Case Report - (2018) Volume 2, Issue 2
Antonella La Brocca*, Paolino De Marco, Kerengi Kabhuli, Giovanna Rizzo, Anna Trapani, Gabriele Barletta, Antonio Giulio Giannone, Ada Maria Florena, Gabriella Militello, Gaspare Gulotta and Sebastiano Bonventre
Unit of General and Emergency Surgery, Department of Surgical, Oncological and Oral Sciences, University of Palermo, Palermo, Italy
*Corresponding Author:
Antonella La Brocca
Unit of General and Emergency Surgery
Department of Surgical, Oncological and Oral Sciences
University of Palermo, Palermo, Italy
Tel: 091517495
E-mail: antonellalabrocca@msn.com
Received date: 22 June, 2018; Accepted date: 16 August, 2018; Published date: 23 August, 2018
Citation: La Brocca A, Paolino DM, Kabhuli K, Rizzo G, Trapani A, et al. (2018) Radiation-induced breast angiosarcoma: a Case Study. Res J Oncol. Vol. 2 No. 2: 2.
Copyright: Copyright: © 2018 La Brocca A, et al. This is an open-access article distributed under the terms of the creative Commons attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Mammary angiosarcoma (AS) is an aggressive vascular tumor. It may appear de novo (primary angiosarcoma) or as a complication of radiotherapy (radiation-induced AS) or chronic lymphedema (Stewart-Treves syndrome). Our attention in this case study is focused on radiationinduced breast AS, a rare but severe long-term complication in the breast-preserving management of breast cancer, treated with breast-conserving surgery and radiotherapy. Contrary to what occurs in primary AS of the breast, which usually appears in women aged 30-50 years, radiation-induced breast AS affects older women (with a median age of 67-71 years) about 10.5 years after radiotherapy as treatment of primary breast cancer (the median onset latency varies from 5-10 years). The former is localized in the parenchyma, the latter in dermal and subcutaneous layers of the skin of radiated fields. The prognosis is poor with a high rate of relapse and an increased tendency to metastasis.
Keywords
Breast cancer; Radiation; Secondary angiosarcoma
Introduction
The incidence of radiation-induced breast AS is increasing because breast-conserving treatment with radiotherapy (RT) has replaced mastectomy as standard of care for early-stage breast cancer [1]. The cumulative incidence of this complication in literature is 0.9 per 1000 breast cancer cases over 15 years [2]. It was described for the first time by Davies et al in 1983 [3]. Angiosarcomas secondary to radiotherapy meet Cahan's criteria: history of exposure to radiotherapy, histological discordance between primary and secondary tumor, and a long latency period between radiation therapy and the appearance of sarcoma [4].
Clinical Presentation
A 75-year-old woman came to Breast Unit of Giaccone University Hospital in March 2015 for the appearance of skin lesions in the internal equatorial quadrant of her right breast. In 2008 she underwent right upper exterior quadrantectomy and lymphadenectomy of the omolateral axilla for an apocrine carcinoma, followed by adjuvant RT, administered without incident. Her medical history included arterial hypertension, chronic renal failure, osteoporosis, appendectomy, cholecystectomy and finally smoking 30 years ago. In March 2015 a cutaneous lesion appeared in the internal equatorial region, detected by edema and erythematous skin with central ulceration areas, measuring 4.0 × 7.0 cm. Further injuries appeared with the same features but lower size along the entire right gland (Figures 1 and 2).
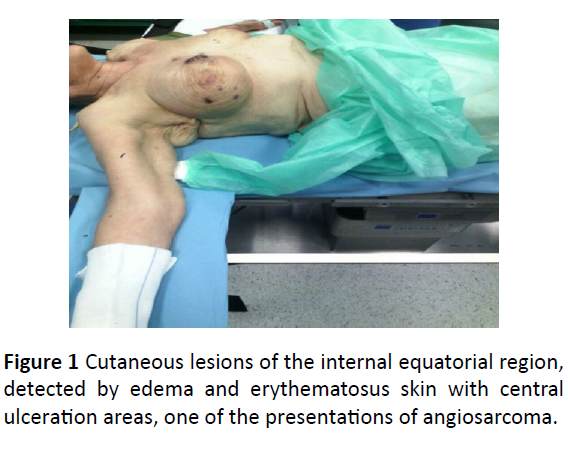
Figure 1: Cutaneous lesions of the internal equatorial region, detected by edema and erythematosus skin with central ulceration areas, one of the presentations of angiosarcoma.
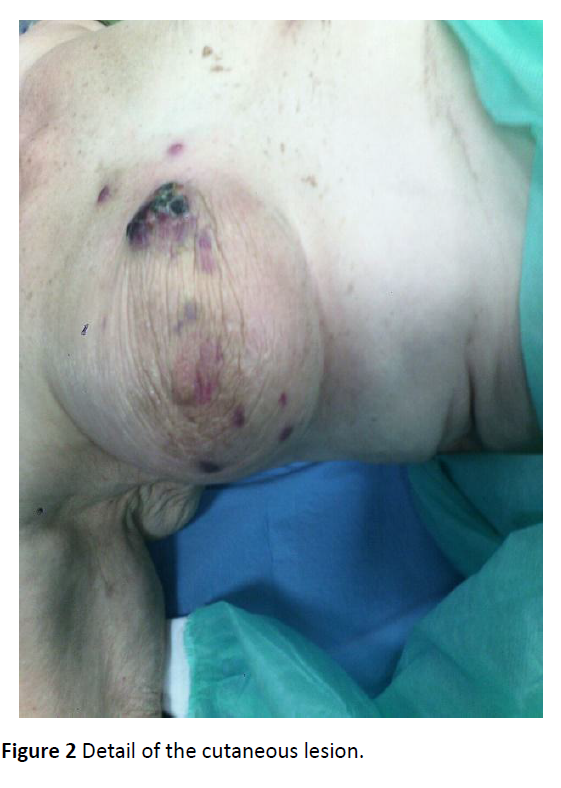
Figure 2: Detail of the cutaneous lesion.
The injury increased over time without pain, so our Surgical Team performed a radical mastectomy with Madden technique in July 2015. The macroscopic anatomopathological examination showed multiple bristle nodules, the largest of which was 4.0 × 3.5 cm. The nodules consisted of atypical cells that expressed the following immunophenotype: CD31+, CD34±, factor VIII±, CKAE1/AE3-, desminate, podoplanin, rate of proliferation (ki67 + cells) >90%, c-myc+ (Figure 3). This immunophenotype was consistent with the diagnosis of breast angiosarcoma. Surgical resection margins were not interested by the tumor. Patient’s postoperative course was uncomplicated. New skin lesions appeared after 6 months. The lesions detected were partly whitish and white-brown, about 1 cm in diameter. The excision of skin and subcutaneous tissue, including muscular plane was conducted under local anesthesia. The macroscopic anatomopathological examination showed proliferation with alternation of solid and vascular growth patterns, consisting of atypical epithelioid and fused morphology cells expressing an immune-phenotype posing for breast angiosarcoma. The lesion extended to the subcutaneous plane, but saved the muscle plane and the resection margins. In May 2016, after four months, new skin lesions appeared again in the right parasternal region and in the pericicatricial area. They were also positive for breast angiosarcoma. After 3 months, the patient developed two additional cutaneous lesions, the first lesion was located next to the mastectomy scar, while the second one appeared on the abdominal wall. Skin lesions were characterized, at the macroscopic anatomopathological examination, by the presence of dermal hypodermic nodules consisting of anastomized vascular structures with marked anisocariosis of endothelial cells. Morphological and immunophenotypic framework (CD31+, c-myc+) were consistent with cutaneous metastases from breast angiosarcoma. In January 2017 the patient returned for a lesion to the right hemithorax, an angiosarcoma again. Neither chemotherapy nor RT was performed during the time intervals between the appearances of lesions. The patient is alive up until the time of this writing.
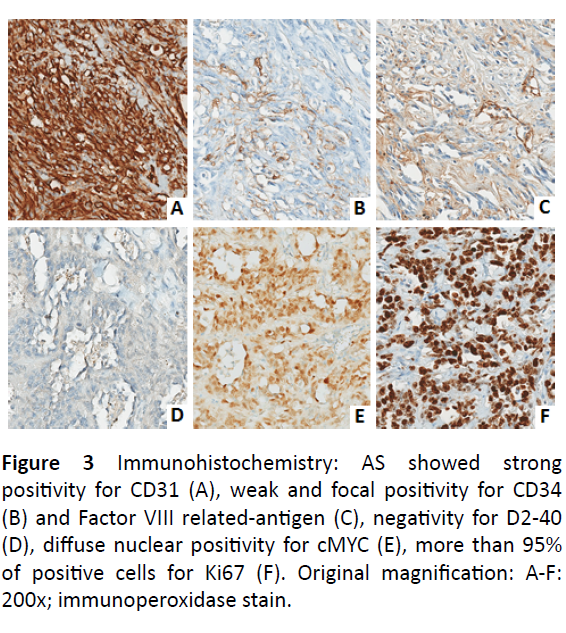
Figure 3: Immunohistochemistry: AS showed strong positivity for CD31 (A), weak and focal positivity for CD34 (B) and Factor VIII related-antigen (C), negativity for D2-40 (D), diffuse nuclear positivity for cMYC (E), more than 95% of positive cells for Ki67 (F). Original magnification: A-F: 200x; immunoperoxidase stain.
Discussion
Our patient was affected by apocrine carcinoma of the breast, a rare, primary breast cancer. This malignancy is characterized by apocrine morphology of the cells, estrogen receptor-negative and androgen receptor-positive profile with a frequent overexpression of Her-2/neu protein [5]. One of the complications associated with the use of RT is the secondary development of malignant tumors, although current evidence suggests that only 3–6% of all sarcomas are related to adjuvant therapy [6]. The risk of developing secondary angiosarcoma is greater if the RT is performed at an early age or if the patient received chemotherapy concurrent with RT [7]. The risk is also greater at the edges of the radiation fields where the radiotherapeutic dose is more heterogeneous [8]. Secondary AS can have different kind of clinical presentations: hematoma, reddish purple multifocal nodules (as our patient) or the appearance of eczema [9]. When tumor increases in size (with an explosive growth in high grade tumors, and more insidious in low grade ones), ulcers or edema may appear, instead pain is uncommon [10]. Radiation-induced breast AS is characterized by a rapid proliferative pattern and a tendency to infiltrate any organ in the body, two thirds of the patients experienced a local recurrence, even after an R0 resection, as in the clinical case described [11]. The pathogenesis is unclear. MYC oncogene mutation in the 8q24 region, p53 gene inactivation [12] and BRCA1 and BRCA2 genes mutations [13] have been found. Radiology, with Magnetic Resonance Imaging, because dermal or intraparenchymal lesions usually enhance the contrast [14]. In any case, biopsy confirmation is always necessary. The treatment of secondary AS is not evidence-based because of the lack of guidelines, due to the rarity of the disease. An aggressive surgery obtained with a R0 mastectomy, defined as free margin >2cm, is recommended [15]. Other authors advance aggressive resections involving the muscle because the lower border is the most frequently affected in the case of incomplete excision [8]. Excision of the tumor, along with all radiated tissue, provided a lower incidence of recurrence [4]. Adjuvant RT could achieve a better control of the disease [16]. Chemotherapy with taxanes, liposomal doxorubicin, and traditional anthracycline is recommended in the case of inoperable disease [9]. In addition to systemic chemotherapy is described a case report in which cutaneous and subcutaneous tumor nodules in the chest wall and in abdominal skin were palliatively treated with local electro-chemotherapy with intravenous bleomicin. Electro-chemotherapy is a combination of a cytotoxic drug and electroporation, in order to obtain a reversible destabilization of the membrane [17]. Despite these innumerable therapeutic approaches the prognosis remains poor.
Conclusion
Post radiation angiosarcoma is a rare and aggressive disease. A multidisciplinary team, consisting of breast cancer surgeon, oncologist, pathologist, radiologist and plastic surgeon, would be advisable to manage the malignancy in an optimal way. More studies are needed, in order to draw up evidence-based guidelines for surgical and non-surgical treatment. Early clinical suspect and subsequent early diagnosis remains the best approach for controlling disease progression.