Review Article - (2018) Volume 8, Issue 3
Syed Hassan Mehdi*, Sana Nafees, Md Zafaryab, Md Asad Khan and Md. Moshahid Alam Rizvi
Department of Biosciences, Jamia Millia Islamia, New Delhi, India
Corresponding Author:
Syed Hassan Mehdi
Department of Biosciences
Jamia Millia Islamia, New Delhi, India
Tel: 9213231962
E-mail: saeedimam@gmail.com
Received Date: April 27, 2018; Accepted Date: May 21, 2018; Published Date: May 30, 2018
Citation: Mehdi SH, Nafees S, Zafaryab MD, Khan MA, Rizvi A (2018) Chrysin: A promising anticancer agent its Current trends and future Perspectives. Eur Exp Biol Vol. 8 No. 3:16. doi:10.21767/2248-9215.100057
Copyright: © 2018 Mehdi SH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Cancer is one of the major public health concerns in the United States and in other developed countries, affecting million deaths approximately every year worldwide. Cancer incidences show inconsistency between emerging and developed nations. Epidemiologic findings intensely propose that incidence of cancer are predisposed by environmental factors including diet, which is essentially unavoidable. Strategies to prevent cancer comprise overlapping approaches viz. Dietary cancer prevention or chemoprevention. Naturally occurring constituents or pharmacological agents prevent or reverse the initiation phase of carcinogenesis or arrest at progression of carcinogenesis through chemoprevention. Cancer prevention through diet may be possible by the intake of fruits and vegetables. Significant consideration has been given in finding plant-derived dietary agents, which can be developed as promising chemo preventives. One such compound is chrysin (5,7-dihydroxylflavone). It has been shown to possess significant anti-inflammatory, antioxidant and anti-carcinogenic properties. Recently significant progress has been made in studying the biological effects of chrysin at cellular and molecular levels. This review scrutinizes the anticancer effects of chrysin, its considerable potential for development as a cancer chemo preventive agent.
Keywords
Chrysin, Anticancer agents; Chemoprevention
Introduction
The natural polyphenolic components called as “flavonoids,” which are mainly dominant in vegetables, fruits and nuts, comprise supplement with outstanding safety profile and smaller amount toxicity; have gained major attention in the field of chemoprevention [1]. Flavonoids consist of about 4000 biologically active compounds with varied functions like inhibition of enzymes, ligand-activation of signaling pathways. A number of subdivisions of flavonoids have anti-inflammatory, anti-oxidant and anti-proliferative proficiencies. Anticarcinogenic property have been attributed to free radical scavenging property, modification of enzymes involved in metabolism inhibition of carcinogen, inhibition of transcription factors and initiation of apoptosis [2].
Chrysin is a hydroxylated flavone chiefly found in honey, propolis and quite a few species of plants e.g. Pelargonium crispum, Passiflora incarnata, Oroxylum indicum etc. [3,4]. Previous studies have evaluated the concentrations of chrysin in different varieties of honey. The chrysin content is 0.
Thus, the target of the current review is to communicate its chemistry and bioavailability as well as anticancer potential of chrysin particularly referring to anti-inflammatory effect, antiapoptotic effects (Figure 1).
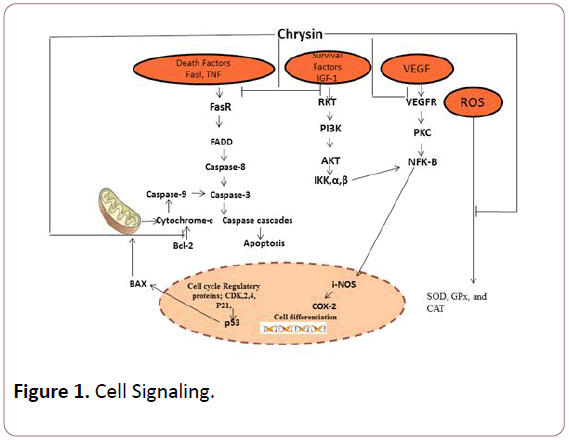
Figure 1: Cell Signaling.
Chemistry and Occurrence
Chrysin (5,7-dihydroxyflavone or 5,7-dihydroxy-2- phenyl-4Hchromen- 4-one) belongs to the flavone class of the 15-carbon skeleton polyphenolic compounds called flavonoids. The distinctive feature of flavones as showed in chrysin is the presence of C2-C3 double bond in ring C and the lack of oxygenation at C-3. Contrasting various flavonoids that have either one commonly at C-40 or two hydroxyl i.e. C30, C40- diortho hydroxyl functional group in ring-B, chrysin lacks oxygenation in this particular ring. Additionally other natural products of chrysin arise due to diversity in ring-A oxygenation as illustrated by the common naturally vital flavonoids like wogonin, oroxylin A and baicalein. Fruits and vegetables are the central nutritive sources of flavonoids. Health aids are attributed to vegetable and fruits when they are consumed daily basis due to the presence of these compounds. Flavonoids are also present in several medicinal plants and have different pharmacological effects described till date. Chrysin and its derivatives have been shown to be the major components of the prominent medicinal plant, Radix scutellariae [5-16]. Further common sources of chrysin eg. Propolis and honey attracted considerable scientific attention recently from pharmacological perspective [17]. Furthermore, various fruits [18], passion flowers like P. caerulea [19] and even mushrooms (e.g. oyster mushroom, Pleurotus ostreatus [20] are renowned to be good sources of chrysin (Figure 2).
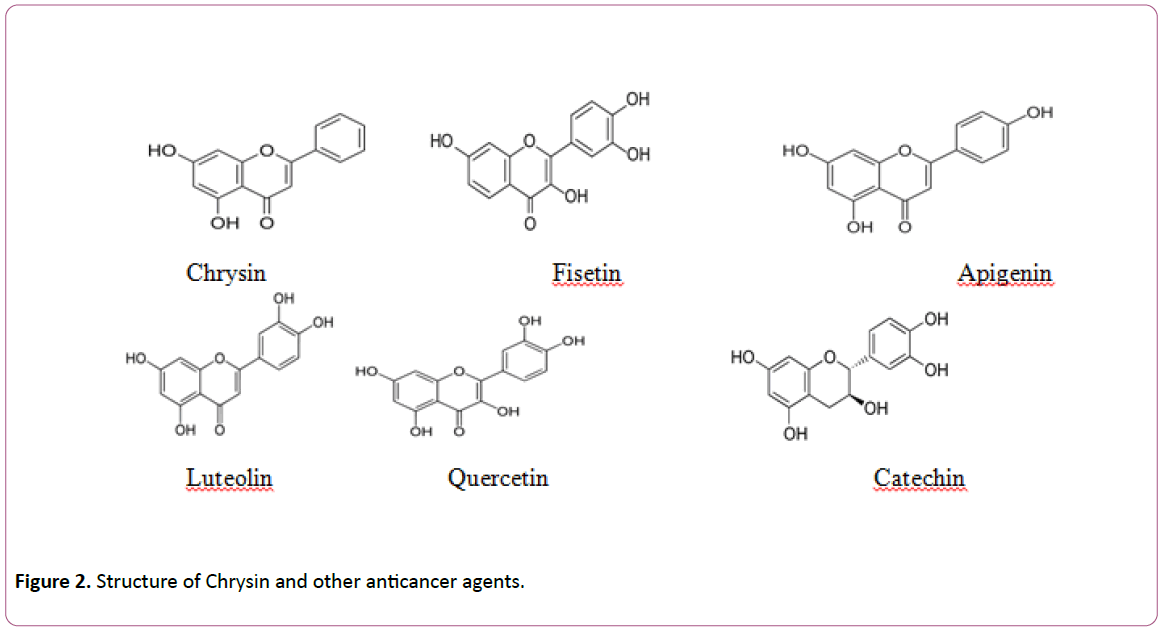
Figure 2: Structure of Chrysin and other anticancer agents.
Chemical properties of chrysin, due to lack of oxygenation on B and C-ring are linked with various pharmacological properties that varies from antioxidant to anticancer properties [21]. Though, differences in the structure of flavones have been revealed to persuade the antioxidant property and the inhibitory effect on the expression of the cyclooxygenase-2 (proinflammatory enzyme). For example, luteolin which is a flavone, showed more Cox-2 inhibition in comparison to chrysin [22] which is due to lack of 30,40 hydroxylation on the “B” ring of chrysin. Luteolin, which has 30,40 hydroxylation demonstrated more antioxidant activity than chrysin. The presence of 30,40 hydroxylation, a double bond between carbons 2 and 3, and the presence of a carbonyl group on carbon 4 have been demonstrated to be crucial to generate antioxidant activity [23]. In contrast to chrysin, luteolin has all these structures and efficiently scavenged the hydroxyl and superoxide radicals in RAW 264.7.
Bioavailability and Toxicity
The useful effect of chrysin and other naturally evolved phytochemicals depends on their bioavailability and solubility of these natural compounds. It has been reported that the concentrations of Chrysin in the plasma is low following a single 400mg oral dose in healthy human volunteers [24]. This reported value is very similar to that reported for the quercetin [25]. A remarkable series of studies have been conducted using the human colon cell line Caco-2 as a model of intestinal absorption has revealed that chrysin has favorable membrane transport properties [26].
Derivatives of chrysin are critical for establishing the whole therapeutic potential of chrysin and its analogues. Some initial studies on formulation such as addition in b-cyclodextrin nanocavity has been shown a promise [27], however more information is needed in this field. Though low dosages of flavonoids present in our daily diet, might be nontoxic for intake by human, intake of higher doses can be hazardous and may lead to toxicity. On the other hand chrysin has been shown to induce toxicity in hepatic cells of trout. The cytotoxicity caused by chrysin has been accredited to the presence of peroxidaselike activity in hepatic cells that leads to oxidation of chrysin to form toxic compounds [28]. It is estimated that this enzyme might be responsible for the toxicity propagated by nutritional flavonoids [29]. Oral administration of chrysin at doses of 1 mg/kg and 10 mg/kg considerably attenuated the enhancement in the production of free radicals and inhibited the activities of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) and inhibition of SOD, CAT and GPx activities of old mice [30] (Figure 3).
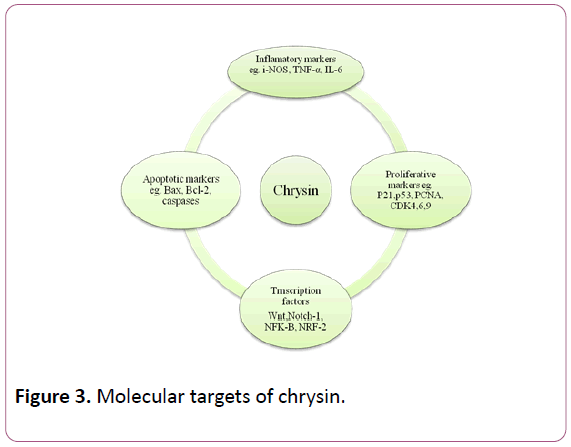
Figure 3: Molecular targets of chrysin.
Anti-cancer Activity of Chrysin
Effects of chrysin on cancer chemoprevention
Chemoprevention is a promising and innovative approach in the area of cancer biology for cancer prevention [31]. Cancer control can be managed by various mechanisms focused at different stages of carcinogenesis like initiation, promotion and progression. The probable methods of prevailing carcinogenesis comprise modulation of biotransformation of carcinogen, scavenging free radicals and changing the expression of genes implicated in signaling pathways, cell proliferation and differentiation [32]. It has been shown that chrysin inhibited the development of 7,12-dimethylbenz(a)anthracene (DMBA)- induced hamster buccal pouch (HBP) carcinomas via various mechanisms with prevention of procarcinogen activation, upregulation of antioxidant and carcinogen detoxification enzymes such as (glutathione (GSH), glutathione peroxidase (GPx), glutathione reductase (GR, GST and QR), apoptosis induction, and metastasis [33]. Some researchers showed that the protective effects of chrysin against DMBA induced breast and two stage skin carcinomas due to its ability to modulate phase I and phase II enzymes [34]. Furthermore, it has been studied that chrysin induce breast cancer resistance protein (BCRP) in Caco-2 cells via induction of aryl hydrocarbon receptor (AhR) and augmented the transport of benzo(a) pyrene-3-sulfate which is a toxic phase II metabolite of lung carcinogen benzo(a)pyrene [35].
In vitro anticancer activity of chrysin
The antitumor cytotoxicity of chrysin has been broadly studied in current years in different cancer cell lines. Chrysin is able to kill cancer cells of lung, breast, cervical, liver, leukemia, colon, nasopharyngeal, prostate, glioblastoma, thyroid and pancreatic cancer [36]. In addition to this, it is interesting to record that chrysin has been shown to demonstrate preferential cytotoxicity against cancer larger than non-cancer cell lines. Fibroblasts and epithelial cells have been reported to be more resistant to the cytotoxic effect of chrysin than cancer cells [37]. Moreover, chrysin induced cell death in numerous models of primary or acquired drug resistance. Chrysin increased sensitivity of doxorubicin resistant BEL-7402 cells (BEL-7402/ ADM) to doxorubicin by inhibiting the expression of nrf2 and its downstream genes hemo oxygenase-1, AKR1B10, and multidrug resistance-associated protein 5 (MRP5) via suppressing phosphatidylinositol-3 kinase-Akt (PI3K-Akt) and extracellularsignal regulated kinase (ERK) pathway [38]. Chrysin can be used alone or in combination to turn over BCRP-mediated multi drug resistance (MDR) as it can successfully inhibit BCRP-mediated efflux to increase the cellular accumulation of BCRP substrates and restore the sensitivity of MDR cells [39].
In vivo anticancer activity of chrysin
In addition to its antitumor activity in various cancer cell lines, chrysin slow down tumor growth in vivo in several animal studies. Chrysin was shown to demonstrate chemopreventive activity by ameliorating oxidative stress and inflammation via NF-kB pathway in an animal model of N-nitrosodiethylamine (DEN) induced and ferric nitrilotriacetate promoted renal carcinogenesis [40]. Chrysin reduced the growth of 1,2- dimethylhydrazine induced colorectal cancer in female Wistar rats by increasing antioxidant mineral levels in the mucosa of intestine which resulted the reduction of nitrosative stress and cell proliferation [41]. N-nitrosodiethylamine (DEN) induced early hepato-carcinogenesis and apoptosis in preneoplastic nodules of male Wistar rats can abrogated by Chrysin [42]. Chrysin has reported chemopreventive activity in azoxymethane induced colon carcinogenesis in both mice and rats via modulation of cryptal cell proliferation activity and apoptosis [43]. The antitumor effect of chrysin was tested in xenograft models and found effective against human anaplastic thyroid carcinoma (HTh7 cells), 4T1 cells (murine breast carcinoma), MDA-MB-231 cells (human breast carcinoma) via inhibition of angiogenesis and generation of apoptosis [36]. Chrysin was shown to inhibit tumor growth by enhancing populations of Tcells and B-cells in the mouse xenograft model of leukemia induced by implanting WEHI-3 cells in Balb/c mice, and by endorsing macrophage phagocytosis and natural killer cell cytotoxicity [44].
Chrysin and cell proliferation
It is well accounted that several natural compounds induce apoptosis in various tumor cells and there are clear evidence that these compounds are effective inhibitors of cancer cell proliferation [45]. It has been reported that chrysin inhibits proliferation and induces apoptosis in cancer cells, making it a possible candidate as anticancer agent [46]. Chrysin diminishes proliferation and induces apoptosis in the human prostate cancer cell line PC-3 [47]. It has been estimated that the proliferation inhibitory effects of chrysin, in leukemia cells seem to be dose-dependent manner [48]. Chrysin and its derivatives exhibited potential anti-cancer effects in cervical carcinoma of human [49]. It was shown that Chrysin (100 μM) showed dosedependent inhibition of U87-MG, MDA-MB-231, U-251 and PC3 proliferation [50,51].
Chrysin and Cell Death
Induction of apoptosis by chrysin
It has been revealed that chrysin possess anticancer activity by endorsing the cell death induced by tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) and increasing TRAIL-induced degradation of caspases 3 and 8 [52,53]. Chrysin guide to the initiation of apoptotic marker proteins in DEN initiated and Fe-NTA promoted renal carcinogenesis by down regulation of Bcl2, which is an anti-apoptotic protein [54]. An inhibitory cause of Chrysin was shown against neuronal cell death via inhibition of mitochondrial apoptosis pathway in SHSY5Y cells [55]. It has been evaluated that chrysin induces apoptotic cell death in human colorectal cancer cells and that induction of pro-apoptotic cytokines tumor necrosis factor (TNF) α and β is involved in apoptosis induced by Chrysin [56]. Chrysin showed dose-dependent apoptosis against the MCF-7 cells [57]. Chrysin induces an in vitro apoptosis of peripheral blood lymphocytes isolated from human chronic lymphocytic leukemia patients through mitochondrial pathway [58]. Chrysin also induced apoptosis in U937, HL-60 and L1210 leukemia cells as demonstrated by the several researchers [59]. Chrysin inhibits the growth of the lung cancer cells by inducing cancer cell apoptosis by Bcl-2 family regulation and activation of cas3 and cas9 that illustrates its anticancer activity [46]. Chrysin induces cell apoptosis by the activation of the p53/bcl-2/caspase-9 pathway in hepato-cellular carcinoma cells viz., HepG2 [52].
Chrysin and autophagy in cancer cells
Autophagy is a pro-death mechanism that causes devastation of cancer cells. There is an evidence that autophagy is vital for the death of cancer cells with deficit in apoptosis. Novel insights into the molecular mechanisms of autophagy are presently leads to the finding of stimulating new drug targets [60]. It has a cytoprotective role essentially via elimination of intracellular pathogens by the initiation of the innate and adaptive immune reactions and it is also noted for its role in maintaining endoplasmic reticulum (ER) and metabolic homeostasis in cancerous cells which go through chronic hypoxia and nutrient reduction. It has been established that chrysin has antiautophagic effects on PC-induced renal toxicity in animal model. Chrysin inhibited TMZ-induced autophagy and MGMT expression and serve as a potential anticancer agent against glioblastomas in Human GBM8901cells [61]. It has demonstrated that chrysin induced autophagy by increasing LC3- II levels to enhance apoptosis in MCF-7 cells [62]. All these reports impose the model that autophagy-associated cell death may constitute a possible adjuvant therapeutic strategy.
Chrysin and inflammation
Inflammation is a protective reaction of the body to infection, physiological stress, drugs and triggers the immune system. The anti-inflammatory mechanism of chrysin prevents inflammatory disorders. Chrysin conquers mast cells mediated allergic inflammation by blocking histamine release and proinflammatory cytokine expression and cause reversal of inflammatory linked dihydrodiol dehydrogenases, overexpression and drug resistance in non-small cell lung cancer cells in combination with wogonin and chrysin [63]. Chrysin alleviates inflammation by the inhibition of COX-2, prostaglandin-E2, histamine, NF-κB pathway, tumor necrosis factor- (TNF-α), i-NOS and cytokines (interleukin-1β, interleukin-2, interleukin-6, and interleukin-12) and activation of peroxisome proliferator-activated factor γ (PPARγ) which suggests that chrysin could be a good candidate in inflammatory diseases [64]. On the other hand chrysin accelerate apoptosis induced by TNF-α which is regulated by NF-κB and increased by TNF-α, and plays a key role in the acceleration of apoptosis [65]. Chrysin showed anti-inflammatory properties associated with its inhibition of nitric oxide, cytokines, chemokines, and growth factors in dsRNA-induced macrophages through the ER stress- CHOP pathway on RAW 264.7 mouse macrophages [66]. It has been shown that chrysin is efficient modulator of PPARγ during the pathogenesis of inflammation by regulating M1/M2 status [67].
Biomedical Applications of Chrysin and Other Flavonoids
Analysis of the biomedical application of flavonoids in cancer therapy is complemented by potential applications in the therapy of specific tumors, including melanoma, colorectal and lung cancers. Chrysin is a natural anticancer bioflavonoid and an emerging potential drug therapy for approximately all types of cancer; however it has poor solubility and bioavailability. New approach to formulate biofunctionalized metallic silver (ChRAgNPs) and gold (ChR-AuNPs) nanoparticles using chrysin as a direct bioreductant and capping agent has been reported. It functionalized metal NPs can be potentially employed as a combinational drug-nano platform for breast cancer therapy [68]. Resveratrol and its naturally dimethylated analog, pterostilbene have analogous biological activities but the higher in vivo bioavailability of pterostilbene has biomedical applications. It has been reported that pterostilbene is an active phytonutrient and a potential drug with several biomedical applications [68]. According to the study conducted earlier it is demonstrated that Silybin augmented the therapeutic potential of doxorubicin, cisplatin and carboplatin in both estrogendependent and -independent human breast carcinoma cells (MCF-7 and MDA-MB468) [69]. The magnetic nanoparticles draw increasing attention due to their prospects in cancer therapy and used as drug carriers for other diseases. It has been investigated that quercetin conjugated superparamagnetic Fe3O4 nanoparticles for in-vitro analysis of breast cancer cell lines for chemotherapy. The results confirmed the prepared quercetin conjugated Fe3O4 nanoparticles are capable anticancer agents for targeted drug delivery and proposed that magnetite nanoparticle based drug conjugation might be an efficient approach to deliver an anticancer drug to the targeted cancer cells and also boost the promising treatment in biological applications [70].
Future Perspectives
Flavonoids are presumed to be nontoxic due to their prevalent distribution in the diet with little or no toxicity. This property is noteworthy as a number of drugs which are used in the form of medicine have adverse side effects. The anticancer properties of chrysin are due to the diverse variety of mechanisms, including variations in the enzymes activity and oncogenic pathways inhibition. Concern has been shown in the anti-cancer activity of chrysin when it was exposed that chrysin together with other flavonoids decreased the enzymes activity which is responsible for the detoxification of carcinogens. Numerous studies have endorsed that chrysin has both therapeutic and anticancer properties. Although most of the evidences about the biological activity of chrysin are based on in vitro studies, preclinical experimental studies will be vital in evaluating the possible usage of chrysin before implementation of any clinical trials. It remains unexplored why and how chrysin upsurges the anticancer effects of chemotherapeutic drugs.
Conclusion
Chrysin has been designated as health-promoting and anticancer compounds. It has shown its activity in several biological assessments suggesting that it may probably be advantageous against many disorders. Chrysin blocks many cancer-related pathways and inhibits cancer by promoting apoptosis and moderating cell death by autophagy. By substantially interacting with signaling pathways, chrysin inhibits several signaling pathways. However more widespread studies in this direction are required in the near future, not only with chrysin, but also numerous additional non-toxic and naturallyoccurring chemopreventive compounds to rationalize their probable clinical application in chemotherapy. Thus we propose that chrysin be explored further for the treatment of cancers either alone or as an adjuvant with other chemotherapeutic drug.
Acknowledgement
The authors would like to acknowledge the University Grants Commission (India) DS Kothari Fellowship (Grant Number: 4-2/2006 (BSR)/BL/13-14/0190) New Delhi, for providing the financial assistance.