbit 3EO of O.GTotal healing with skin dryness after 4 ± 2 days
Table 3: Results of in vivo evaluation of the efficacy of the preparations on rabbits infested with Tricophyton mentagrophytes.
Figure 2. Cream with 10% Ocimum gratissimum EO.
The formulation complies with the microbiological standards of the european pharmacopeia, 9th edition (absence of pathogens and AMGs < 102 CFU/g sample).
Skin reaction scores after 72 hours of observation revealed no erythema, pressure ulcer or edema formation. The ocular scoring showed no evidence of major irritability of the cornea, iris, conjunctiva and eyelids for which the swelling observed in the early hours disappeared after 72 hours.
Antifungal activity was evaluated on yeasts (Candida albicans) and dermatophytes (Trichopyton mentagrophytes). The study was conducted to determine whether the EO in cream form retains its antifungal activity on the germs studied. The growth of Candida albicans was inhibited at MICs and FCMs equal to 1.25 mg/ml for EO. For cream 10% EO the MIC was 12.5 mg/ml and the FCM was 100 mg/ml. The inhibition diameters were 20 ± 2.1 mm and 10.7 ± 1.2 mm for EO and cream 10% EO respectively. The inhibition diameters of EO were greater than that of Fluconazole (12.3 ± 0.09 mm) but smaller than those of Econazole cream (28.3 ± 3.2 mm and 21.3 ± 2 mm). Moreover, the MIC / MFC reports show that only EO has fungicidal activity on Candida, unlike the cream 10% EO, which despite its very high percentage of EO remains fungistatic.
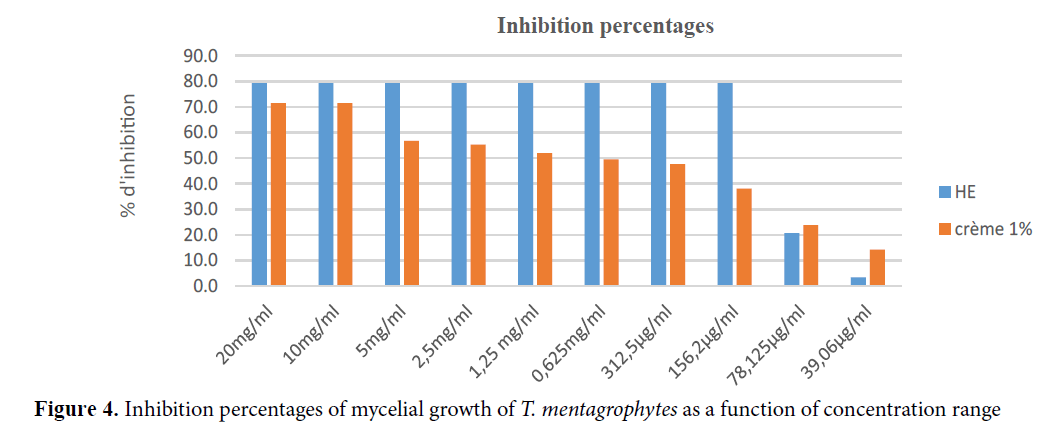
The growth of Trichophyton mentagrophytes was influenced by our products with MICs and FMCs of 156.5 μg/ml for EO and 10 mg/ml for cream 10% EO. The preparations were all fungicidal at a concentration of 0.15 mg/ml, and 10 mg/ml respectively for EO, and cream 10% EO (Figure 4).
Figure 3. Dynamic viscosity curves as a function of cream shear rates 1%.
Figure 4.Inhibition percentages of mycelial growth of T. mentagrophytes as a function of concentration range.
Compared to Candida albicans, EO and cream showed better in vitro activity on Trichophyton mentagrophytes.
The average healing time observed in all rabbits infected with T. mentagrophytes and treated with our different preparations (EO, cream 10% EO and econazole cream) was on average 4 ± 2 days (Table 3). The formulation used (1% cream) proved to be just as effective with a result quite identical to that of the reference antifungal agent (econazole cream). The treatment with EO resulted in complete healing but with dry skin.
Discussion
The value of the extraction yield is consistent with that of Tchoumbougnang et al., who obtained an extraction yield of 0.60% on dry Ocimum gratissimum leaves collected in Cameroon [20]. Dabiré and co-workers, in evaluating the effect of drying on the extraction yield of Grangea maderaspatana EO, showed that the yield increased by 50% after a few days of drying (i.e. 0.15% at D0 and 0.4% after drying) [21, 22]. The increase in extraction yield after drying observed in our work and that of Dabiré and co-workers could be explained by the fact that biosynthesis of some EOs would continue after harvest during the drying time, due to enzymatic phenomena [22]. The chemical composition of our essential oil is in accordance with the work of Tchoumbougnang and teammates in Cameroon and Kouassi et al. in Ivoiry Coast, which described samples characterized by a high level of three compounds (thymol, p-cymene and -terpinene) [18,20]. Although the majority compounds are the same, the percentages remain different and this could be attributed to several factors: location, harvest period, but also climatic factors.
The pH value of the cream of 5.4 corroborated with the work of Siessina and teammates who, in making the shea butter creams, found pH values between 3.69 and 5.84 [17]. These values suggest that the pH of the shea butter preparations obtained is compatible with dermal use since they are close to that of the pH on the skin surface, which is about 5[17]. The rheofluidifying and thixotropic characteristics obtained with the cream obtained are in agreement with the work of Moghimi and co-workers in 2009, who had produced an anti-dermatophytic cream based on Eucalyptus camaldulensis [23]. Indeed, this behaviour is described for non-Newtonian liquids including creams. Viscosity decreases with increasing shear rates and does not return to its initial state quickly enough when the stress is stopped, due to the slow restructuring of the system [17].
The results of the evaluation of in vitro anti-candidotic activity obtained are like those of Kouassi et al. [18]. They had shown that Ocimum gratissimum EO showed an antifungal activity on the in vitro growth of Candida albicans. This activity would be due to the nature of the chemical composition of this oil. The results of the antidermatophytic activity agree with the work of Chaumont and teammates which had demonstrated an anti-dermatophytic activity of EO on T. mentagrophytes at an MIC/MFC of 200 μg/ml [24]. These similar results would be more related to the nature of the chemical composition of EO, which is mainly rich in thymol, in γ-terpinene and para-cymene [25].
The results obtained in vivo showed that the EO, although it has an antifungal activity in vivo as significant as the reference antifungal agent (Econazole cream) and the formulation used (1% cream), causes a very pronounced dryness of the skin (loss of elasticity). The astringent character of essential oils could explain this result. Indeed, EOs, by their healing properties, cause dehydration of the skin, which leads to a loss of elasticity and consequently to skin dryness [26]. This result would further justify the presentation of EO in dosage forms to overcome this problem.
Conclusion
The cream developed from the essential oil of Ocimum gratissimum complies with the criteria of the european pharmacopeia, 9th edition. This formula, whose antifungal activity has been tested, both in vitro and in vivo, has shown that the essential oil of Ocimum gratissimum, when formulated as a cream, retains its antifungal activity.
References
- Colette van H, Naafs B. Common skin diseases in africa, an illustrated guide. 2009.
- Thes P, Zirihi GM, Adou KMK. Action of soap made from oils of Mitracarpus scaber (rubiaceae) «misca», Mareya micrantha (euphorbiaceae) «g243» and Cassia alata (caesalpiniaceae) on in vitro growth of Candida albicans and Trichophyton mentagrophytes. J Sci Pharm Biol. 2006; 7: 28-36
- Silva-Rocha WP, De Azevedo MF, Chaves GM. Epidemiology and fungal species distribution of superficial mycoses in Northeast Brazil. Journal de Mycologie Médicale. 2017; 27: 57-64.
- Koffi A, Dally I, Aka B, et al. Galenic development of a Mitracarpus scaber oil-based ointment for the treatment of five types of superficial mycoses. J Pharm Biol Sci. 2007; 8: 18-24.
- Havlickova B, Czaika VA, Friedrich M. Epidemiological trends in skin mycoses worldwide. Mycoses. 2008; 51: 2-15
- Kouotou EA, Kechia FA, Iwewe YS, et al. Mycological profile of onychomycosis in Yaoundé, Cameroon. J Mycol Med. 2017; 27: 238-44.
- Talarmin JP, Boutoille D, Tattevin P, et al. Candidaemia epidemiology: one-year prospective observational study in the West of France. Med Infect Dis. 2009; 39: 877-85.
- Burt S. Essential oils: their antibacterial properties and potential applications in foods-a review. Int J Food Microbiol. 2004; 94: 223-53.
- Bakkali F, Averbeck S, Averbeck D, et al. Biological effects of essential oils–a review. Food Chem Toxicol. 2008; 46: 446-75.
- Pitman V. Aromatherapy: A pratical approach. 2nd ed. India: Lotus Publishing; 2004.
- Mohr FB, Lermen C, Gazim, ZC, et al. Antifungal activity, yield, and composition of Ocimum gratissimum essential oil. Genet Mol Res. 2017; 16: 1-10.
- Pandey S. Antibacterial and antifungal activities of Ocimum gratissimum L. Proteus. 2017; 25:56.
- Prabhu KS, Lobo R., Shirwaikar AA, et al. Ocimum gratissimum: A review of its chemical, pharmacological and ethnomedicinal properties. Open Complement Med J. 2009; 1: 1-15.
- Hzounda FJ, Jazet DM, Fekam BF, et al. Antioxydant and antifungal activities of the essential oils of Ocimum gratissimum from Yaoundé and Dschang. J Pharm Pharmacol. 2014; 2: 257-68.
- Kaloustian J, Hadji-Minaglou F. Knowledge of essential oils: quality and aromatherapy: Between science and tradition for a reasoned medical application. Springer Science & Business Media. 2013.
- Kouame NM, Kamagate M, Koffi C, et al. Cymbopogon citratus (DC.) Stapf: ethnopharmacology, phytochemistry, pharmacology and toxicology. Phytother Res. 2016; 14: 384-392.
- Siessina Lawaldlia NTM TOÉ. Tests on the formulation of creams and body milks based on shea butter from Burkina Faso. University of Ouagadougou. 2004.
- Kouassi EK, Ouattara S, Seguin C, et al. Study of some biological properties of Ocimum Gratissimum L, a lamiaceae harvested in Daloa (Côte d'Ivoire). Eur Sci J. 2018; 14: 477.
- Grover RK, Moore JD. Toxicometric studies of fungicides against brown rot organisms, Sclerotinia fructicola and s-laxa. Phytopathology. 1962; 52: 876-80.
- Tchoumbougnang F, Dongmo PM, Sameza ML, et al. Larvicide activity on Anopheles gambiae Giles and chemical composition of essential oils extracted from four crops in Cameroon. 2009; 13: 77-84.
- Constantin MD, Eloi P, Roger HCN, et al.Mouhoussine Essential oil of Grangea maderaspatana (L.) poir. of Burkina Faso: extraction yield and chemical composition. J West AfrSoc Chem. 2009; 28:81-86.
- Dabire C, Nebie RHC, Belanger A, et al. Effect of drying of plant material on the chemical composition of essential oil and antioxidant activity of essential oil extracts Ocimum basilicum L. Int J Biol Chem Sci. 2011; 5: 1082-95.
- Moghimipour E, Ameri A, Saudatzadeh A, et al. Formulation of an antidermatophytic cream from hydro-alcoholic extract of Eucalyptus camaldulensis leaves. J Nat Pharm Prod. 2009; 4: 32-40.
- Chaumont JP, Mandin D, Sanda K, et al. In vitro antimicrobial activities of five essential oils of Togolese Lamiaceae with respect to germs representative of the cutaneous microflora. Acta Botanica Gallica. 2001; 148: 93-101.
- Oussou KR, Kanko C, Guessend N, et al. Antibacterial activities of the essential oils of three aromatic plants of Côte-d'Ivoire. Comptes Rendus Chimie. 2004; 7: 1081-6.
- Jouault S. The quality of essential oils and their influence on their effectiveness and toxicity. PhD Thesis. 2012.