Keywords
Polymorphism, Pharmaceutical, Stability, Bioavailability, Solubility
Introduction
As per the internal structure, a solid can exist in a crystalline or amorphous form. If a substance exists in more than one form of crystalline, those varied forms are designated as polymorphs and the phenomenon is known as polymorphisms [1]. The word polymorph is derived from the Greek words ‘poly’, which means many and ‘morph’ which means ‘form’. Hence polymorphism refers to different structural forms of a chemical substance [2]. Among polymorphs of certain compounds, one is more stable than the other under all conditions. Whereas in the case of other compounds, one polymorph is stable within a particle range of temperature and pressure while another is stable under a different set of conditions [3]. More than 59% of the substances are known to exist in more than one crystal form. At least 38% can form polymorphic modifications 30% hydrates and 12% are solvates [4].
The arrangement of the molecules in a crystal determines the physical properties and chemical properties of a drug. The physicochemical properties of the drug molecule exhibit their influence on its performance of drug. The shape and particles of the solid drug can affect the pharmaceutical operations, such as milling, mixing, filtering, washing, drying, tableting, and dissolution. Crystalline solids can exists as polymorphs, hydrates or solvates, or combinations of Organic molecules can be able to forming different crystal lattices through two different mechanisms. One is termed as packing polymorphism or orientational polymorphism, which provides instances in which molecules that are conformationalLy relatively rigid, can be turned into different 3-dimentional structures through different intermolecular arrangements and another mechanism is conformational polymorphism, which arises when a flexible molecule bends into different conformations, and subsequently can be packed into alternative crystal structure [5].
Classification
Crystals are defined by a high degree of order, as a three dimensional periodicity of atoms or ions with their particular positions and additionally with configurational periodicity for molecules that form the crystal”. Most solid forms of drugs are consumed in the crystalline state, and most pharmaceutically relevant crystals are formed by organic molecules. So, most of the drugs are organic molecules. The crystals formed by these molecules are held together by dipole-dipole interactions, hydrogen bonds, or London forces. Since these are at least in an order of magnitude weaker than ionic or covalent interactions, the melting points of molecular crystals are lower than those of the aforementioned crystalline structures. For examples the melting points of paracetamol (molecular crystals) and NaCl (metal crystal) are 169°C and 801°C respectively [6].
Every crystal is periodically organized depending upon a specific unit cell with its specific characteristic. A unit cell is defined as the smallest unit of a crystal, which, if repeated, could generate the whole crystal. It is possible to summarize the types of unit cells into seven crystal systems, which can be understood as unique unit cell shapes and which can be differentiated with respect to the relative lengths on their axes and angles between the axes. These seven crystal systems are cubic, tetragonal, orthorhombic, monoclinic, triclinic, rhombohedral and hexagonal (Figure 1).
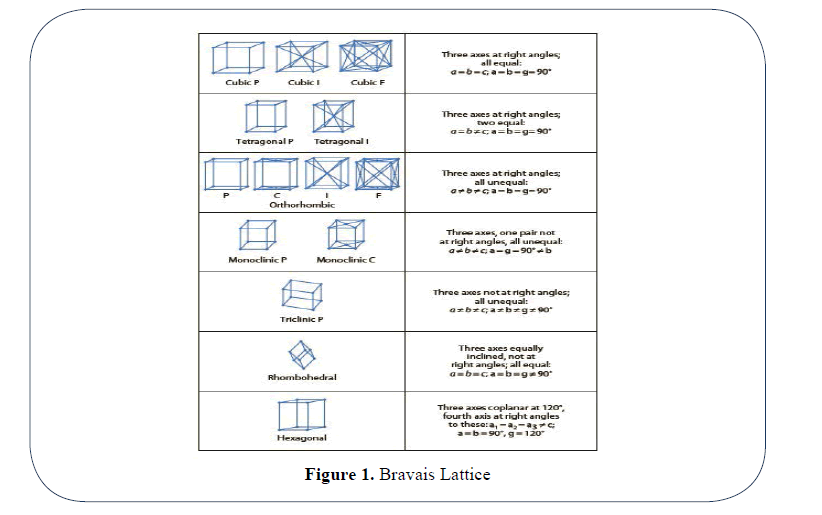
Figure 1: Bravais Lattice
It is important to note that different modifications of a drug may not necessarily form crystals with a different type of Bravais lattice [7].
Polymorphs are broadly classified into two types. They are 1. Monotropes 2. Enantiotropes. For a monotropic system, plots of free energy of the various polymorphs against temperature do not cross before all polymorphs become melt. For an enantiotropic system, a plot of the free energy against temperature shows a crossing point at various melting points, and it may be possible to convert reversibly the two polymorphs on heating and cooling. Enantiotropes are members of a pair of polymorphs whose mutual transitional temperature is less than the melting point of either of the polymorph [7] [8].
Table 1: Crystals grouped by properties [8]
| TYPES |
CHARACTERISTICS |
| Covalent Crystals |
A covalent crystal has true covalent bonds between all of the atoms in the crystal. Many covalent crystals have extremely high melting points. Examples: Diamond and Zinc sulfide crystals. |
| Metallic Crystals |
Individual metal atoms of metallic crystals sit on lattice sites. This leaves the outer electrons of these atoms free to float around the lattice. Metallic crystals tend to be very dense and have high melting points. |
| Ionic Crystals |
The atoms of ionic crystals are held together by electrostatic forces. Ionic crystals are hard and have relatively high melting points. Examples: Sodium chloride and Potassium chloride crystals. |
| Molecular Crystals |
These crystals contain recognizable molecules within their structures. A molecular crystal is held together by non-covalent interactions, like Vander Waals forces and Hydrogen bonding. Molecular crystals tend to be soft with relatively low melting points. Example: Rock candy, the crystalline form of table sugar or sucrose. |
Amorphous Form [9-11]
The amorphous materials will be in solid forms in which the molecules are not organized in a definite lattice pattern. This means that the active sites that would be interacting with each other to build the crystal are now exposed more directly to the solid environment. Their structure can affect several properties such as product stability, storage, processing, compatibility, hygroscopicity and dissolution. For example, Novobiocin exists in crystalline form and amorphous form. But the amorphous form shows 10 times more dissolution rate than that of crystalline form. Even the amorphous structure compound shows more enhanced solubility than that of metastable state crystal form due to large intermolecular spaces in the structure and their weak bonding between each structure.
Amorphous solids have two characteristic properties. When they are broken, they produce fragments with irregular and often curved surfaces which are not arranged in a regular array. An amorphous trans lucent solid is called glass. Some solids are intrinsically amorphous because either their components can form a stable crystal lattice or they contain impurities that distrusts the lattice.
Amorphous solids are isotropic in nature as their physical properties will remain same in all directions.
Examples: Carbon, Tar, Candlewax, Glass
“Glass is an amorphous translucent solid form.” (Figure 2)
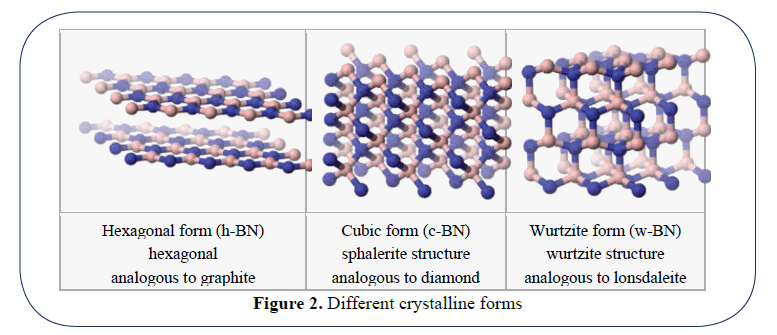
Figure 2: Different crystalline forms
Metastable Form
Depending on their relative stability, one of the several polymorphic forms will be physically more stable than the others and such a stable polymorph represents the lowest energy state, which has the highest melting point and least aqueous solubility. The remaining polymorphs’ are called metastable form [12]. The solvents used in the recrystallization process play a key role in the formulation of met nature form which shows more aqueous solubility.
In contrast the controlled use of a metastable form can offer considerable benefits’ when it has advantageous physical properties such as improved solubility [13].
Importance of Polymorphism
Solubility and bioavailability
The most critical issues related to drug substance polymorphism is equilibrium solubility. Equilibrium solubility is defined as the concentration of drug dissolved when there is equilibrium between the solid drug substance and solution.
A study on equilibrium solubility studies helps to assess the effect of crystal structure or polymorphism. Particle size and wettability of a solid can be modified by processing parameters, but equilibrium solubility is determined by polymorphic form. Equilibrium solubility should be assessed according to procedures recommended in the biopharmaceutical classification system (BSC) guidance [14] (Table 2).
Table 2: BSC classification system
| Class |
Solubility |
Permeability |
| I |
High |
High |
| II |
Low |
High |
| III |
High |
Low |
| IV |
Low |
Low |
The most recent illustration, which shows the impact of polymorphs on solubility and dissolution rate is the protease inhibitor Ritonavir. This form II is more soluble and showing high dissolution rate when compared with the form I. The two crystal forms differ substantially in their physical properties such as solubility and dissolution rate.
The characteristics of drugs in solid state are known to be potentially exerted with a significant influence on the solubility parameter. Polymorphs of a drug substance can have different apparent aqueous solubility and dissolution rate, and such differences will change the bioavailability of the drug and it is often difficult to formulate a bioequivalent drug product using a different polymorph [15].
Polymorphic differences and transformation that result in different apparent solubility and dissolution rate are generally detected by dissolution testing. If there are differences in the solubility of various polymorphs, they will show effect upon drug product, bioavailability, bioequivalence, gastrointestinal motility and intestinal permeability. A drug with dissolution as the rate limiting step, the polymorphic form affects the bioavailability and bioequivalence. If a drug has permeability as the rate limiting step, solubility of polymorphic forms show less effect on bioavailability and bioequivalence.
Stability
Polymorphs of a pharmaceutical solid may have different physical and chemical solid state properties. The most stable polymorphic form of a drug substance is often used because it has the lowest potential for conversion from one polymorphic form to another while the metastable form may be used to enhance the bioavailability. The relative polymorphic stability may be determined by an appropriate examination of the relative apparent solubility of supersaturated solution of polymorphic pairs. Since the rate of conversion to the more stable form is often rapid when mediated by the solution phase, the less stable polymorph with the grater apparent solubility dissolves, while the more stable polymorph with the lower apparent solubility crystallizes out upon sanding. One polymorph may convert to another during manufacturing and storage, particularly when a metastable form is used.
Applications
Better processing characters: It is used to change the micrometrics properties of the drug such as flow property and compressibility.
Easy of handling: Crystallization facilities various operations such as transportation during operations and storage conditions.
Improved bioavailability: Some drugs are more effective in their crystalline form. Examples are penicillin G does not dissolve immediately in the gastric fluids. Hence, the bioavailability of drug will increase [16].
Sustain release; size and shape of drug substance also play a vital role, when they are used as sustained release dosage e.g. protamine zinc insulin.
Polymorphism in pharmaceuticals
Acetaminophen is a common analgesic. It is usually obtained as monoclinic prisms on crystallization from water a less stable orthorhombic polymorph having better physical properties for pressing into tablets [17].
Gray Nicolas was found that paracetamol crystallization of the from 1 and form 2, he also analyzed the characterization of signals crystal by x-ray diffraction, powder X-ray diffraction, DSC, thermo microscopy, optical crystallography, scanning electron microscopy, compaction study.
Stephen et al., was studied on the c825T polymorphism of the gene encoding the βasubunit of G proteins has been associated with increased sodium-hydrogen exchange and low renin in patients with essential hypertension H. He concluded that C825Tpolymorphism of the G-protein β3-subunit may help identify patients with essential hypertension who are more responsive to diuretic therapy [18].
The Three polymeric forms of Chloramphenicol palmitate A, B and C. B form shows best availability and A form is virtually inactive biologically [19].
Conclusion
The study of polymorphism is necessary to acquire knowledge on rapid absorption of low solubility of drugs in systemic circulation, to improve dissolution rate and bioavailability. From the above information we can conclude that the order of solubility of different forms of polymorphs are amorphous > metastable > stable form, whereas in terms of either stability the order is stable > amorphous > metastable.
References
- Liebermann and Lachman. Theory and Practices of Industrial Pharmacy. Varghese publication house. 187-93.
- Raza K, Kumar P, Ratan S, et al. Polymorphism: The Phenomenon Affecting the Performance of Drugs. SOJ Pharm Pharm Sci 2014; 1(2):1-10.
- Haleblian J and Walter M. Pharmaceutical Applications of Polymorphism. J Pharm Sci 1969; 58(8):911-29.
- https://www.eurostar-science.org/conferences/abst9/Griesser.pdf
- Datta S and Grant D J. Crystal Structures of Drugs: Advances in Determination, Prediction and Engineering. Nat Rev Drug Discov 2004; 3:42-57.
- Loyd V and Allen J. Remington: An Introduction to Pharmacy. Pharmaceutical Press 2013; 2-4.
- Linda A. Felton. Remington Essentials of Pharmaceutics. Pharmaceutical Press 2012; 193-4.
- Thiruvengadam E and Vellaisamy G. Polymorphism in Pharmaceutical Ingredients A Review. World Journal of Pharmacy and Pharmaceutical Sciences 2014; 3(3):621-33.
- John F. Bauer. Pharmaceutical Solids- The Amorphous Phase. Journal of Validation Technology 2009.
- https://chemwiki.ucdavis.edu/?title=Textbook_Maps/General_Chemistry_Textbook_Maps/Map:_Chemistry_(Averill_%26_Eldredge)/12:_Solids/12.1:_Crystalline_and_Amorphous_Solids
- https://en.wikipedia.org/wiki/Boron_nitride
- Brahmankar D M and Jaiswal S B. Biopharmaceutics rand pharmacokinetics a treatise. Vallabh prakashan 1995.
- https://www.chem.ucl.ac.uk/cposs/caseforsupport.html
- Inamdar N, Rathi A A and Saifee M. Drug Polymorphism: A Review. International Journal of Health Research 2009; 2(4):291-306.
- Gandhi S and Chandrul K. Pharmaceutical Solid Polymorphism in Abbreviated New Drug Application (ANDA) - A Regulatory Perspective. J Chem Pharm Res 2011; 3(3):6-17.
- https://www.academia.edu/24152922/Polymorphism_in_Pharmaceutical_Compounds?auto=download
- https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/physprop.htm
- Kadam K P and Chavan R P. Evaluation of Various Polymorphs by Different Techniques and Their Characterization A Review. The International Journal of Engineering and Science 2016; 5(6):29-34.
- Omar M, Makary P and Wlodarski M. A Review of Polymorphism and the Amorphous State in the Formulation Strategy of Medicines and Marketed Drugs. UK Journal of Pharmaceutical and Biosciences 2015; 3(6):60-6.