Research Article - (2019) Volume 9, Issue 2
Oral LD50 of total saponins and tannins isolated from Dialium guineense stem bark
Abu OD1*, Adeogun EF1 and Ebhohon SO2
1Department of Biochemistry, University of Benin, Benin City, Edo State, Nigeria
2Department of Biochemistry, College of Natural Sciences, Michael Okpara University of Agriculture, Umudike, Umuahia, Abia State, Nigeria
Corresponding Author:
Osahon Abu
Department of Biochemistry, Faculty of Life Sciences
University of Benin, Benin City, Edo State, Nigeria
Tel: + 2347086427636
E-mail: osahon.abu@uniben.edu
Received date: June 17, 2019; Accepted date: June 24, 2019; Published date: July 10, 2019
Citation: Abu OD, Adeogun EF, Ebhohon SO (2019) Oral LD50 of total saponins and tannins isolated from Dialium guineense stem bark. Eur Exp Biol. Vol.9 No.2:8.
Background and Objective: The safety of bioactive compounds isolated from plant extracts has become a global concern. The aim of this study was to determine the oral LD50 of total saponins and tannins isolated from Dialium guineense stem bark using Sprague Dawley rats.
Materials and Methods: Male Sprague Dawley rats (n=13) weighing 150-180 g (mean weight=165 ± 15 g) were used for this study. The aqueous, ethanol and methanol extracts of the plant stem bark were obtained using cold maceration method. The lorke method was used to determine oral LD50 of isolated total saponins and tannins of D. guineense. Signs of toxicity and possible death of rats were also monitored for twenty-four (24) h.
Results: The major signs of toxicity observed within 24 h were: difficulty in breathing, loss of appetite and general weakness. No deaths were recorded in both phases and all the animals in each phytochemical group survived. The oral LD50 of total saponins and tannins of D. guineense were greater than 5000 mg/kg body weight (bwt).
Conclusion: The findings of this study have shown that saponins and tannins of D. guineense stem bark are not toxic at doses not exceeding 5000 mg/kg bwt.
Keywords
Dialium guineense; Acute toxicity; Rats, Extract; Lethal dose
Introduction
In recent times, plant-derived substances have become of huge importance to man due to their many applications. Extraction methods involve the separation of medicinally active portions of plant tissues from the inactive/inert components using selective solvents. These plant components exist as complex mixtures of many medicinal metabolites, such as alkaloids, glycosides, terpenoids, phenols, flavonoids, and lignans [1]. Medicinal plants have long been recognized as important sources of therapeutically active compounds. Evidence-based research supports the medical and pharmacological benefits of plant-derived compounds with interest in the identification and characterization of bioactive compounds from natural sources [2].
Dialium guineense (Velvet Tamarind) is a tall, tropical, fruitbearing tree which belongs to the Leguminosae family. It has small, typically grape-sized edible fruits with brown hard inedible shells. Dialium guineense grows in dense forests in Africa along the southern edge of the Sahel and it can be found in West African countries such as Ghana where it is known as “Yoyi”, Sierra Leone, Senegal, Guinea-Bissau and Nigeria where it is known as “Awin” or "Igbaru" in Yoruba, “Icheku” in Igbo, “Tsamiyarkurm”in Hausa and “Amughen” in Edo. The bark and leaves have medicinal properties and are used against several diseases [3]. Despite the widespread use of D. guineense in medical research, the safety of its bioactive components is rarely reported. Saponins are a class of chemical compounds found in abundance in various plants species. They are amphipathic glycosides grouped phenomenologically by the soap-like foam they produce in aqueous solutions when shaken, and structurally by having one or more hydrophilic glycoside moieties combined with a lipophilic triterpene or steroid derivative [4]. Tannins are phenolic compounds of high molecular weight, which are soluble in water and alcohol, and are found in the root, bark, stem and outer layers of plant tissue. Their acidic nature in reactions is attributed to the presence of phenolic or carboxylic acid group [5]. The aim of this study was to determine oral LD50 of total saponins and tannins isolated from D. guineense stem bark using Sprague Dawley rats.
Methods
Experimental rats
Male Sprague Dawley rats (n=13) weighing 150-180 g (mean weight=165 ± 15 g) were obtained from the Department of Anatomy, University of Benin, Benin City. The rats were housed in metal cages under standard laboratory conditions: temperature of 25°C, 55%-65% humidity and 12 h light/12 dark cycle. They had free access to rat feed (pelletized growers mash) and clean drinking water. Prior to the commencement of the study, the rats were acclimatized to the laboratory environment for one week. The study protocol was approved by the Faculty of Life Sciences Ethical Committee on Animal Use.
Collection of plant material
The stem barks of D. guineense were obtained from Auchi Area of Edo State, Nigeria and authenticated at the herbarium of the Department of Plant Biology and Biotechnology, University of Benin, Benin City.
Plant preparation and extraction
The stem bark was brushed and shade dried at 30°C for a period of two weeks and crushed into small pieces using clean mortar and pestle. Aqueous, ethanol and methanol extracts of the stem bark were obtained using the cold maceration method as described previously [6].
Qualitative phytochemical analysis
The aqueous, ethanol and methanol extracts were screened for the presence of saponins and tannins based on standard methods [7,8].
Acute toxicity test
This was carried out using Lorke’s method [9-11], which comprised two phases. In the first phase, nine (9) rats were divided into three groups of three (3) rats each. Each group of rats was administered total saponins and tannins of D. guineense stem bark orally at doses of 10, 100 and 1000 mg/kg bwt. The rats were placed under observation for 24 h to monitor their behavior and mortality. In the second phase, 4 rats were randomly assigned to four groups of 1 rat each. The rats were administered higher doses of total saponins and tannins of D. guineense stem bark orally at doses of 1600, 2900 and 5000 mg/kg bwt, and then, observed for 24 h for signs of behavior and mortality (Table 1).
| Phase 1 | ||
|---|---|---|
| Groups | No. of rats | Dose (mg/kg bwt) |
| 1 | 3 | 10 |
| 2 | 3 | 100 |
| 3 | 3 | 1000 |
| Phase 2 | ||
| 1 | 1 | 1500 |
| 2 | 1 | 2500 |
| 3 | 1 | 2900 |
| 4 | 1 | 5000 |
Table 1: Acute toxicity study on total saponins and tannins of D. guineense stem bark on Sprague Dawley rats.
The lethal dose (LD50) of total saponins and tannins was calculated thus:
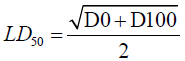
Where D0=Highest dose that gave no mortality, D100=Lowest dose that produced mortality.
Statistical Analysis
Data are expressed as mean ± SEM, and statistical analysis was performed using GraphPad Prism Demo (6.07).
Results
Phytochemical screening of ethanol, methanol and aqueous extracts of D. guineense stem bark
As shown in Table 2, saponins and tannins were relatively abundant in the ethanol and aqueous extracts of the plant, while saponins were absent in the methanol extract.
| Parameters | Extracts | ||
|---|---|---|---|
| Ethanol | Methanol | Aqueous | |
| Saponins | + | ND | ++ |
| Tannins | ++ | ++ | + |
+: present
++: relatively abundant
ND: not detected
Table 2: Qualitative phytochemical analysis.
Results of acute toxicity study on total saponins and tannins of D. guineense stem bark on Sprague Dawley rats
No mortality was recorded even at 5000 mg/kg bwt in the saponins and tannins groups (Tables 3 and 4).
| Dose (mg/kg bwt) | No. of rats | No. of deaths | Survival | Mortality ratio |
|---|---|---|---|---|
| 10 | 3 | 0 | 3 | 0/3 |
| 100 | 3 | 0 | 3 | 0/3 |
| 1000 | 3 | 0 | 3 | 0/3 |
| 1600 | 1 | 0 | 1 | 0/1 |
| 2900 | 1 | 0 | 1 | 0/1 |
| 5000 | 1 | 0 | 1 | 0/1 |
Data are a number of death and survival of rats
No. of deaths recorded=Nil
No. of rats that survived=All
Mortality ratio=no. of death/no. of survival
Oral LD50>5000 mg/kg bwt
Table 3: Outcome of acute toxicity study of total saponins of D. guineense stem bark extracts.
| Dose (mg/kg bwt) | No. of rats | No. of deaths | Survival | Mortality ratio |
|---|---|---|---|---|
| 10 | 3 | 0 | 3 | 0/3 |
| 100 | 3 | 0 | 3 | 0/3 |
| 1000 | 3 | 0 | 3 | 0/3 |
| 1600 | 1 | 0 | 1 | 0/1 |
| 2900 | 1 | 0 | 1 | 0/1 |
| 5000 | 1 | 0 | 1 | 0/1 |
Data are a number of death and survival of rats
No. of deaths recorded=Nil
No. of rats that survived=All
Mortality ratio=no. of death/no. of survival
Oral LD50>5000 mg/kg bwt
Table 4: Outcome of acute toxicity study on total tannins of D. guineense bark extracts.
Discussion and Conclusion
There have been growing interests in the toxicity of substances purified from plants basically to determine their safety. The aim of this study was to determine the oral LD50 of total saponins and tannins isolated from D. guineense stem bark using Sprague Dawley rats. The results showed that no death was recorded in both phases after 24 h and all the rats in each group survived. The major signs of toxicity observed within 24 h were: difficulty in breathing, loss of appetite and general weakness. Thus, the median lethal dose LD50 (oral) of total saponins and tannins of D. guineense stem bark were greater than 5000 mg/kg bwt. These results suggest that the total saponins and tannins may be relatively safe [8]. One major and overriding criterion in the selection of herbal medicine for use in health services is safety. Phytochemicals present in plant extracts should not only be clinically effective, but safe for consumption. Therefore, screening of bioactive components present in plant extracts to identify their toxic potentials is the need for selection of plants for drug formulations.
Significant Statement
The findings of this study have shown that saponins and tannins of D. guineense stem bark are not toxic at doses not exceeding 5000 mg/kg bwt.
Authors’ Contribution
This work was conceived, designed and developed by the authors. Abu wrote the manuscript and was proofread by Adeogun.
Acknowledgment
The authors wish to appreciate Metro-Research and Biotechnology Africa for their technical assistance and to the editorial staff and reviewers who took part in the success of this special issue.